Concept 18.1: Events in Earth’s History Can Be Dated
Some evolutionary changes happen rapidly enough to be studied directly and manipulated experimentally. Plant and animal breeding by agriculturalists and evolution of resistance to pesticides are examples of rapid, short-term evolution that we saw in Chapter 15. Other evolutionary changes, such as the appearance of new species and evolutionary lineages, usually take place over much longer time scales.
To understand long-term patterns of evolutionary change, we must not only think in time scales spanning many millions of years, but also consider events and conditions very different from those we observe today. Earth of the distant past was so unlike our present Earth that it would seem like a foreign planet inhabited by strange organisms. The continents were not where they are now, and climates were sometimes dramatically different from those of today. We know this because much of Earth’s history is recorded in its rocks.
We cannot tell the ages of rocks just by looking at them, but we can visually determine the ages of rocks relative to one another. The first person to formally recognize this fact was the seventeenth-century Danish physician Nicolaus Steno. Steno realized that in undisturbed sedimentary rocks (rocks formed by the accumulation of sediments), the oldest layers of rock, or strata (singular stratum), lie at the bottom, and successively higher strata are progressively younger.
Geologists subsequently combined Steno’s insight with their observations of fossils contained in sedimentary rocks. They developed the following principles of stratigraphy:
- Fossils of similar organisms are found in widely separated places on Earth.
- Certain fossils are always found in younger strata, and certain other fossils are always found in older strata.
- Organisms found in younger strata are more similar to modern organisms than are those found in older strata.
These patterns revealed much about the relative ages of sedimentary rocks and the fossils they contain, as well as patterns in the evolution of life. But the geologists still could not tell how old particular rocks were. A method of dating rocks did not become available until after radioactivity was discovered at the beginning of the twentieth century.
Radioisotopes provide a way to date rocks
Radioactive isotopes of atoms—radioisotopes—decay in a predictable pattern over long periods. Over a specific time interval, known as a half-life, half of the atoms in a radioisotope decay to become a different, stable (nonradioactive) isotope (FIGURE 18.1A). The use of this knowledge to date fossils and rocks is known as radiometric dating.
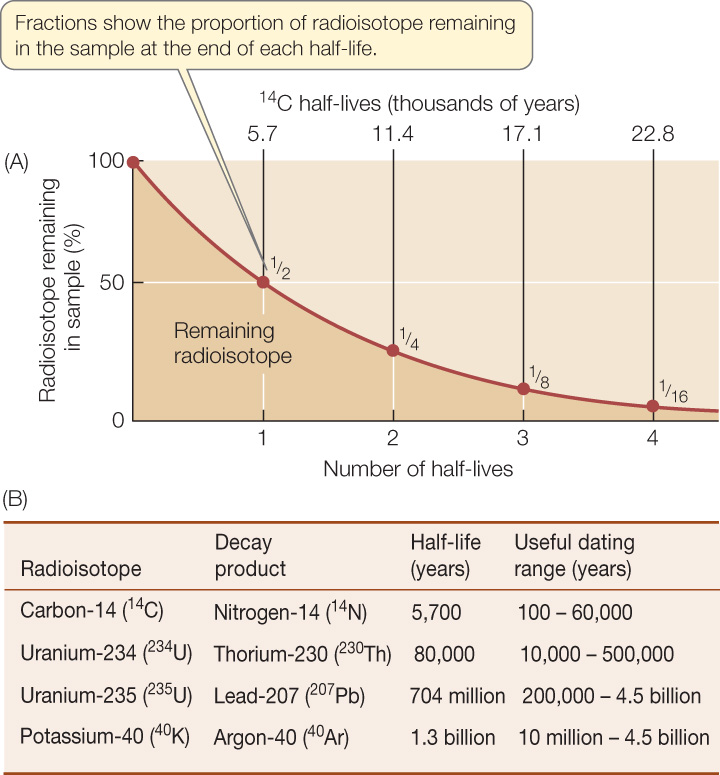
To use a radioisotope to date a past event, we must know or estimate the concentration of that isotope at the time of that event, and we must know the radioisotope’s half-life. In the case of carbon-14, a radioisotope of carbon, the production of new carbon-14 (14C) in the upper atmosphere—by the reaction of neutrons with nitrogen-14 (14N, a stable isotope of nitrogen)—just balances the natural radioactive decay of 14C into 14N. Therefore the ratio of 14C to the more common stable isotope of carbon, carbon-12 (12C), is relatively constant in living organisms and in their environment. As soon as an organism dies, however, it ceases to exchange carbon compounds with its environment. Its decaying 14C is no longer replenished, and the ratio of 14C to 12C in its remains decreases over time. Paleontologists can use the ratio of 14C to 12C in fossil material to date fossils that are less than 60,000 years old (and thus the sedimentary rocks that contain those fossils). If fossils are older than that, so little 14C remains that the limits of detection using this particular isotope are reached.
Radiometric dating methods have been expanded and refined
Sedimentary rocks are formed from materials that existed for varying lengths of time before being weathered, fragmented, and transported, sometimes over long distances, to the site of their deposition. Therefore the radioisotopes in sedimentary rock do not contain reliable information about the date of its formation. Radiometric dating of rocks older than 60,000 years requires estimating radioisotope concentrations in igneous rocks, which are formed when molten material cools. To date sedimentary strata, geologists search for places where volcanic ash or lava flows have intruded into the sedimentary rock.
359
APPLY THE CONCEPT: Events in Earth’s history can be dated
Imagine you have been assigned the job of producing a geological map of volcanic rocks that were formed between 400 and 600 million years ago. You collect samples from ten sites (1–10 on the map below). Then you can determine the ratio of 206Pb to 238U for each sample and use these ratios to estimate the ages of the rock samples, as given in the table below. Use the data to answer the following questions.

- Use the table shown here and Table 18.1 to assign each sample to a geological period.
- Use these estimated ages and geological periods of the samples to mark rough boundaries between the geological periods among the sample locations on the map below.
- If you wanted to refine the boundary between the Ordovician and Silurian on your map, which of three new sampling sites—x, y, or z—would you add to your analysis next?

A preliminary estimate of the age of an igneous rock determines which radioisotopes can be used to date it (FIGURE 18.1B). The decay of potassium-40 (which has a half-life of 1.3 billion years) to argon-40, for example, has been used to date many of the ancient events in the evolution of life. Fossils in the adjacent sedimentary rock that are similar to those in other rocks of known ages provide additional clues to the rock’s age.
Scientists have used several methods to construct a geological time scale
Radiometric dating of rocks, combined with fossil analysis, is the most powerful method of determining geological age. But in places where sedimentary rocks do not contain suitable igneous intrusions and few fossils are present, paleontologists turn to other dating methods.
One method, known as paleomagnetic dating, relates the ages of rocks to patterns in Earth’s magnetism, which change over time. Earth’s magnetic poles move and occasionally reverse themselves. Both sedimentary and igneous rocks preserve a record of Earth’s magnetic field at the time they were formed, and that record can be used to determine the ages of those rocks. Other dating methods use information about continental drift, information about sea level changes, and molecular clocks.
LINK
Molecular clocks are described in Concept 16.3
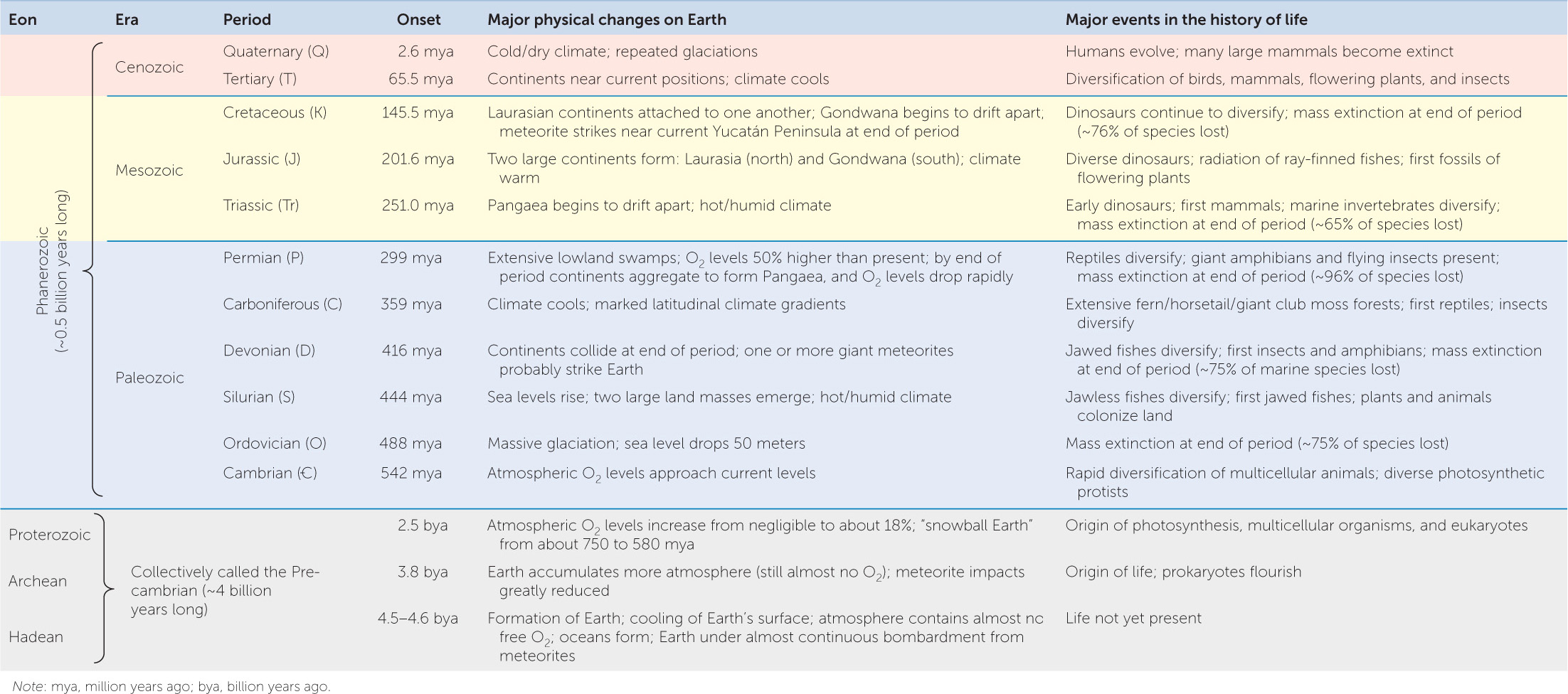
Using all of these methods, geologists developed a geological time scale (TABLE 18.1). They divided the broad history of life into four eons. The Hadean eon refers to the time on Earth before life evolved. The early history of life occurred in the Archean eon, which ended about the time that photosynthetic organisms first appeared on Earth. Prokaryotic life diversified rapidly in the Proterozoic eon, and the first eukaryotes in the fossil record date from this time. These three eons are sometimes referred to collectively as Precambrian time, or simply the Precambrian. The Precambrian lasted for approximately 3.8 billion years and thus accounts for the vast majority of geological time. It was in the Phanerozoic eon, however—a mere 542-million-year time span—that multicellular eukaryotes rapidly diversified. To emphasize the events of the Phanerozoic, Table 18.1 shows the subdivision of this eon into eras and periods. The boundaries between these divisions of time are based largely on the striking differences geologists observe in the assemblages of fossil organisms contained in successive strata. This geological record of life reveals a remarkable story of a world in which the continents and biological communities are constantly changing.
360
CHECKpoint CONCEPT 18.1
- What observations about fossils suggested to geologists that they could be used to determine the relative ages of rocks?
- Why are radioisotopes not measured directly from sedimentary rocks to determine their ages?
- Given the problems of dating sedimentary rocks using radioisotopes, what other methods can geologists use to date sedimentary rocks?
As geologists began to develop accurate ways to age Earth, they began to understand that Earth is far older than anyone had previously understood. During its 4.5-billion-year history, Earth has undergone massive physical changes. These changes have influenced the evolution of life, and life, in its turn, has influenced Earth’s physical environment.