SUMMARY
SUMMARY
Enzymes adopt conformations that are structurally and chemically complementary to the transition states of the reactions that they catalyze. Sets of interacting amino acid residues make up sites with the special structural and chemical properties necessary to stabilize the transition state. Enzymes use five basic strategies to form and stabilize the transition state. The first (1) involves the use of binding energy to promote both specificity and catalysis. The others are (2) covalent catalysis, (3) general acid–
The cleavage of peptide bonds by chymotrypsin is initiated by the attack by a serine residue on the peptide carbonyl group. The attacking hydroxyl group is activated by interaction with the imidazole group of a histidine residue, which is, in turn, linked to an aspartate residue. This Ser-
Other proteases employ the same catalytic strategy. Some of these proteases, such as trypsin and elastase, are homologs of chymotrypsin. Other proteases, such as subtilisin, contain a very similar catalytic triad that has arisen by convergent evolution. Active-
Carbonic anhydrases catalyze the reaction of water with carbon dioxide to generate carbonic acid. The catalysis can be extremely fast: some carbonic anhydrases hydrate carbon dioxide at rates as high as 1 million times per second. A tightly bound zinc ion is a crucial component of the active sites of these enzymes. Each zinc ion binds a water molecule and promotes its deprotonation to generate a hydroxide ion at neutral pH. This hydroxide ion attacks carbon dioxide to form bicarbonate ion, 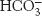 . Because of the physiological roles of carbon dioxide and bicarbonate ions, speed is of the essence for this enzyme. To overcome limitations imposed by the rate of proton transfer from the zinc-
. Because of the physiological roles of carbon dioxide and bicarbonate ions, speed is of the essence for this enzyme. To overcome limitations imposed by the rate of proton transfer from the zinc-
282
A high level of substrate specificity is often the key to biological function. Restriction endonucleases that cleave DNA at specific recognition sequences discriminate between molecules that contain these recognition sequences and those that do not. Within the enzyme–
Some enzymes discriminate between potential substrates by binding them with different affinities. Others may bind many potential substrates but promote chemical reactions efficiently only on specific molecules. Restriction endonucleases such as EcoRV endonuclease employ the latter mechanism. Only molecules containing the proper recognition sequence are distorted in a manner that allows magnesium ion binding and, hence, catalysis. Restriction enzymes are prevented from acting on the DNA of a host cell by the methylation of key sites within its recognition sequences. The added methyl groups block specific interactions between the enzymes and the DNA such that the distortion necessary for cleavage does not take place.
Finally, myosins catalyze the hydrolysis of adenosine triphosphate (ATP) to form adenosine diphosphate (ADP) and inorganic phosphate (Pi). The conformations of myosin ATPase domains free of bound nucleotides and with bound ATP are quite similar. Through the use of ADP and vanadate 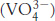 , an excellent mimic of the transition state for ATP hydrolysis bound to the myosin ATPase domain can be produced. The structure of this complex reveals that dramatic conformational changes take place on formation of this species from the ATP complex. These conformational changes are used to drive substantial motions in molecular motors. The rate of ATP hydrolysis by myosin is relatively low and is limited by the rate of product release from the enzyme. The hydrolysis of ATP to ADP and Pi within the enzyme is reversible with an equilibrium constant of approximately 10, compared with an equilibrium constant of 140,000 for these species free in solution. Myosins are examples of P-
, an excellent mimic of the transition state for ATP hydrolysis bound to the myosin ATPase domain can be produced. The structure of this complex reveals that dramatic conformational changes take place on formation of this species from the ATP complex. These conformational changes are used to drive substantial motions in molecular motors. The rate of ATP hydrolysis by myosin is relatively low and is limited by the rate of product release from the enzyme. The hydrolysis of ATP to ADP and Pi within the enzyme is reversible with an equilibrium constant of approximately 10, compared with an equilibrium constant of 140,000 for these species free in solution. Myosins are examples of P-
283