20.2 Natural Selection in Action: An Exemplary Case
For nearly a century after the publication of On the Origin of Species, there was not one example of natural selection that had been fully elucidated, that is, where the agent of natural selection was known, the effect on different genotypes could be measured, the genetic and molecular basis of variation was identified, and the physiological role of the gene or protein involved was well understood.
The first such “integrated” example of natural selection on a molecular variant was elucidated in the 1950s, before the genetic code was even deciphered. Remarkably, this trailblazing work revealed natural selection operating on humans. It still stands today as one of the most detailed and important examples of evolution by natural selection in any species.
The story began when Tony Allison, a Kenyan-
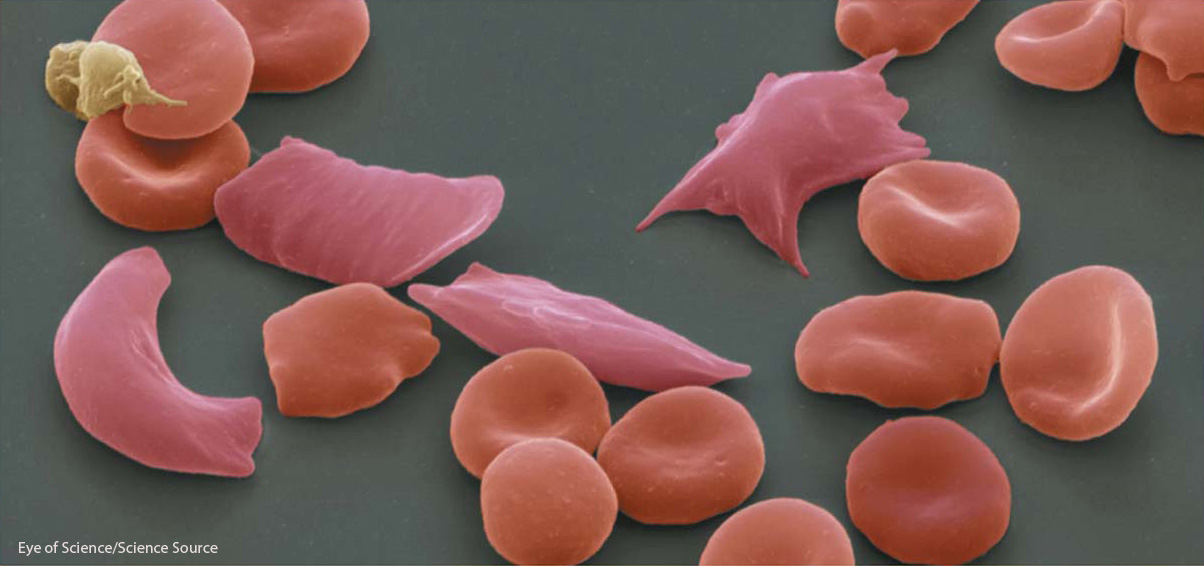
In 1949, the very year Allison went into the field, Linus Pauling’s research group demonstrated that patients with sickle-
Allison collected blood specimens from members of the Kikuyo, Masai, Luo, and other tribes across the very diverse geography of Kenya. While he did not see any particularly striking association between ABO or MN blood types among the tribes, he measured remarkably different frequencies of HS. In tribes living in arid central Kenya or in the highlands, the frequency of HbS was less than 1 percent; however, in tribes living on the coast or near Lake Victoria, the frequency of HbS often exceeded 10 percent and approached 40 percent in some locations (Table 20-1).
|
Tribe |
Ethnic affinity |
District/region |
%HbS |
|---|---|---|---|
|
Luo |
Nilotic |
Kisumu (Lake Victoria) |
25.7 |
|
Suba |
Bantu |
Rusingo Island |
27.7 |
|
Kikuy |
Bantu |
Nairobi |
0.4 |
The allele frequencies were surprising for two reasons. First, since sickle-
Allison’s familiarity with the terrain, tribes, and tropical diseases of Kenya led him to the crucial explanation. Allison realized that the HbS allele was at high frequency in low-
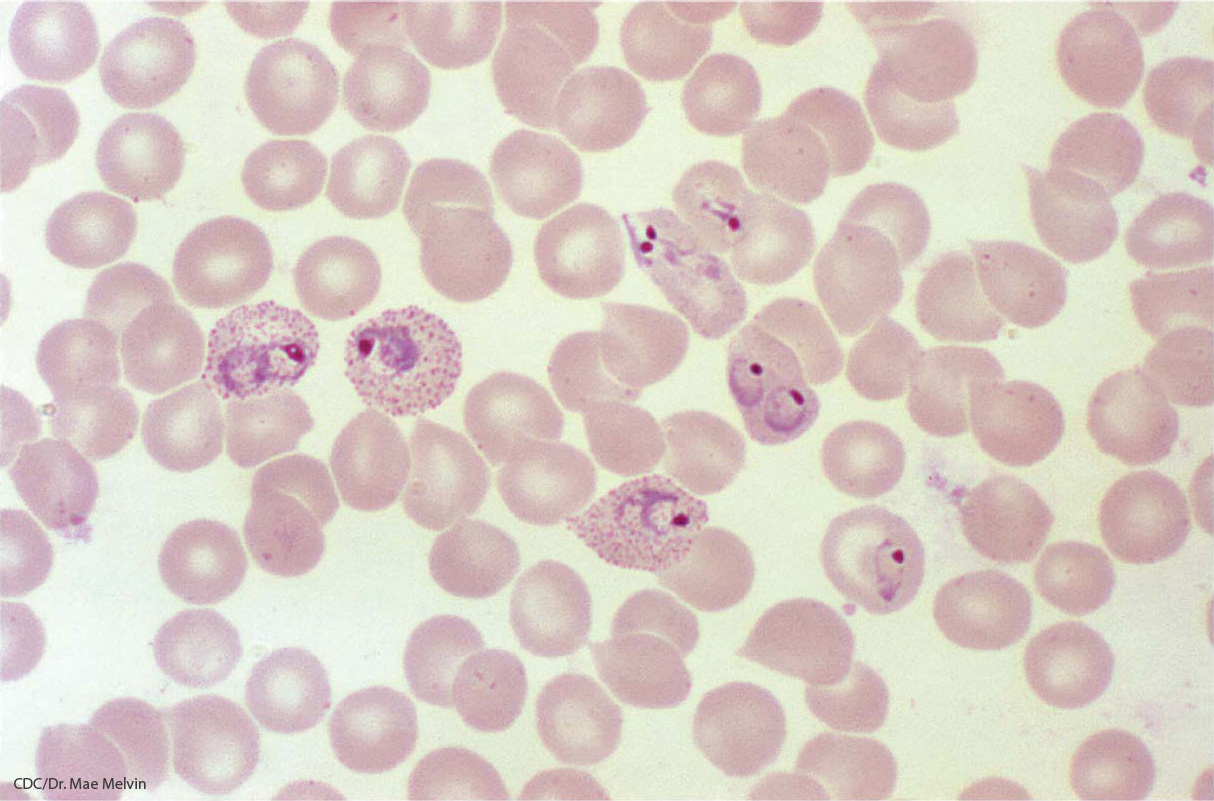
The selective advantage of HbS
In order to test this idea, Allison carried out a much larger survey of HbS frequencies across eastern Africa, including Uganda, Tanzania, and Kenya. He examined about 5000 individuals representing more than 30 different tribes. Again, he found HbS frequencies of up to 40 percent in malarial areas and frequencies as low as 0 percent where malaria was absent.
The link suggested that the HbS allele might affect parasite levels, so Allison also undertook a study of the level of parasites in the blood of heterozygous AS children versus wild-
The advantage to AS heterozygotes was especially striking in light of the disease suffered by SS homozygotes. Allison noted:
The proportion of individuals with sickle cells in any population, then, will be the result of a balance between two factors: the severity of malaria, which will tend to increase the frequency of the gene, and the rate of elimination of the sickle-
In other words, the sickle-
How much of an advantage do AS individuals experience? This can be calculated by measuring the frequency of the HbS allele in populations and examining how these frequencies differ from the frequencies expected under the assumptions of the Hardy–
|
Genotype |
Observed phenotype frequency |
Expected phenotype frequency |
Ratio of observed/expected |
W (relative fitness) |
Selective advantage |
|---|---|---|---|---|---|
|
SS |
29 |
187.4 |
0.155 |
0.155/1.12 = 0.14 |
|
|
AS |
2993 |
2672.4 |
1.12 |
1.12/1.12 = 1.00 |
1.0/0.88 = 1.136 |
|
AA |
9365 |
9527.2 |
0.983 |
0.983/1.12 = 0.88 |
|
|
Total |
12,387 |
12,387 |
This selective advantage has been well documented by long-
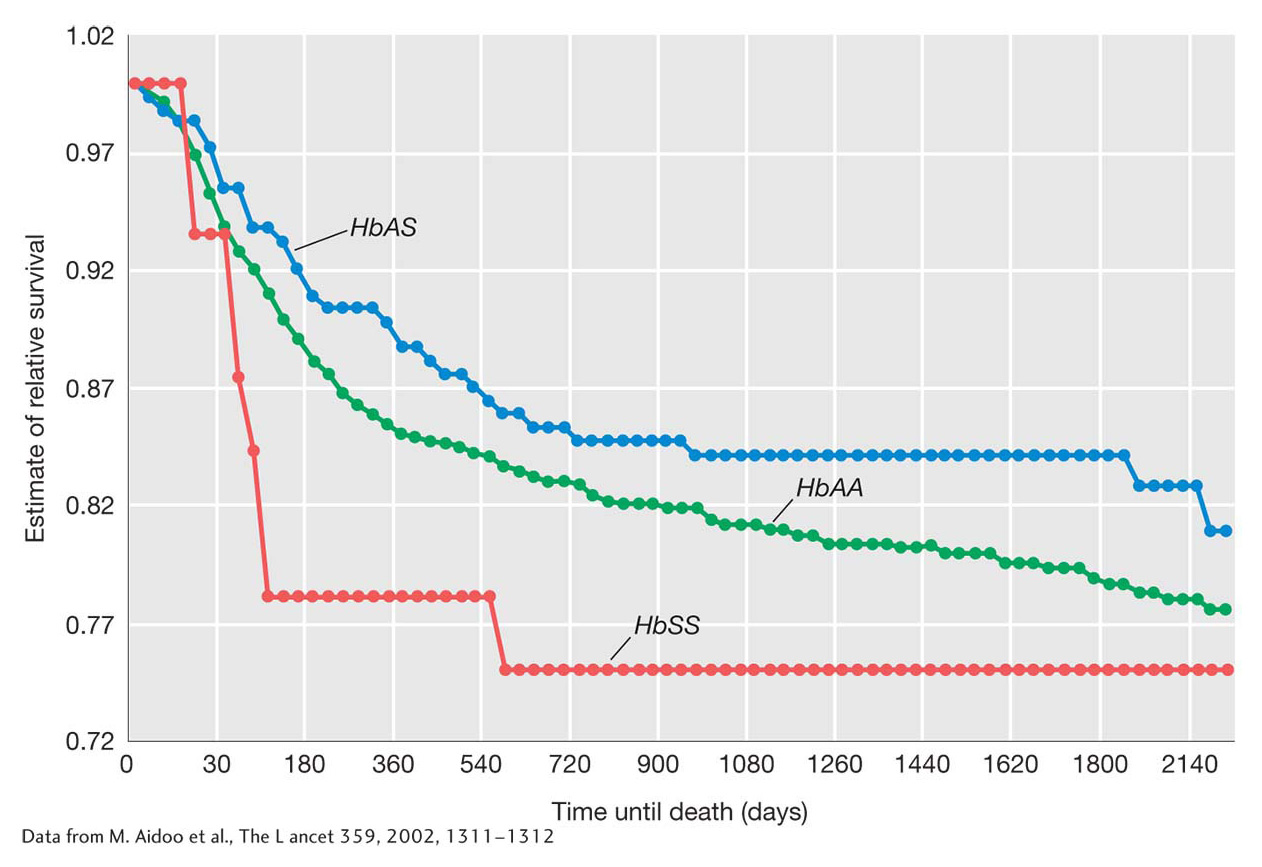
KEY CONCEPT
The sickle-The molecular origins of HbS
After Allison’s discovery, there was keen interest in determining the molecular basis of the difference(s) between HbS and HbA. Protein sequencing determined that HbS differs from HbA by just one amino acid, a valine in the place of a glutamic acid residue. This single amino acid change alters the charge of hemoglobins and causes it to aggregate into long rodlike structures within red blood cells. Once the genetic code was deciphered and methods for sequencing DNA were developed, HbS was determined to be caused by a single point mutation (CTC → CAC) in the glutamic acid codon encoding the sixth amino acid of the ß-globin subunit within the hemoglobin protein.
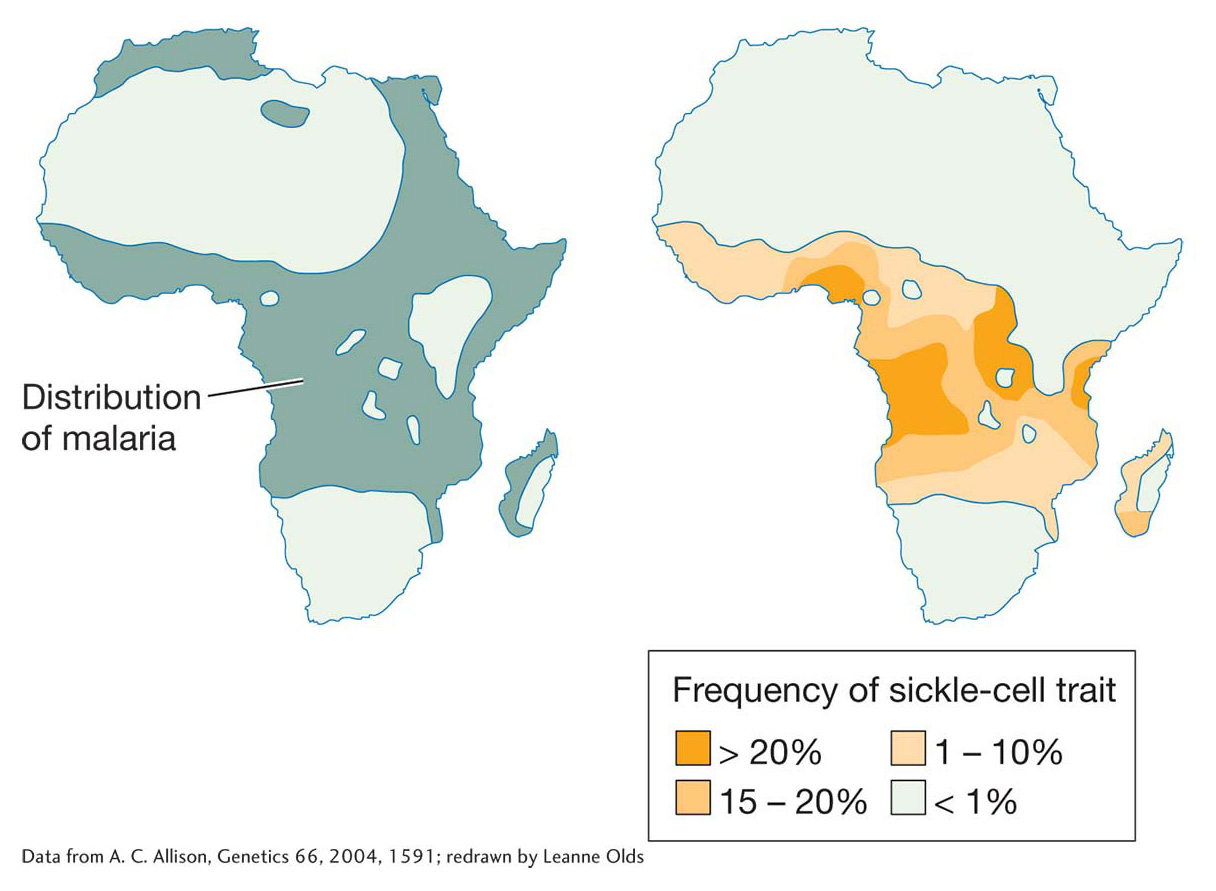
Interestingly, Allison also noted a high incidence of HbS outside of Africa, including in Italy, Greece, and India. Other blood-
KEY CONCEPT
The role of sickle-The role of HbS in conferring resistance to malaria illustrated three important facets of the evolutionary process:
Evolution can and does repeat itself. The multiple independent origins and expansions of the HbS mutation demonstrate that given sufficient population size and time, the same mutations can arise and spread repeatedly. Many other examples are now known of the precise, independent repetition of the evolution of adaptive mutations, and we will encounter several more in this chapter.
Fitness is a very relative, conditional status. Whether a mutation is advantageous or disadvantageous, or neither, depends very much on environmental conditions. In the absence of malaria, HbS is very rare and disfavored. Where malaria is present, it can reach high frequencies despite the disadvantages imparted to SS homozygotes. In African Americans, the frequency of HbS is on the decline because of selection against the allele in the absence of malaria in North America.
Page 771Natural selection acts on whatever variation is available, and not necessarily by the best means imaginable. The HbS mutation, while protective against malaria, also causes a life-
threatening condition. In areas where malaria is prevalent, where over 40 percent of the world’s population lives, the imperative of combating malaria counterbalances the deleterious effect of the sickle- cell mutation.