Concept 6.1: ATP and Reduced Coenzymes Play Important Roles in Biological Energy Metabolism
In Chapters 2 and 3 we introduced the general concepts of energy, enzymes, and metabolism. Energy is stored in the chemical bonds of molecules, and it can be released and transformed by the metabolic pathways of living cells. There are five general principles governing metabolic pathways:
- A complex chemical transformation occurs in a series of separate, intermediate reactions that form a metabolic pathway.
- Each reaction is catalyzed by a specific enzyme.
- Most metabolic pathways are similar in all organisms, from bacteria to plants to humans.
- In eukaryotes, many metabolic pathways are compartmentalized, with certain reactions occurring inside specific organelles.
- Each metabolic pathway is controlled by key enzymes that can be inhibited or activated, thereby determining how fast the reactions will go.
Chemical energy available to do work is termed free energy (G). According to the laws of thermodynamics, a biochemical reaction may change the form of energy but not the net amount. A biochemical reaction is exergonic if it releases energy from the reactants, or endergonic if energy must be added to the reactants.
LINK
You can review the principles of energy transformations in Concept 2.5
In the chemistry lab, energy can be released or added in the form of heat. But in cells, energy-transforming reactions are often coupled; that is, an energy-releasing (exergonic) reaction is coupled in time and location to an energy-requiring (endergonic) reaction. Two widely used coupling molecules are the coenzymes ATP and NADH.
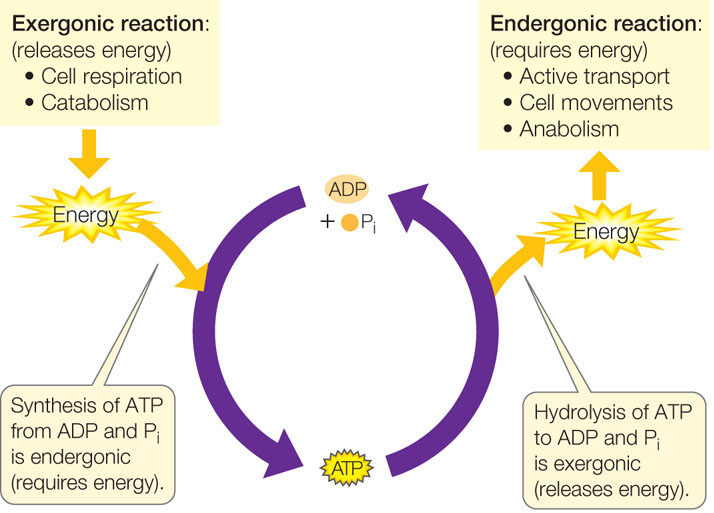
ATP hydrolysis releases energy
Cells use adenosine triphosphate (ATP) as a kind of “energy currency.” Just as it is more effective, efficient, and convenient for you to trade money for a lunch than to trade your actual labor, it is useful for cells to have a currency for transferring energy between different reactions and cell processes. Some of the energy that is released in exergonic reactions is captured in chemical bonds when ATP is formed from adenosine diphosphate (ADP) and inorganic phosphate (hydrogen phosphate; commonly abbreviated as Pi). The ATP can then be hydrolyzed at other sites in the cell, releasing free energy to drive endergonic reactions (FIGURE 6.1).
Go to ACTIVITY 6.1 ATP and Coupled Reactions
PoL2e.com/ac6.1
An active cell requires the production of millions of molecules of ATP per second to drive its biochemical machinery. You are already familiar with some of the activities in the cell that require free energy derived from the hydrolysis of ATP:
- Active transport across a membrane (Concept 5.3)
- Condensation reactions that use enzymes to form polymers (Concept 2.2)
- Motor proteins that move vesicles along microtubules (Concept 4.4)
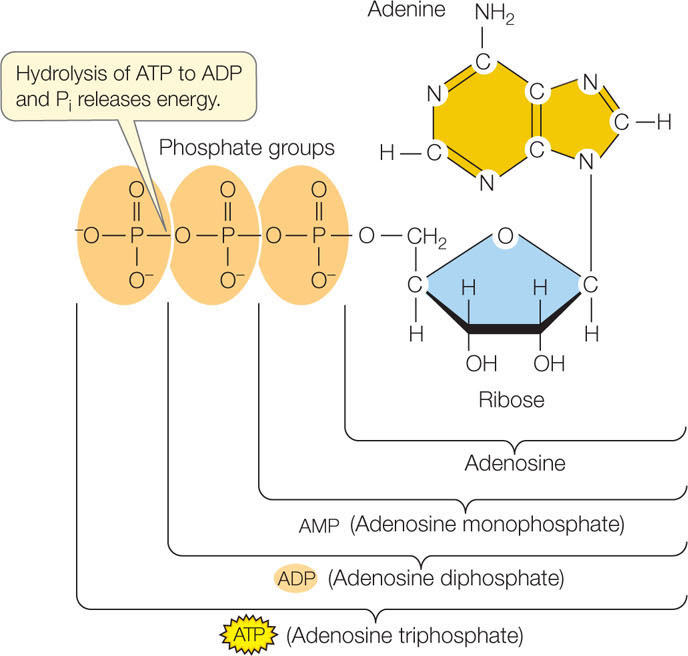
An ATP molecule consists of the nitrogen-containing base adenine bonded to ribose (a sugar), which is attached to a sequence of three phosphate groups (FIGURE 6.2). The hydrolysis of a molecule of ATP yields free energy, ADP, and the inorganic phosphate ion (Pi):
ATP + H2O → ADP + Pi + free energy
107
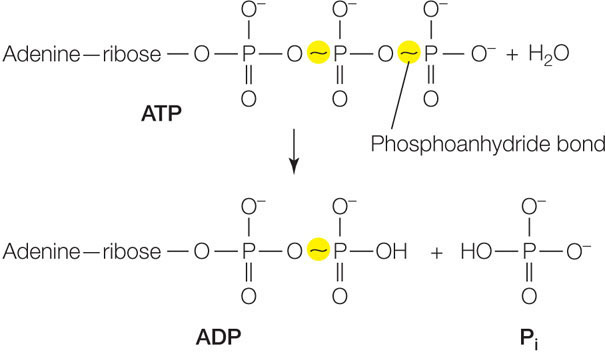
The important property of this reaction is that it is exergonic, releasing free energy. Under standard laboratory conditions, the change in free energy for this reaction (ΔG) is about −7.3 kcal/mol (−30 kJ/mol). Recall that a negative change in free energy means that the product molecules (in this case, ADP and Pi) have less free energy than the reactants (ATP and H2O), so the change is negative. A molecule of ATP can also be hydrolyzed to adenosine monophosphate (AMP) and a pyrophosphate ion (P2O74−; commonly abbreviated as PPi). In this case, additional energy may be released by the subsequent conversion of PPi to two molecules of Pi.
Energy is released as a result of ATP hydrolysis because the P—O bonds in a free hydrogen phosphate (Pi) molecule are stronger and more stable than the relatively weak P—O bonds (called phosphoanhydride bonds) between the phosphate groups in ATP. (Phosphoanhydride bonds are often denoted by wavy lines in chemical structures, as highlighted below). Recall that in general, stable bond formation is an exergonic process, whereas breaking bonds requires an input of energy. In this case, the amount of energy released by the formation of a new P—O bond in the Pi molecule is greater than the energy needed to break the phosphoanhydride bond.
In some reactions, ATP is formed by substrate-level phosphorylation—the enzyme-mediated direct transfer of phosphate from another molecule (the substrate) to ADP. This is the case for some reactions of glycolysis, as we will see in Concept 6.2. But most of the ATP in living cells is formed by oxidative phosphorylation, which we will discuss shortly.
Redox reactions transfer electrons and energy
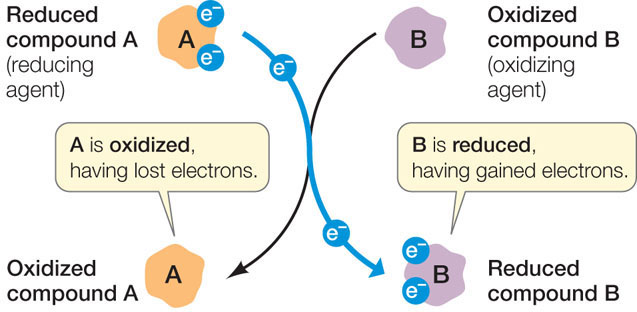
Another way of transferring energy in chemical reactions is to transfer electrons. A reaction in which one substance transfers one or more electrons to another substance is called a reduction–oxidation reaction, or redox reaction.
- Reduction is the gain of one or more electrons by an atom, ion, or molecule.
- Oxidation is the loss of one or more electrons.
Oxidation and reduction always occur together: as one chemical is oxidized, the electrons it loses are transferred to another chemical, reducing it. Thus some molecules are called oxidizing agents and others are reducing agents:
Although oxidation and reduction are defined in terms of traffic in electrons, it is often helpful to think in terms of the gain or loss of hydrogen atoms. Transfers of hydrogen atoms involve transfers of electrons (H = H+ + e−). So when a molecule loses a hydrogen atom, it becomes oxidized.
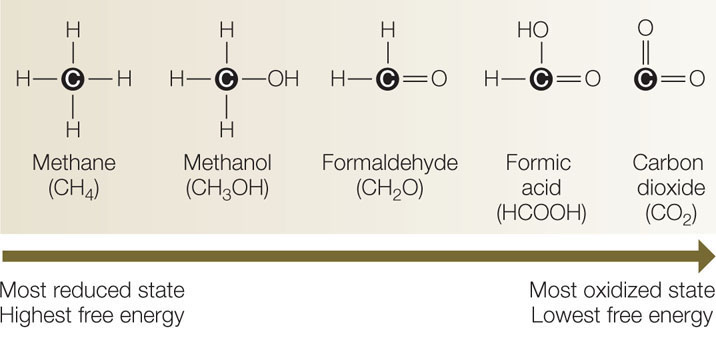
In general, the more reduced a molecule is, the more energy is stored in its covalent bonds (FIGURE 6.3). Indeed, highly reduced molecules can be used as energy sources; for example, methane and methanol can be burned as fuel. However, oxidized molecules such as CO2 cannot be used as sources of energy. In a redox reaction, some energy is transferred from the reducing agent to the reduced product. Some energy remains in the reducing agent (now oxidized), and some is lost to entropy.
Cells use the coenzyme nicotinamide adenine dinucleotide (NAD) as an electron carrier in redox reactions (FIGURE 6.4). NAD exists in two chemically distinct forms, one oxidized (NAD+) and the other reduced (NADH). The reduction reaction
NAD+ + H+ + 2 e− → NADH
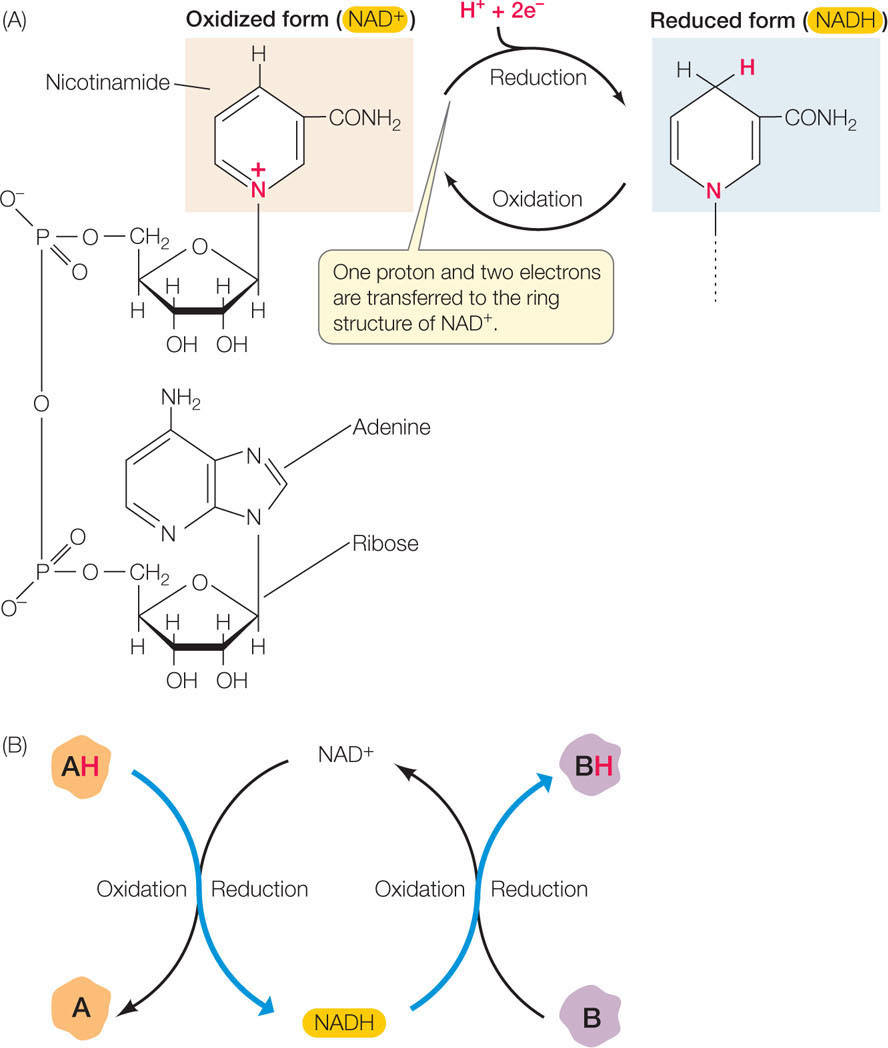
involves the transfer of a proton (the hydrogen ion, H+) and two electrons, which are released by an accompanying oxidation reaction.
The reduction of NAD+ is highly endergonic, and within the cell, the electrons do not remain with NADH. Oxygen is highly electronegative and readily accepts electrons from the reduced NADH molecule. The oxidation of NADH by O2 (which occurs in several steps):
NADH + H+ + ½ O2 → NAD+ + H2O
108
is highly exergonic, releasing energy with a ΔG of −52.4 kcal/mol (−219 kJ/mol). Note that the oxidizing agent appears here as “½ O2” instead of “O.” This notation emphasizes that it is molecular oxygen (O2) that acts as the oxidizing agent. This is clearer if the molecules of the reaction above are doubled:
2 NADH + 2 H+ + O2 → 2 NAD+ + 2 H2O
Because the oxidation of NADH releases more energy than the hydrolysis of ATP, NADH can be thought of as a larger package of free energy than ATP. NAD+ is a common electron carrier in cells, but not the only one. Others include flavin adenine dinucleotide (FAD), which also transfers electrons during glucose metabolism (see Concept 6.2), and nicotinamide adenine dinucleotide phosphate (NADP+), which is used in photosynthesis (see Concept 6.5).
The processes of NADH oxidation and ATP production are coupled
In order to carry out the many metabolic processes needed to sustain life, cells release and reuse the energy contained in chemical bonds. The energy-coupling coenzymes (in particular, ATP and NADH) play vital roles in the transfer of energy between cellular reactions that release energy (catabolism) and those that require energy (including anabolism).
LINK
See Concept 2.5 to review the principles of catabolism and anabolism
How do these coenzymes participate in the flow of energy within cells? The release and reuse of cellular energy can be summarized as follows:
- Catabolism releases energy by oxidation; this energy can be trapped by the reduction of coenzymes such as NAD+.
- ATP supplies the energy for many energy-requiring processes, including anabolism. For example, as we noted in Chapter 5, active transport requires ATP.
In other words, most of the energy-releasing reactions in the cell produce NADH (or similar reduced coenzymes), but most of the energy-consuming reactions require ATP. Cells need a way to connect the two coenzymes; that is, to transfer energy from NADH to the phosphoanhydride bond of ATP. This transfer is accomplished in a process called oxidative phosphorylation—the coupling of NADH oxidation to the production of ATP. We will discuss the mechanisms of this process in Concept 6.2.
CHECKpoint CONCEPT 6.1
- For each of the reactions
- C6H12O6 + 6 O2 → 6 CO2 + 6 H2O
- 6 CO2 + 6 H2O → C6H12O6 + 6 O2
- What kinds of coenzymes might be involved in the following reactions? Explain your answer.
- Glucose → glucose 6-phosphate
- Fatty acid → CO2 + H2O
- A typical, active young man requires 2,800 kilocalories of food energy a day to fuel metabolism, movement, active transport, etc. The energy stored in the third phosphodiester bond of ATP is 0.0145 kcal/gram.
- If the energy from the man’s food were all stored as ATP, how much ATP would be produced each day from ADP and Pi?
- The man actually has about 50 grams of ATP. What does this mean in terms of ATP hydrolysis and synthesis?
In this concept we saw that both ATP and NADH function as energy-coupling coenzymes, which are used by cells to store and transfer energy. We will now look at how cells capture energy from the catabolism of glucose to produce NADH, and then transfer this energy from NADH to ATP.
109