Concept 29.1: Animals Eat to Obtain Energy and Chemical Building Blocks
Animals are heterotrophs—organisms that require preformed organic molecules as food (see Concept 45.3). They obtain their energy by breaking the chemical bonds of organic compounds obtained from other organisms, and they build their tissues from preexisting organic compounds obtained from other organisms. Animals, in other words, eat organic matter, such as starch or fat, that other organisms synthesized, and they use that organic matter as their source of both energy and chemical building blocks.
In contrast, autotrophs obtain energy from inorganic sources and can synthesize organic compounds from inorganic precursors. Plants are examples of autotrophs. They obtain energy from sunlight and synthesize organic compounds, such as the sugar glucose, principally from H2O and CO2.
In most ecosystems, sunlight is the primary energy input. Plants and algae use some of the light energy in sunlight to carry out photosynthesis and build organic compounds from simple precursors. In the process, a small fraction of the light energy in sunlight is incorporated into the chemical bonds of the organic compounds that plants and algae make. Animals then ingest the organic compounds in the plant or algal material and use them as their sources of energy and chemical building blocks.
LINK
The flow of energy through ecological communities is discussed in Concept 44.3
Animals need chemical building blocks to grow and to replace chemical constituents throughout life
Why does an animal need the chemical building blocks it obtains by eating? One reason is growth. When an animal grows during its early development, it requires chemical building blocks—such as fatty acids and amino acids—to build each new cell it adds to its body.
Even after they are fully grown, animals need chemical building blocks throughout their lives. This important fact was discovered by using isotopic forms of carbon atoms and nitrogen atoms (see Concept 2.1 for a discussion of isotopes). Suppose you make some food in which the nitrogen atoms are nitrogen-15 instead of the usual isotope, nitrogen-14. Suppose, then, that you feed that food to a full-grown dog or other animal. After a few weeks you will find that many of the nitrogen-14 atoms in the dog’s body have been replaced with nitrogen-15 atoms—even though the dog looks no different. If you then start to feed the dog regular food (with nitrogen-14 atoms) again, you will find that the nitrogen-15 atoms in the dog’s body are gradually replaced with nitrogen-14 atoms.
Experiments such as this demonstrate that the atoms inside an adult animal’s body are constantly exchanged with atoms in the animal’s surrounding environment. For example, our red blood cells live for only about 4 months. When a red blood cell is at the end of its life span, it is broken down and replaced. As it breaks down, some of its constituents are excreted. For example, some of the iron from the cell is excreted. To replace the cell, our bodies need to get replacement constituents—such as replacement iron—from our environment.
Nearly all the cells in our body are engaged in the process of breakdown and replacement (although not necessarily on a 4-month schedule). As the process takes place, some atoms originally inside our body are lost, and some atoms are taken in from the environment. In humans, 2–3 percent of body protein is broken down and rebuilt every day! In the process, some of the amino acids in the initial proteins are lost and must be replaced. For this reason, we need to eat amino acid–containing foods all our lives, even after we are fully grown.
We have seen that individual atoms constantly come and go in an animal. Over time, therefore, an animal is not defined by its atomic building blocks. Instead it is defined by the organization of its body.
Animals need inputs of chemical-bond energy to maintain their organized state throughout life
Organization is the most essential attribute of life. Animals are organized at multiple levels. In individual cells, atoms are organized into molecules such as phospholipids and proteins. In turn, these individual molecules are organized into cellular structures such as the cell membrane and into cellular biochemical processes such as sequences of enzyme-catalyzed reactions.
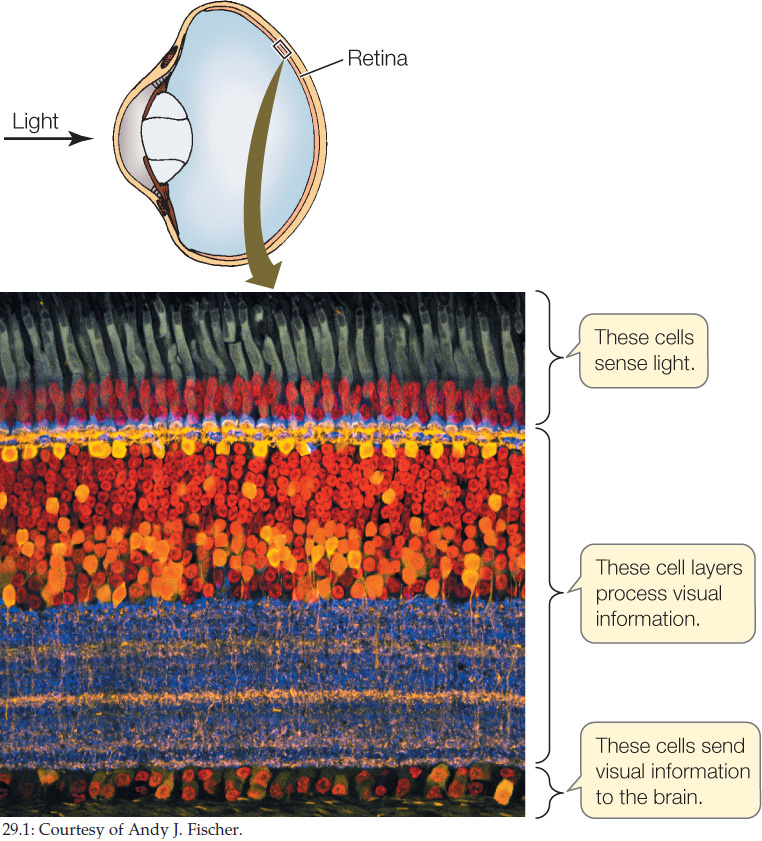
Organization is also evident in animals at the level of the organism, in the ways cells relate to each other in tissues, organs, and multi-organ systems. Consider the structure of an individual tissue such as the retina of the vertebrate eye. In each part of the retina, individual nerve cells are highly organized in relation to each other (FIGURE 29.1), and this structural organization among cells is essential for vision. Another type of organization at the organismal level is the interaction of tissues and organs to form systems of organs that work together. A large set of organs—including the stomach, intestines, pancreas, and brain—must interact in an organized, coordinated way to carry out digestion, for instance (we will return to these topics in greater detail in Concept 29.4).
Recall the second law of thermodynamics, which tells us that, left to itself, any organized system tends to lose organization and become more random as time passes. Animals must combat this tendency because any loss of organization in an animal’s body threatens the animal’s well-being or even life itself.
LINK
You can review the laws of thermodynamics in Concept 2.5
For biologists, the best definition of energy is that it is the capacity to create or maintain organization. In essence, the reason animals need to obtain energy is so they can do work (of several types) to maintain their organization by combating the effects of the second law of thermodynamics.
To fully understand the need for energy, we need to recognize that energy exists in several forms, and that only some forms can do work. In living organisms, heat is a form of energy that is unable to do work. To do work, animals need chemical-bond energy, also known as chemical energy. Moreover, when animals use chemical energy to do work, they convert that energy to heat, which they are unable to convert back to chemical energy. These principles explain why animals need to eat to obtain new chemical energy from their foods throughout their lives. As animals use chemical energy to do work, they convert it to heat. Thus they continually need more chemical energy, which they get by ingesting organic molecules that they can break down.
607
CHECKpoint CONCEPT 29.1
- From what sources do autotrophs obtain chemical energy and chemical building blocks?
- Why is it necessary for animals to continue gathering chemical building blocks even after they’ve reached their full adult sizes?
- In what form does energy enter a heterotroph’s body, and in what form does the majority of that energy inevitably leave?
Animals vary tremendously in how large their energy needs are. Among animals of any particular body size, some need 10 or even 50 times as much energy as others. Next we will discuss some of the reasons for this.