Concept 2.3: Carbohydrates Consist of Sugar Molecules
Carbohydrates are a large group of molecules that all have similar atomic compositions but differ greatly in size, chemical properties, and biological functions. Carbohydrates usually have the general formula CmH2nOn, where m and n represent numbers. This makes them appear to be hydrates of carbon [associations between water molecules and carbon in the ratio Cm(H2O)n], hence their name. However, carbohydrates are not really “hydrates” because the water molecules are not intact. Rather, the linked carbon atoms are bonded with hydrogen atoms (—H) and hydroxyl groups (—OH), the components of water. Carbohydrates have four major biochemical roles:
- They are a source of stored energy that can be released in a form that organisms can use.
- They are used to transport stored energy within complex organisms.
- They function as structural molecules that give many organisms their shapes.
- They serve as recognition or signaling molecules that can trigger specific biological responses.
Some carbohydrates are relatively small, such as the simple sugars (for example, glucose) that are the primary energy source for many organisms. Others are large polymers of simple sugars; an example is starch, which is stored in seeds.
Monosaccharides are simple sugars
Monosaccharides (mono, “one”) are relatively simple molecules with up to seven carbon atoms. They differ in their arrangements of carbon, hydrogen, and oxygen atoms (FIGURE 2.9).
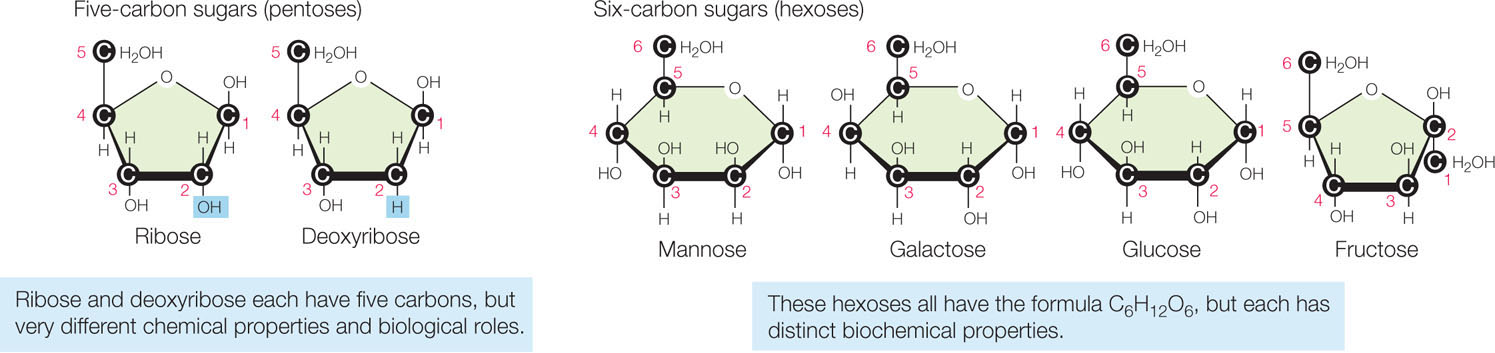
Pentoses (pente, “five”) are five-carbon sugars. Two pentoses are of particular biological importance: the backbones of the nucleic acids RNA and DNA contain ribose and deoxyribose, respectively.
LINK
For a description of the nucleic acids RNA and DNA see Concept 3.1
The hexoses (hex, “six”) all have the formula C6H12O6. They include glucose, fructose (so named because it was first found in fruits), mannose, and galactose.
Go to ACTIVITY 2.2 Forms of Glucose
PoL2e.com/ac2.2
Glycosidic linkages bond monosaccharides
The disaccharides, oligosaccharides, and polysaccharides are all constructed from monosaccharides that are covalently bonded by condensation reactions that form glycosidic linkages. A single glycosidic linkage between two monosaccharides forms a disaccharide. For example, sucrose—common table sugar—is a major disaccharide formed in plants from a glucose molecule and a fructose molecule (see Figure 2.9 for complete structures):
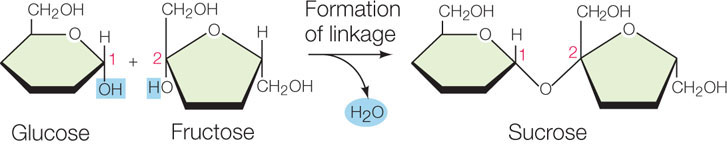
Another disaccharide is maltose, which is formed from two glucose units and is a product of starch digestion. Maltose is an important carbohydrate for making beer.
Oligosaccharides contain several monosaccharides bound together by glycosidic linkages. Many oligosaccharides have additional functional groups, which give them special properties. Oligosaccharides are often covalently bonded to proteins and lipids on the outer surfaces of cells, where they serve as recognition signals. For example, the different human blood groups (the ABO blood types) get their specificity from oligosaccharide chains.
Polysaccharides store energy and provide structural materials
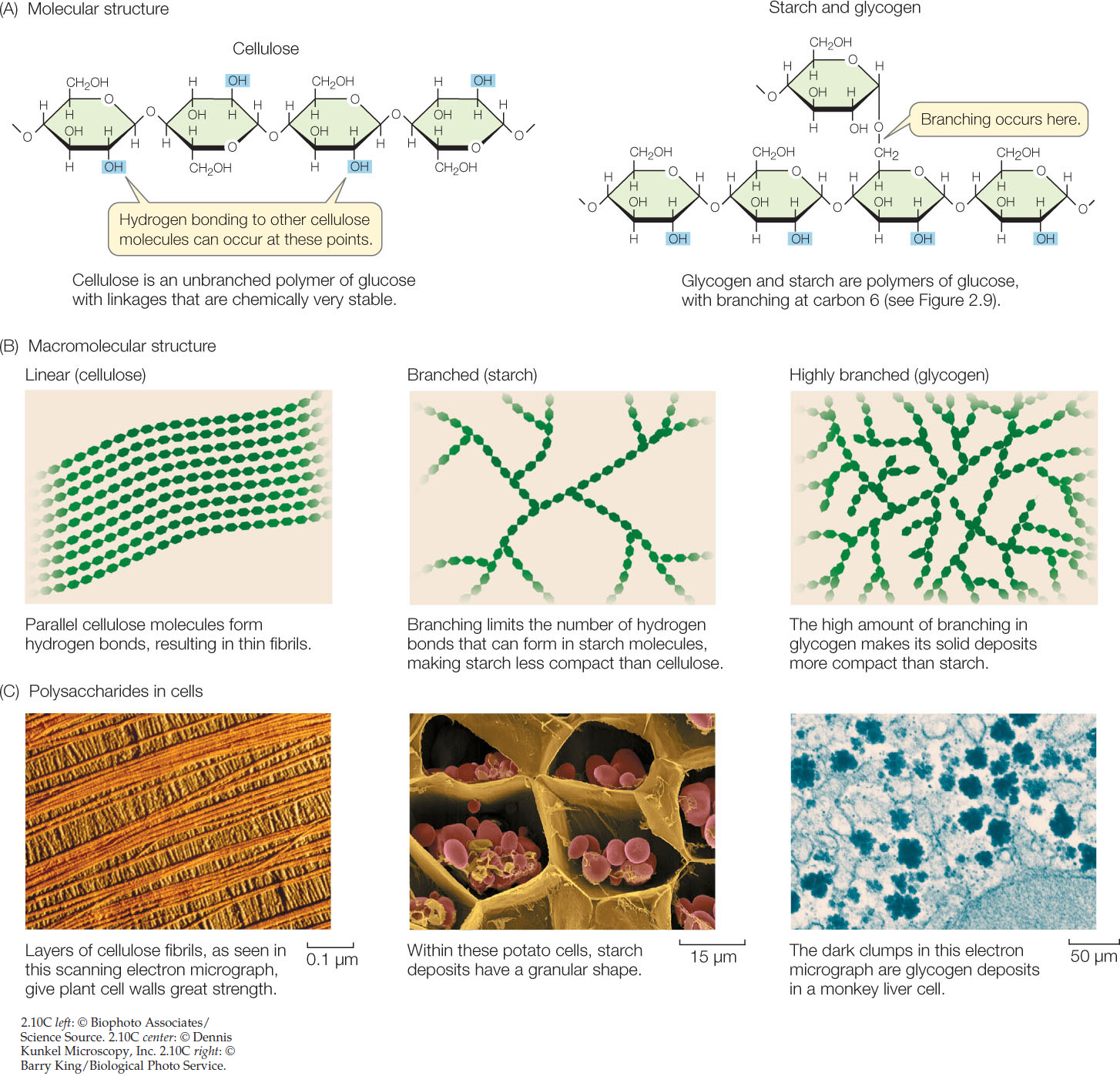
Polysaccharides are large polymers of monosaccharides connected by glycosidic linkages (FIGURE 2.10). Polysaccharides are not necessarily linear chains of monomers. Each monomer unit has several sites that are capable of forming glycosidic linkages, and thus branched molecules are possible.
28
Starches comprise a family of giant molecules that are all polysaccharides of glucose. The different starches can be distinguished by the amount of branching in their polymers. Starch is the principal energy storage compound of plants.
Glycogen is a water-insoluble, highly branched polymer of glucose that is the major energy storage molecule in mammals. It is produced in the liver and transported to the muscles. Both glycogen and starch are readily hydrolyzed into glucose monomers, which in turn can be broken down to liberate their stored energy.
If glucose is the major source of fuel, why store it in the form of starch or glycogen? The reason is that 1,000 glucose molecules would exert 1,000 times the osmotic pressure of a single glycogen molecule, causing water to enter the cells (see Concept 5.2). If it were not for polysaccharides, many organisms would expend a lot of energy expelling excess water from their cells.
As the predominant component of plant cell walls, cellulose is by far the most abundant carbon-containing (organic) compound on Earth. Like starch and glycogen, cellulose is a polysaccharide of glucose, but its glycosidic linkages are arranged in such a way that it is a much more stable molecule. Whereas starch is easily broken down by chemicals or enzymes to supply glucose for energy-producing reactions, cellulose is an excellent structural material that can withstand harsh environmental conditions without substantial change.
LINK
Most animals cannot digest (hydrolyze) cellulose; Chapter 30 describes adaptations in some animals to use cellulose as an energy source
29
CHECKpoint CONCEPT 2.3
- Draw the chemical structure of a disaccharide formed from two glucose monosaccharides.
- Examine the glucose molecule shown in Figure 2.9. What are the functional groups on the molecule?
- Notice the large number of hydrogen bonding groups present in the linear structure of cellulose (see Figure 2.10A). Why is this structure so strong?
- Some sugars have other functional groups in addition to those typically present. Draw the structure of the amino sugar glucosamine, which has an amino group bonded at carbon 2 of glucose. Would this molecule be more or less polar than glucose? Explain why.
We have seen that carbohydrate structure is an example of the monomer–polymer theme in biology. Now we will turn to lipids, which are unusual among the four classes of biological macromolecules in that they are not, strictly speaking, polymers.