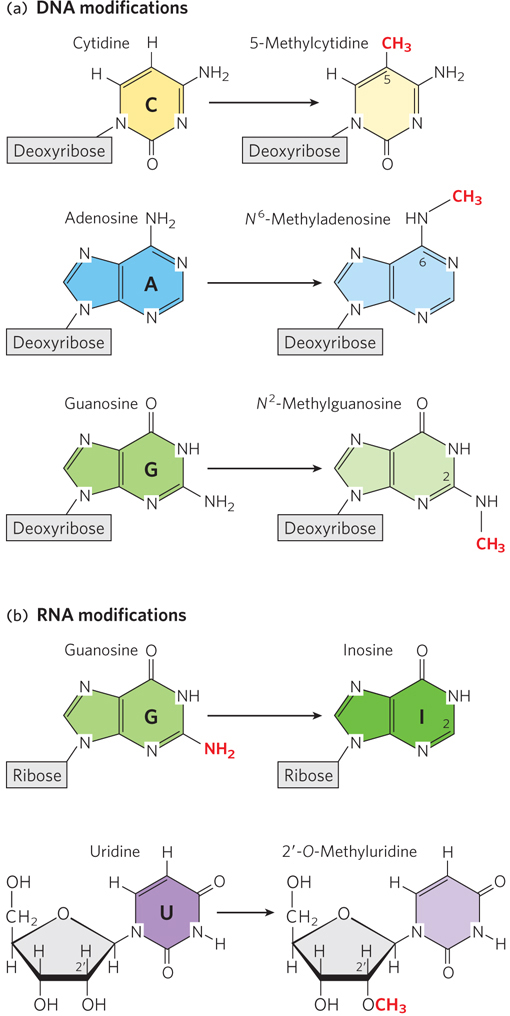
Chemical modification of nucleotide sugars and bases. (a) Examples of methylation modifications in DNA nucleotides. The extra methyl group on 5- methylcytidine, N6-methyladenosine, and N2-methylguanosine is highlighted in red. (b) Examples of modifications in RNA nucleotides. Loss of the amino group attached to the guanine ring (the exocyclic amine) gives rise to inosine; uridine can be methylated at the 2′ position to produce 2′-O-methyluridine. Modification sites are indicated in red.