CONCEPT6.4 Catabolic and Anabolic Pathways Are Integrated
Metabolic transformations are a hallmark of life. The pathways we have seen thus far in the chapter, including glycolysis and the citric acid cycle, do not operate in isolation. Rather, there is an interchange of molecules into and out of these pathways, to and from the metabolic pathways for the synthesis and breakdown of amino acids, nucleotides, fatty acids, and other building blocks of life. Carbon skeletons (a term describing molecules with covalently linked carbon atoms) can enter catabolic pathways and be oxidized to release their energy, or they can enter anabolic pathways to be used in the formation of the macromolecules that are the major constituents of the cell. These relationships are summarized in FIGURE 6.13 and comprise a metabolic system (see Figure 3.17). The inputs and outputs of this system can be described, and predictions can be made regarding what would happen if the concentration of one component changes.
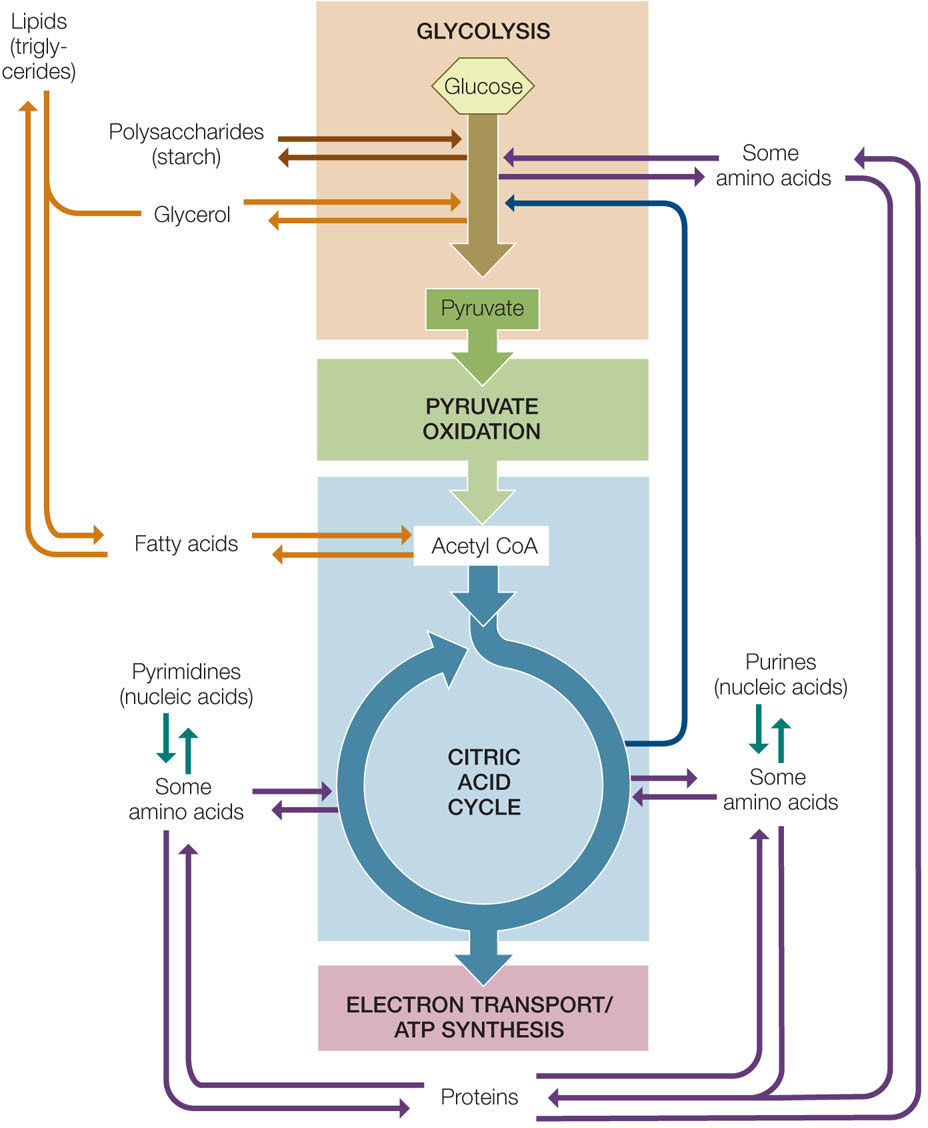
Catabolism and anabolism are linked
A hamburger or veggie burger on a bun contains three major sources of carbon skeletons: carbohydrates, mostly in the form of starch (a polysaccharide); lipids, mostly as triglycerides (three fatty acids attached to glycerol); and proteins (polymers of amino acids). Look at Figure 6.13 to see how each of these three types of macromolecules can be hydrolyzed and used in catabolism or anabolism.
Catabolic Interconversions
Polysaccharides, lipids, and proteins can all be broken down to provide energy:
- Polysaccharides can be hydrolyzed to glucose. Glucose then passes through glycolysis, pyruvate oxidation, and the respiratory chain, where its energy is captured in ATP.
- Lipids are broken down into their constituents—glycerol and fatty acids. Glycerol is converted into dihydroxyacetone phosphate, an intermediate in glycolysis. Fatty acids are highly reduced molecules that are converted to acetyl CoA in a process called β-oxidation, catalyzed by a series of oxidation enzymes inside the mitochondrion. For example, the β-oxidation of a C16 (16-carbon) fatty acid occurs in several steps:
C16 fatty acid + CoA → C16 fatty acyl CoA
C16 fatty acyl CoA + CoA → C14 fatty acyl CoA + acetyl CoA
(Repeat 6 times) → 8 acetyl CoA
The acetyl CoA can then enter the citric acid cycle and be catabolized to CO2. - Proteins are hydrolyzed to their amino acid building blocks. The 20 different amino acids feed into glycolysis or the citric acid cycle at different points. For example, the amino acid glutamate is converted into α-ketoglutarate, an intermediate in the citric acid cycle.
Anabolic Interconversions
Many catabolic pathways can operate essentially in reverse, with some modifications. Glycolytic and citric acid cycle intermediates, instead of being oxidized to form CO2, can be reduced and used to form glucose in a process called gluconeogenesis (which means “new formation of glucose”). Likewise, acetyl CoA can be used to form fatty acids. The most common fatty acids have even numbers of carbons: 14, 16, or 18. These are formed by the addition of two-carbon acetyl CoA “units” one at a time until the appropriate chain length is reached.
Some intermediates in the citric acid cycle are reactants in pathways that synthesize important components of nucleic acids. For example, α-ketoglutarate and oxaloacetate are starting points for purines and pyrimidines, respectively.
Catabolism and anabolism are integrated into a system
A carbon atom from a protein in your burger can end up in DNA, fat, or CO2, among other fates. How does the organism “decide” which metabolic pathways to follow, in which cells? With hundreds of enzymes and all the possible interconversions, you might expect that the cellular concentrations of various biochemical molecules would fluctuate widely. Remarkably, the levels of these substances in what is called the metabolic pool—the sum total of all the biochemical molecules in a cell—are usually quite constant. Metabolic changes in the cell are a bit like the changes in traffic patterns in a city: if an accident blocks traffic on a major road, drivers take alternate routes, where the traffic volume consequently changes.
Consider what happens to the starch in your burger bun. In the digestive system, starch is hydrolyzed to glucose, which enters the blood. If it is needed, the glucose is distributed to the rest of the body. But if there is already enough glucose in the blood to supply the body’s needs, the excess glucose is converted into glycogen and stored in the liver. If not enough glucose is supplied by food, glycogen is broken down, or other molecules are used to make glucose by gluconeogenesis. The end result is that the level of glucose in the blood is remarkably constant. How does the body accomplish this?
Metabolic enzymes (including those of glycolysis, the citric acid cycle, and the respiratory chain) are subject to regulation, and often the regulatory mechanisms involve allosteric effects. An example is feedback inhibition, illustrated in Figure 3.21. In a metabolic pathway, a high concentration of the final product can inhibit the enzyme that catalyzes the commitment step. For example, the product of glucose catabolism, ATP, feedback inhibits key enzymes in both glycolysis and the citric acid cycle. Sometimes feedback involves the product of one pathway speeding up reactions in another pathway. Feedback regulation generally occurs rapidly, affecting a pathway within minutes.
LINK
Review the discussion of enzyme regulation in Concept 3.4
The rate of a biochemical reaction can also be controlled by reducing or increasing the number of enzyme molecules present relative to substrate (see Figure 11.6). This can be done by altering the transcription of the genes that encode the enzymes. These events take time, and typically the effects on metabolism will take days to appear. For example, excess levels of glucose and other dietary factors can lead to increased transcription of the gene for fatty acid synthase, a key enzyme in the synthesis of fatty acids. Excess citrate produced by the citric acid cycle is broken down to acetyl CoA, which in turn is used in fatty acid synthesis. This is one reason why people accumulate fat after eating too much. The fatty acids may be catabolized later to produce more acetyl CoA.
LINK
The regulation of gene transcription is described in Chapter 11
ATP and reduced coenzymes link catabolism, anabolism, and photosynthesis
Thus far in this chapter we have discussed the major catabolic pathway called cellular respiration, in which a reduced molecule, the carbohydrate glucose, is oxidized, often all the way to CO2:
Glucose + 6 O2 → 6 CO2 + 6 H2O + chemical energy
We have also seen how the energy derived from cellular respiration is transferred between catabolic and anabolic pathways (depending on a cell’s needs), and that the coenzymes ATP and NADH play key roles in these energy transfers.
But where does the energy for all these processes come from? As we will see in the next two concepts, almost all living organisms on Earth ultimately derive their energy from the sun (exceptions include organisms that derive energy from geothermal vents deep within oceans). In addition to cellular respiration, green plants, algae, some protists, and some prokaryotes also carry out photosynthesis—a major anabolic pathway that converts light energy into chemical energy in the form of carbohydrates:
CO2 + H2O + light energy → O2 + carbohydrates
Just as catabolism and anabolism are linked, cellular respiration and photosynthesis are also linked—not only by their reactants and products (O2, CO2, and carbohydrates), but also by the energy “currency” of ATP and reduced coenzymes (FIGURE 6.14). The pathways of cellular respiration and photosynthesis can occur in the same cell. For example, cells in green plants often carry out both photosynthesis and cellular respiration simultaneously.
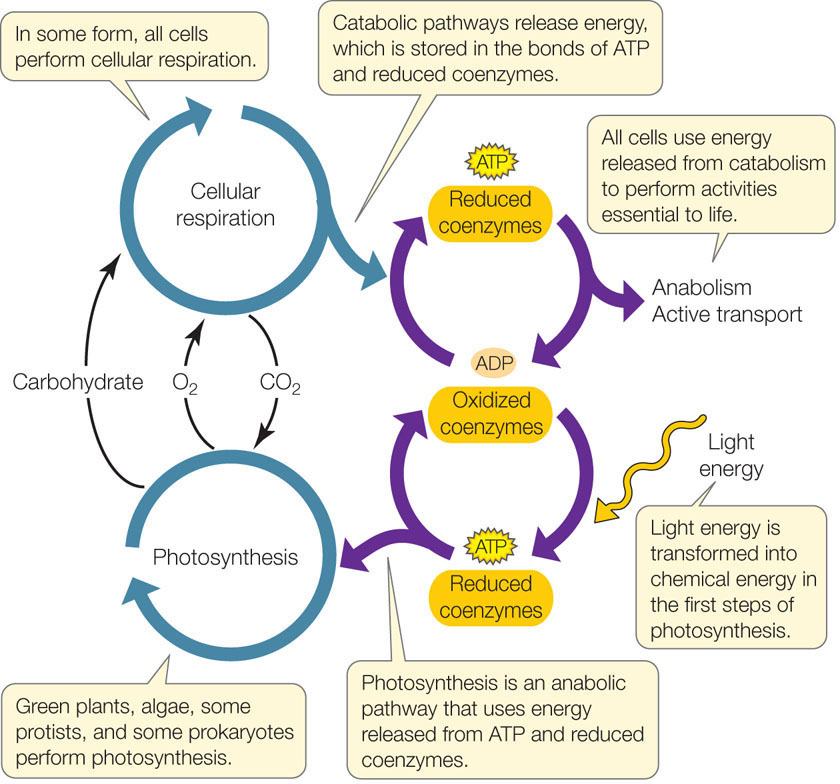
More often, however, cells do not carry out photosynthesis for themselves. Even in a green plant, a root cell is not photosynthetic and relies on carbohydrates transported from the leaf to carry out cellular respiration. Humans do not carry out photosynthesis anywhere in their bodies; they rely on carbohydrates obtained in their diet (ultimately derived from photosynthesis) to carry out cellular respiration, which provides the chemical energy for their bodies’ activities, such as active transport and anabolism.
CHECKpointCONCEPT6.4
- Give examples of the catabolic conversion of a lipid and the anabolic conversion of a protein.
- Trace the biochemical pathway by which a carbon atom from a starch molecule in rice eaten today can end up in a muscle protein tomorrow.
- Describe what might happen if there were no mechanisms for modulating the level of acetyl CoA.
We have seen how cellular respiration allows organisms to harvest chemical energy from organic molecules. For the rest of the chapter, we’ll look at how plants and other photosynthetic organisms produce these organic molecules using energy from light.