Chapter 1.
Introduction
The Wittig reaction is one of the most widely used methods for forming carbon-carbon double bonds, because it is easy to carry out and often gives high yields of pure product. It is named after its discoverer, German chemist Georg Wittig.1 The reaction involves the addition of a phosphorus ylide to an aldehyde or ketone to form a double bond with the elimination of phosphine oxide.
1. Wittig, G. Pure & Appl. Chem.1964, 9, 245–254
Reaction

Lab Objective
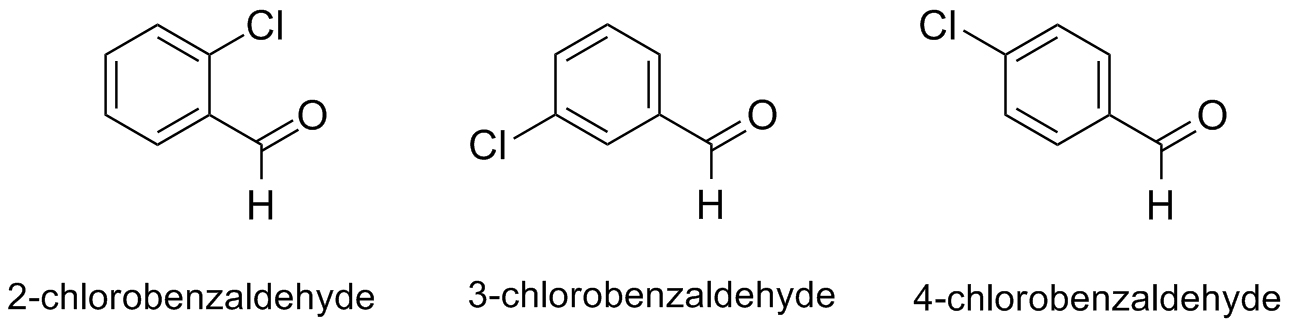
Ths is a group experiment. You will work in a small group to carry out variations of the Wittig reaction and to answer the focus question in the next tab. Each group will perform the Wittig reaction with commercially available ylide (carbethoxymethylene)triphenylphosphorane and one of the three aldehydes (2-, 3-, or 4-chlorobenzaldehyde) below. Be sure to complete the group design sheet.
Focus Question
Dichloromethane is typically used as a solvent in the Wittig reaction, because it can dissolve a wide variety of compounds. However, dichloromethane presents significant health hazards. What other solvents can be used effectively in the Wittig reaction?
Note: In selecting a greener alternative solvent there are several important factors to keep in mind. The alternative solvent must perform similarly to the solvent it replaces. Specifically, the reaction should proceed with similar efficiency, taking into account the solubility and its influence on reactivity and ease of purification. Furthermore, appreciable improvements in the toxicity of the solvent, its environmental impact, and potential for reducing exposure should also be considered.2 Potential available alternative solvents include diethyl ether, ethyl acetate, hexanes, petroleum ether, and water.
2. Doxsee, K.M. and Hutchison, J.E. “Green Organic Chemistry: Strategies, Tools, and Laboratory Experiments” Brooks/Cole: Toronto, Canada, 2004
Procedure
Dissolve chlorobenzaldehyde (50 mg) in dichloromethane (3 mL) in a dram vial equipped with a stir vane. Add 1.2 mol equivalents of the ylide (mol. wt. 348.38 g/mol) portion-wise while stirring. Stir at room temperature for two hours while monitoring the reaction by TLC. When the reaction is complete evaporate the dichloromethane solvent with a stream of N2 gas and dissolve the reaction mixture in 25% diethyl ether in hexanes (2–3 mL). Note the formation of a white precipitate, which is triphenylphosphine oxide. Transfer the solution to a clean vial and evaporate the majority of the solvent. Purify the crude product using a microscale wet column. Group members will work together to modify the purification procedure, as necessary, based on the alternative solvent selected, and to identify appropriate solvents for use in TLC and purification by microscale wet column chromatography.
Reagents
| Compound | CAS Number | mol. wt. (g mol-1) | Concentration or Density | m.p. or b.p. (°C) | SAFETY |
|---|---|---|---|---|---|
| 2-chlorobenzaldehyde | 89-98-5 | 140.57 | 1.248 g/mL | 209–215 (bp) | corrosive |
| 3-chlorobenzaldehyde | 587-04-2 | 140.57 | 1.241 g/mL | 213–214 (bp) | toxic |
| 4-chlorobenzaldehyde | 104-88-1 | 140.57 | -- | 213–214 (bp) | toxic |
| dichloromethane | 75-09-2 | 84.93 | 1.325 g/mL | 39.8–40 (bp) | health hazard |
| (carbethoxymethylene) triphenylphosphorane | 1099-45-2 | 348.37 | -- | 128–130 (mp) | -- |