LESSON 20: Getting Connected: Ionic Compounds
THINK ABOUT IT
Sodium and chlorine atoms combine to form sodium chloride, NaCl, when sodium transfers a valence electron to chlorine. There are no other compounds that form between sodium and chlorine. For example, NaCl4 and Na3Cl are not possible compounds. Why is NaCl the compound that forms, rather than NaCl4 or Na3Cl?
How can valence electrons be used to predict chemical formulas?
To answer this question, you will explore
Ionic Compounds
The Rule of Zero Charge
More Complex Ionic Compounds
Ionic Compounds
EXPLORING THE TOPIC
Ionic Compounds
There are millions of different compounds on the planet. Each compound is a result of a combination of atoms and has a chemical formula. However, some combinations of atoms are simply not possible. Valence electrons are the key to figuring out chemical formulas and to determining which compounds are possible.
COMPOUNDS BETWEEN METALS AND NONMETALS
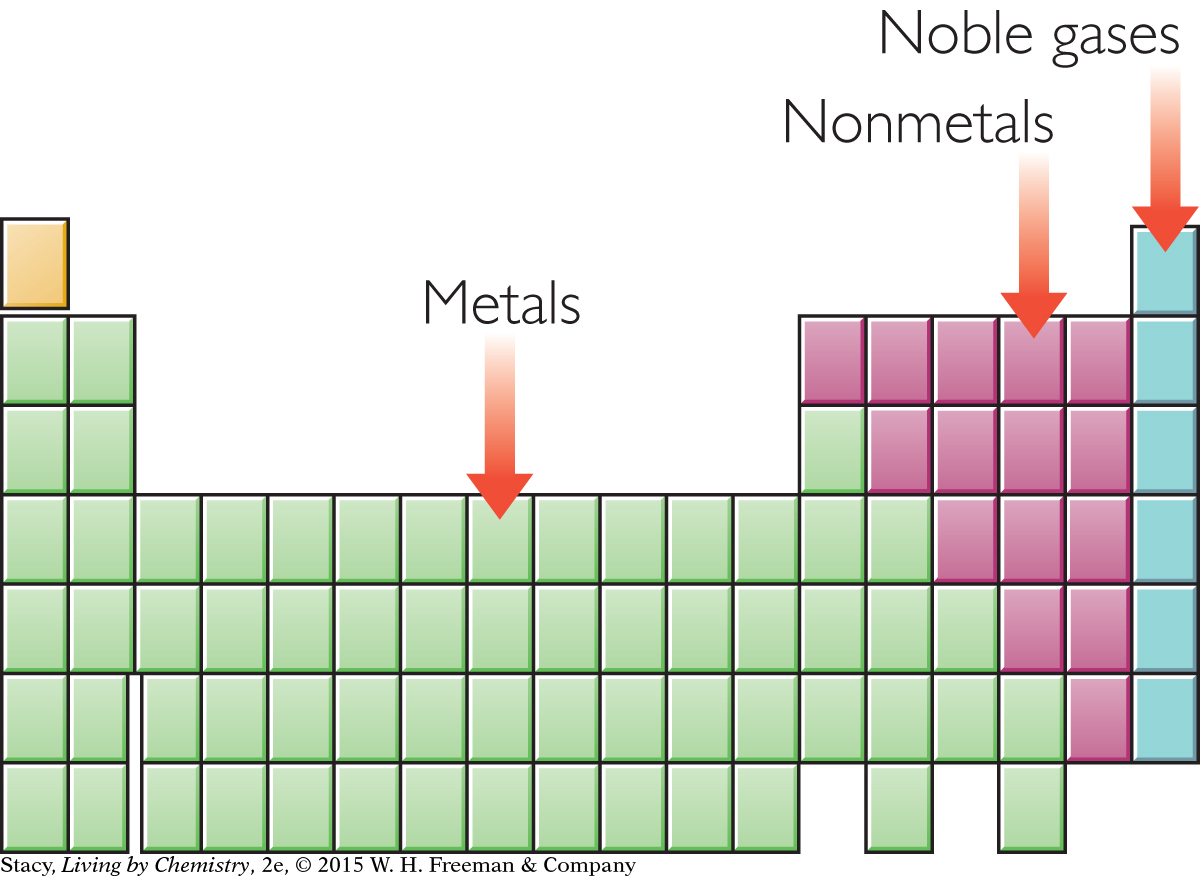
Atoms combine with other atoms to achieve the stability of the noble gases. One way that atoms accomplish this is to transfer valence electrons to other atoms. When atoms gain or lose electrons, the resulting atoms are no longer neutral. They become ions with charges. The compounds that form in this way are called ionic compounds. Ionic compounds, like the noble gases, are very stable.
Ionic compounds form between metal atoms and nonmetal atoms. The metal atoms, on the left side of the periodic table, tend to give up electrons and become cations, which have positive charges. The nonmetal atoms, on the right side of the periodic table, tend to accept electrons and become anions, which have negative charges. Because the cation and anion have opposite charges, they are strongly attracted to one another.
Big Idea
Big Idea
Ionic compounds form when valence electrons are transferred between atoms.
FORMULAS OF SIMPLE IONIC COMPOUNDS
The table below contains data on three simple ionic compounds. These compounds are all composed of a metal atom and a nonmetal atom in a 1:1 ratio. Pay particular attention to the total charge and the number of valence electrons on each of the atoms that combine.

The Rule of Zero Charge
The Rule of Zero Charge
You can see a distinct pattern to the ions that form in ionic compounds. Every time a metal atom and a nonmetal atom bond, they form a compound with an overall zero charge. This is known as the rule of zero charge.
CONSUMER CONNECTION
CONSUMER
CONNECTION
All ionic compounds form crystals. If you look at grains of table salt with a magnifying glass, you will see that the pieces are shaped like tiny boxes.

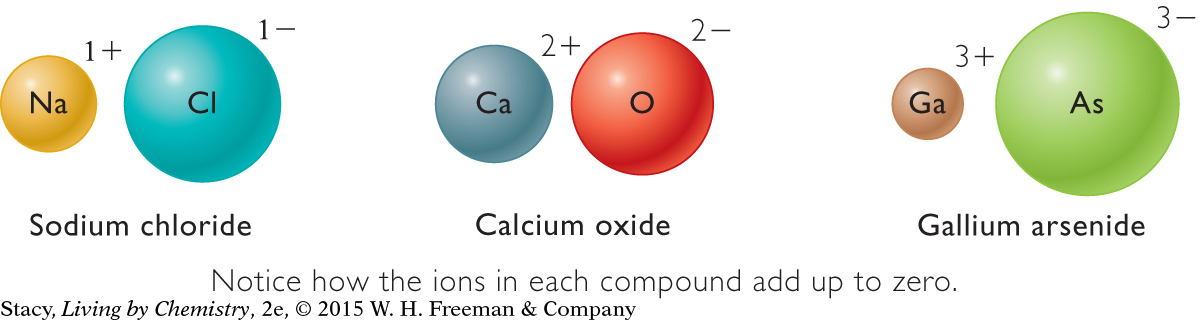
The charges on the metal cations and nonmetal anions add up to zero. The rule of zero charge is useful in predicting the chemical formulas for ionic compounds, which form between a metal and a nonmetal.
PREDICTING IONIC BONDING USING THE PERIODIC TABLE
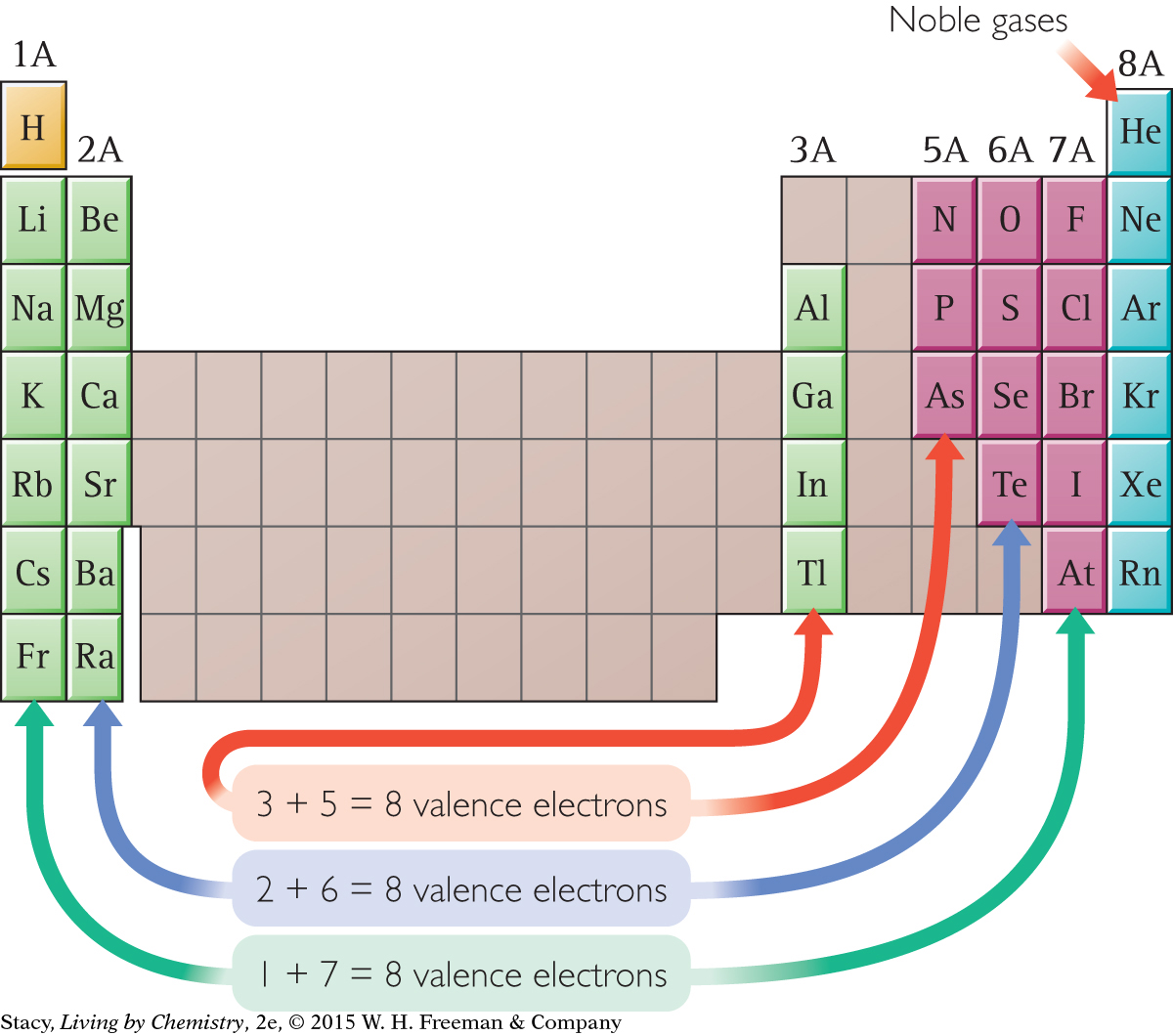
You may have noticed that the atoms that combine to form ionic compounds come from very predictable places on the periodic table. Sodium, with one valence electron, can combine with chlorine, fluorine, or bromine, each with seven valence electrons. In fact, any Group 1A element can combine with any Group 7A element to form an ionic compound.
Important to Know
The atoms in Group 4A can either transfer or accept electrons.
The drawing illustrates how you can use the periodic table to decide which atoms will combine in a 1:1 ratio to form an ionic compound. There are dozens of possible combinations. For example, an atom in Group 3A can form a compound with an atom in Group 5A.
More Complex Ionic Compounds
More Complex Ionic Compounds
CONSUMER CONNECTION
CONSUMER
CONNECTION
Energy drinks help replace necessary ions, known as electrolytes, in your body.

All of the compounds discussed in this lesson so far have a metal-to-nonmetal ratio of 1:1. However, it is also possible to have compounds with different ratios. For example, one magnesium atom can combine with two chlorine atoms to form magnesium chloride, MgCl2. Before bonding, each magnesium atom has two valence electrons. These two electrons are transferred to two different chlorine atoms, each of which needs one more electron to have the same number of electrons as the noble gas argon, Ar.
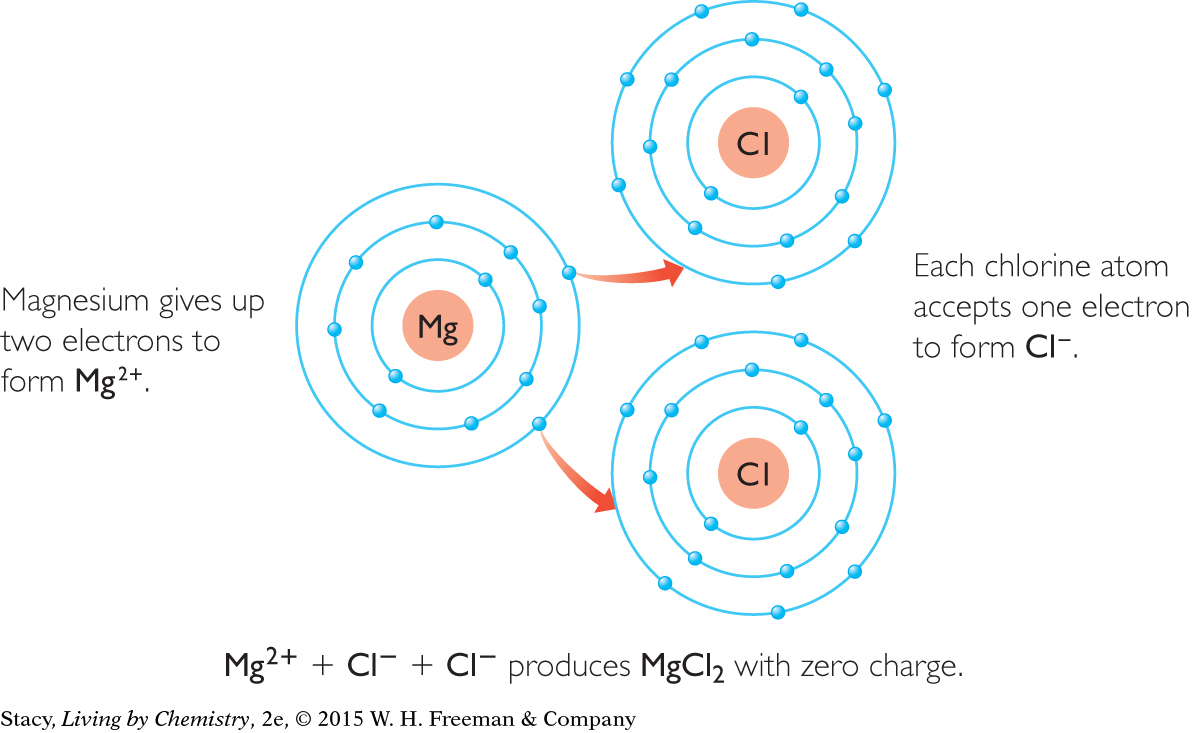
Notice in this table that the atoms combine in such a way that the resulting compound has a zero charge.
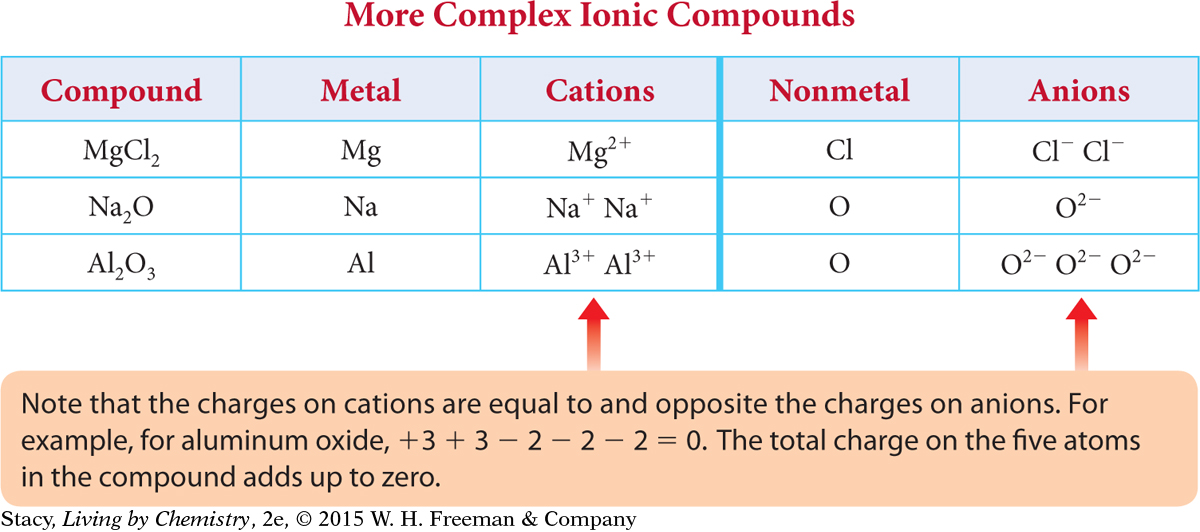
You can see that sometimes more than one atom of a particular element must combine to result in a compound with a zero charge. So, ionic compounds in ratios of 1:2, 2:1, or even 2:3 may result.
Example
Sodium Sulfide
The chemical formula for sodium sulfide is Na2S.
What is the charge on each sodium ion, Na? On each sulfur ion, S?
Show that the charges on the ions add up to zero.
Is Na2Cl a possible ionic compound? Why or why not?
Solution
Sodium, Na, is in Group 1A and has one valence electron. Sodium atoms give up one electron to have a charge of +1. Sulfur, S, is in Group 6A and has six valence electrons. Sulfur atoms gain two electrons to have a charge of –2.
Check the charge on the compound:
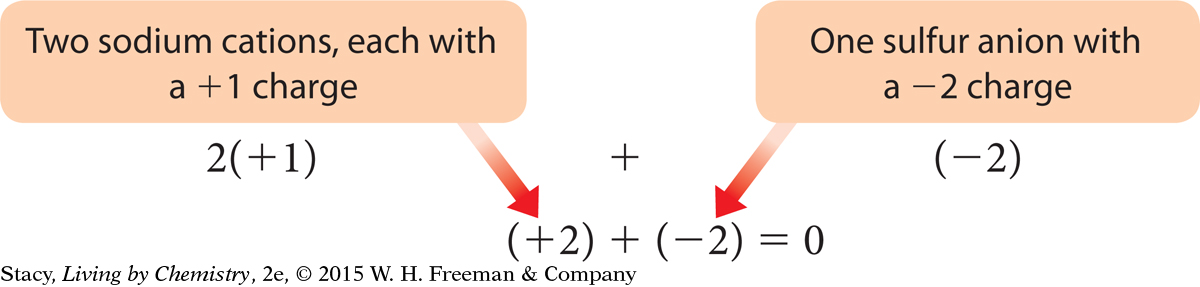
The charges on the ions add up to zero, as they should.
Each sodium atom has one valence electron and each chlorine atom has seven valence electrons. If two sodium atoms gave up electrons to one chlorine atom, the chlorine atom would have nine valence electrons, which is not a stable number resembling a noble gas. So Na2Cl is not a compound that is likely to form. Also, such a compound would not have a neutral charge.
LESSON SUMMARY
LESSON SUMMARY
How can valence electrons be used to predict chemical formulas?
KEY TERMS
ionic compound
rule of zero charge
Metal atoms and nonmetal atoms combine to form ionic compounds through electron transfer. The metal atoms give up electrons. They form ions with a positive charge, called cations. The nonmetal atoms accept electrons and form ions with a negative charge, called anions. You can use the positions of the elements on the periodic table to predict the ionic compounds they can form. When an ionic compound forms, the total charge on the atoms adds up to zero. This is known as the rule of zero charge.
Exercises
Reading Questions
How can you use valence electrons to predict which ionic compounds will form?
How does the rule of zero charge help you predict the formula of an ionic compound?
Reason and Apply
Lithium nitride has the formula Li3N.
What is the charge on the lithium ion?
What is the charge on the nitrogen ion?
Show that the charges on the ions add up to zero.
What is the total number of valence electrons in all the atoms in Li3N?
Aluminum arsenide has the formula AlAs.
What is the charge on the aluminum ion?
What is the charge on the arsenic ion?
Show that the charges on the ions add up to zero.
What is the total number of valence electrons in all the atoms in AlAs?
For each of these compounds, show that the charges on the ions add up to zero.
KBr
CaO
Li2O
CaCl2
AlCl3
What are the total numbers of valence electrons in all the atoms in each of these compounds?
KBr
CaO
Li2O
CaCl2
AlCl3
Explain why the following compounds do not form.
NaCl2
CaCl
AlO