PROBLEMS
PROBLEMS
Question 31.1
Missing genes. Predict the effects of deleting the following regions of DNA:
The gene encoding lac repressor
The lac operator
The gene encoding CAP
Question 31.2
Minimal concentration. Calculate the concentration of lac repressor, assuming that one molecule is present per cell. Assume that each E. coli cell has a volume of 10−12 cm3. Would you expect the single molecule to be free or bound to DNA?
939
Question 31.3
Counting sites. Calculate the expected number of times that a given 8-
Question 31.4
The same but not the same. The lac repressor and the pur repressor are homologous proteins with very similar three-
Question 31.5
The opposite direction. Some compounds called anti-
Question 31.6
Inverted repeats. Suppose that a nearly perfect 20-
Question 31.7
Broken operators. Consider a hypothetical mutation in OR2 that blocks both λ repressor and Cro binding. How would this mutation affect the likelihood of bacteriophage λ entering the lytic phase?
Question 31.8
Promoters. Compare the −10 and −35 sequences for the λ repressor and Cro genes in the right operator. How many differences are there between these sequences?
Question 31.9
Positive and negative feedback. What is the effect of an increased Cro concentration on the expression of the gene for the λ repressor? Of an increased concentration of λ repressor on the expression of the Cro gene? Of an increased concentration of λ repressor on the expression of the λ repressor gene?
Question 31.10
Leaderless. The mRNA for the λ repressor begins with 5′-AUG-
Question 31.11
Quorum count. Suppose you have a series of compounds that you wish to test for the autoinducer activity in Vibrio fischeri. Propose a simple assay, assuming that you can grow V. fischeri cultures at low cell densities.
Question 31.12
Codon utilization. There are four codons that encode threonine. Consider the leader sequence in Figure 31.22A What codons are used and with what frequency?
Mechanism Problem
Question 31.13
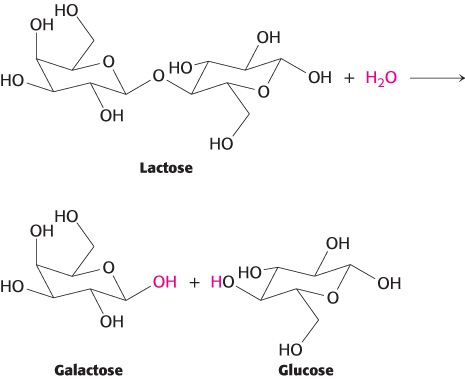
Follow the stereochemistry. The hydrolysis of lactose is catalyzed by β-galactosidase. Does the overall reaction proceed with retention or inversion of configuration? Given that each step likely proceeds with inversion of configuration, what does the overall change in stereochemistry suggest about the mechanism? A key residue in the reaction has been identified to be Glu 537. Propose an overall mechanism for the hydrolysis of lactose.
Data Interpretation Problems
Question 31.14
Leaving tracks. A powerful method for examining protein–
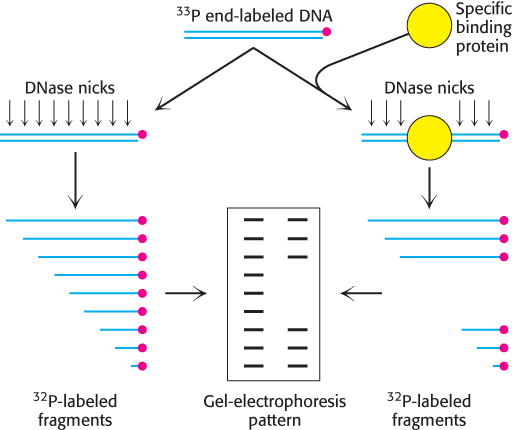
940
This method is applied to a DNA fragment containing a single binding site for the λ repressor in the presence of different concentrations of the λ repressor. The results are shown below (b):
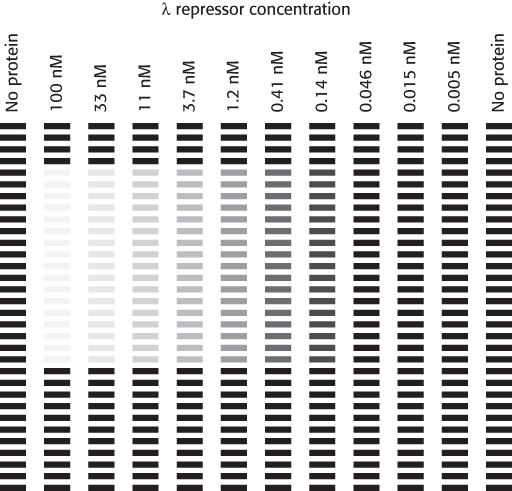
Estimate the dissociation constant for the λ repressor–