LESSON 26: Electron Glue: Bonding
THINK ABOUT IT
HISTORY CONNECTION
HISTORY
CONNECTION
Stephanie Kwolek started working for DuPont in 1946. In 1965, she succeeded in creating synthetic fibers of exceptional strength due to their extremely strong bonds. This led to Kevlar, the material used in bulletproof vests.

All of the objects in our everyday world, including ourselves, are made up of individual atoms. But what holds those atoms together? Why don’t objects just crumble into piles of individual atoms? Something must be holding the atoms together. And why is the desk in your classroom solid, while water simply runs through your fingers? Something about the way atoms are connected must give substances the properties we observe.
How are atoms connected to one another?
To answer this question, you will explore
Bonds: The “Glue” between Atoms
Models of Bonding
Relating Bonds and Properties
Bonds: The “Glue” between Atoms
EXPLORING THE TOPIC
Bonds: The “Glue” between Atoms
Chemists call the attraction that holds atoms together a chemical bond. As you will discover, several different types of chemical bonds exist. All bonds involve the electrons in some way. A chemical bond is an attraction between the positive charges on the nucleus of one atom and the negative charges on the electrons of another atom. This attraction is so great that it keeps the atoms connected to one another.
Types of Bonding
Types of Bonding
Recall from Lesson 25 that most substances can be divided into four categories, based on their physical properties. These four categories can be explained by different models of bonding.
A bond is a force of attraction, so it is not possible to see the actual bonds between atoms. However, a model can help to explain how atoms are bonded in substances. Models can also help us to understand how bonding accounts for certain properties of substances that we observe.
Big Idea
Big Idea
There are four main models of chemical bonds between atoms.
The four models of bonding are called ionic, molecular covalent, metallic, and network covalent. Take a moment to locate the valence electrons in each model on the next page. The red spheres represent the nuclei of atoms and the core electrons, while the blue areas suggest where the valence electrons are located.
Four Models of Bonding
|
MODEL 1: IONIC Properties of ionic substances: Dissolve in water Conduct electricity when dissolved Tend to be brittle solids Made of metal and nonmetal atoms combined 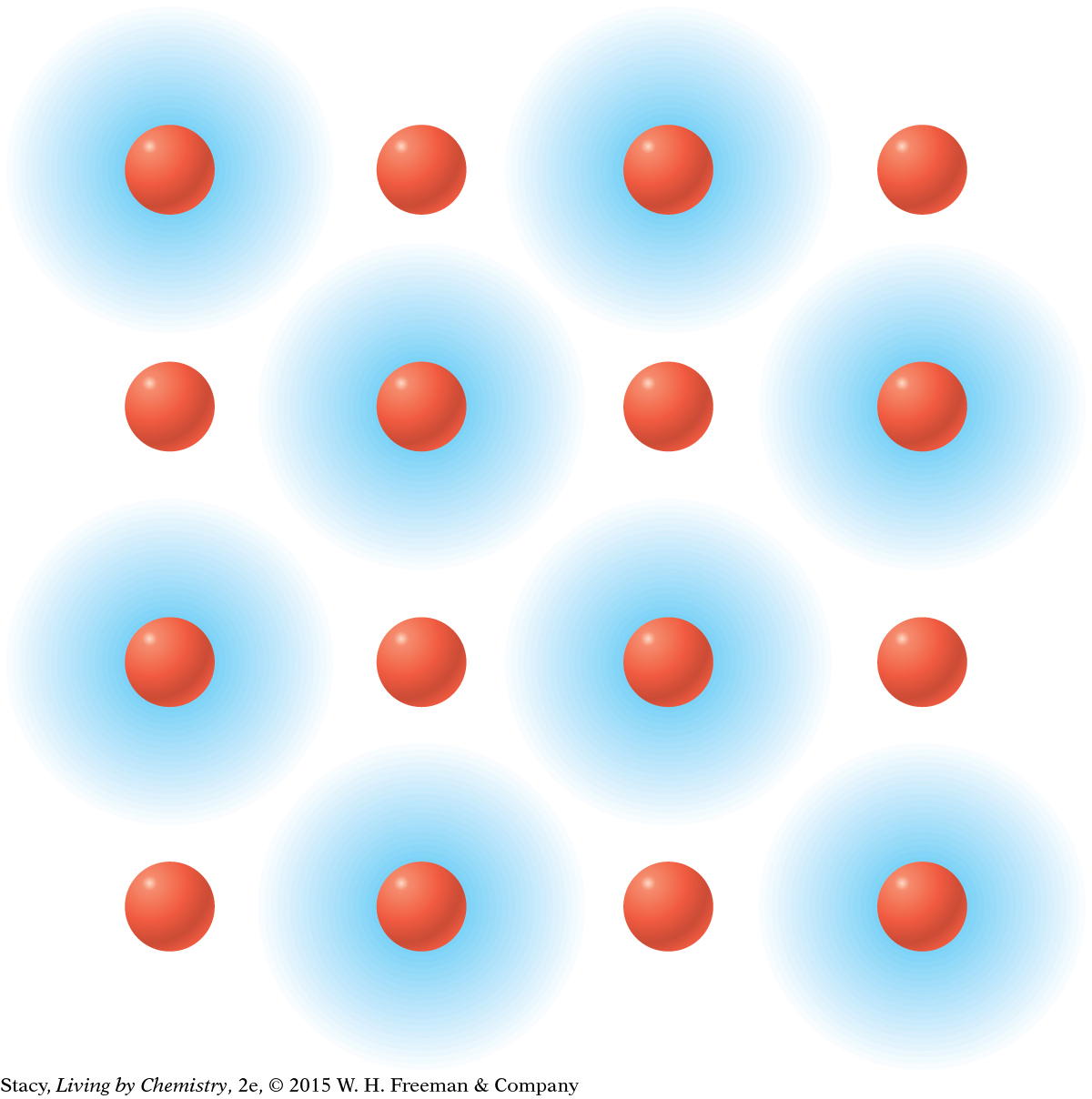
In ionic bonding, the valence electrons are transferred from one atom to another. Metal atoms transfer their valence electrons to nonmetal atoms. |
MODEL 2: MOLECULAR COVALENT Properties of molecular covalent substances: Some dissolve in water, some do not Do not conduct electricity Some are liquids or gases Made entirely of nonmetal atoms 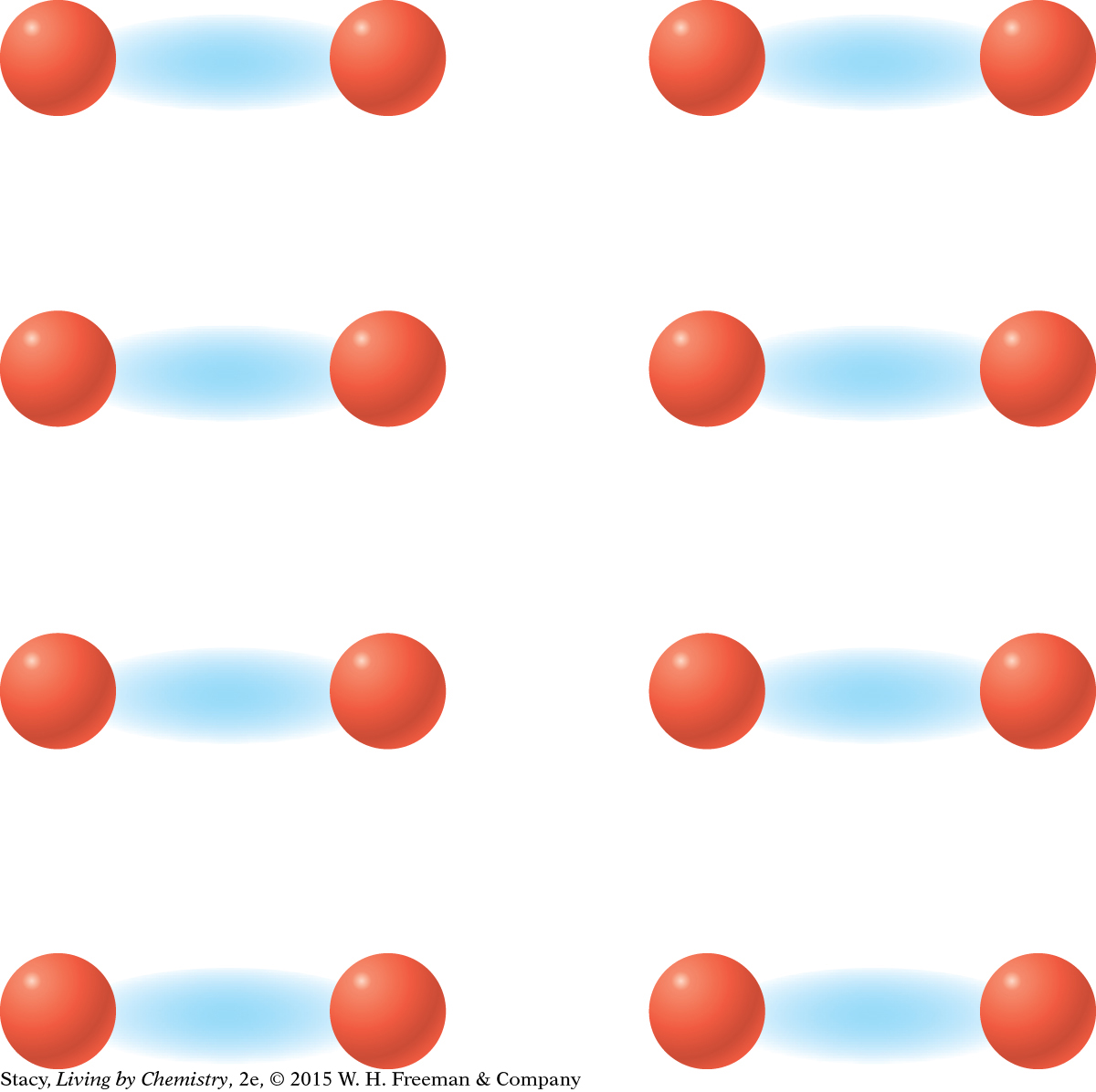
In molecular covalent bonding, the valence electrons are shared between pairs or groups of atoms. This creates small stable units, called molecules, within the substance. |
|
MODEL 3: METALLIC Properties of metallic substances: Do not dissolve in water Conduct electricity Bendable, malleable solids Made entirely of metal atoms 
In metallic bonding, the valence electrons are free to move about the substance. |
MODEL 4: NETWORK COVALENT Properties of network covalent substances: Do not dissolve in water Do not conduct electricity Extremely hard solids Made entirely of nonmetal atoms 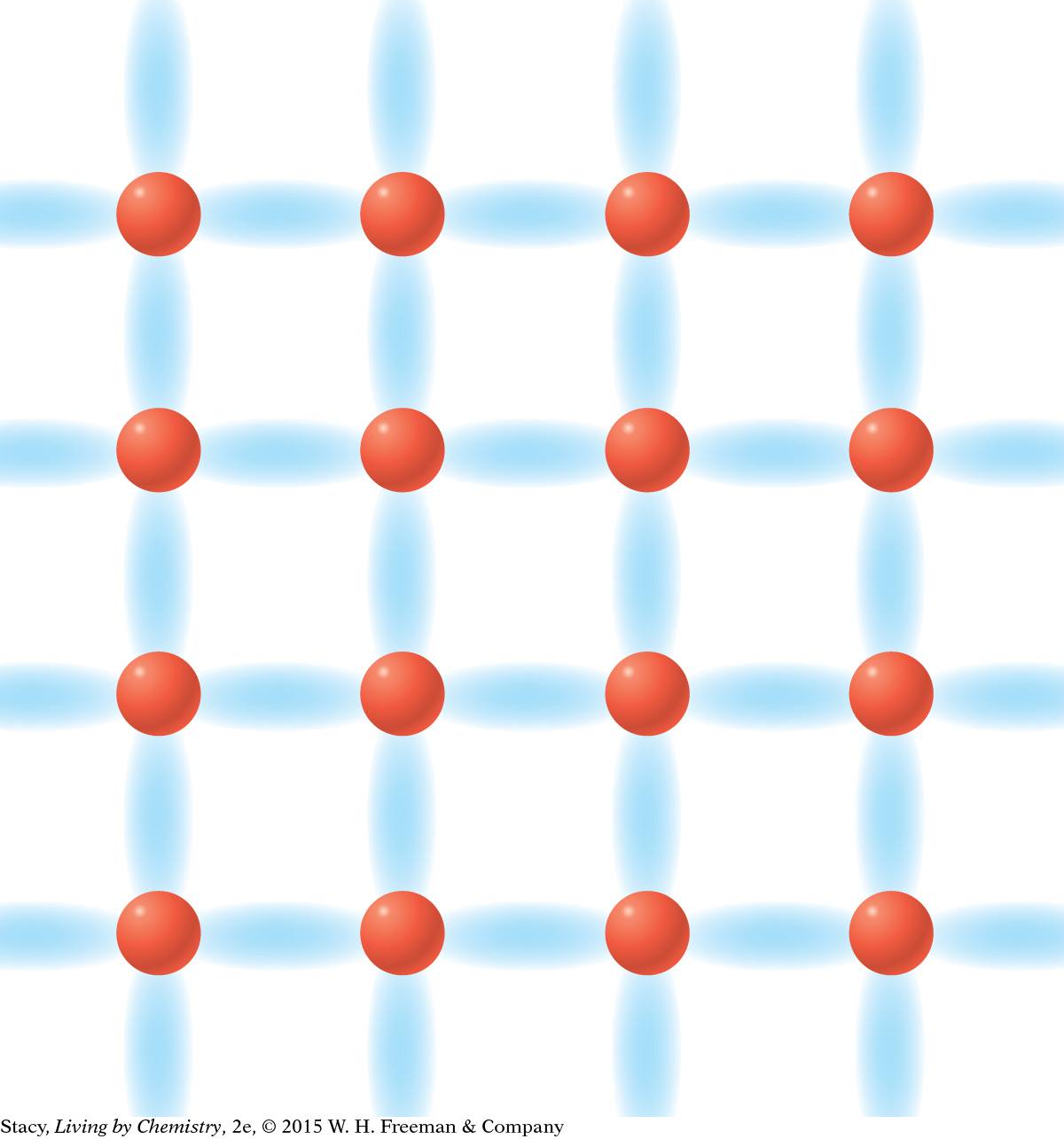
Network covalent bonding is similar to molecular covalent bonding, but the valence electrons are shared throughout the entire substance. |
You have already been introduced to ionic compounds. These all have ionic bonding in which metal atoms transfer valence electrons to nonmetal atoms. The resulting oppositely charged ions are strongly attracted to each other. This attraction is what holds the ions together.
In covalent bonding, the nucleus of one atom is attracted to the valence electrons of another atom. Unlike in ionic bonding, one atom does not transfer an electron to the other. Instead, both atoms share the valence electrons between them.
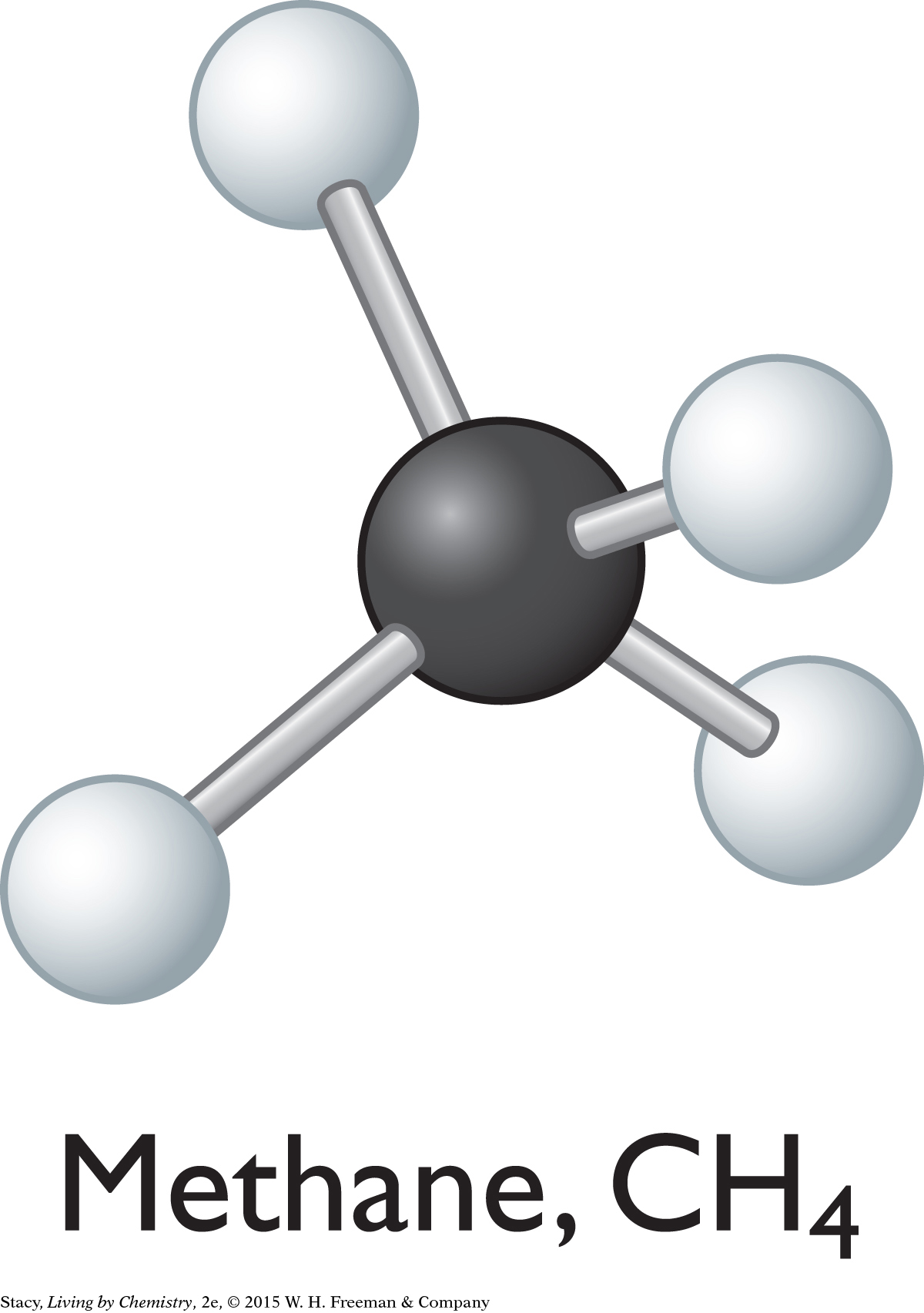
Covalent bonding can happen in two different ways. In molecular covalent bonding, the atoms bond to form individual clusters called molecules, such as the methane molecule shown here.
In network covalent bonding, the valence electrons are shared between atoms but form a highly regular extended network, creating a very durable structure. Diamond consists of carbon atoms that are covalently bonded in a network.
CONSUMER CONNECTION
CONSUMER
CONNECTION
Chalk, which is brittle, is an example of an ionic substance. The chemical name and formula for chalk are calcium carbonate, CaCO3.

In a metal, the valence electrons are distributed throughout the substance in what is sometimes called a “sea” of electrons. The valence electrons are free to move throughout the substance. The atoms are bonded by the attraction between the positively charged atoms and the negatively charged “sea” of electrons.
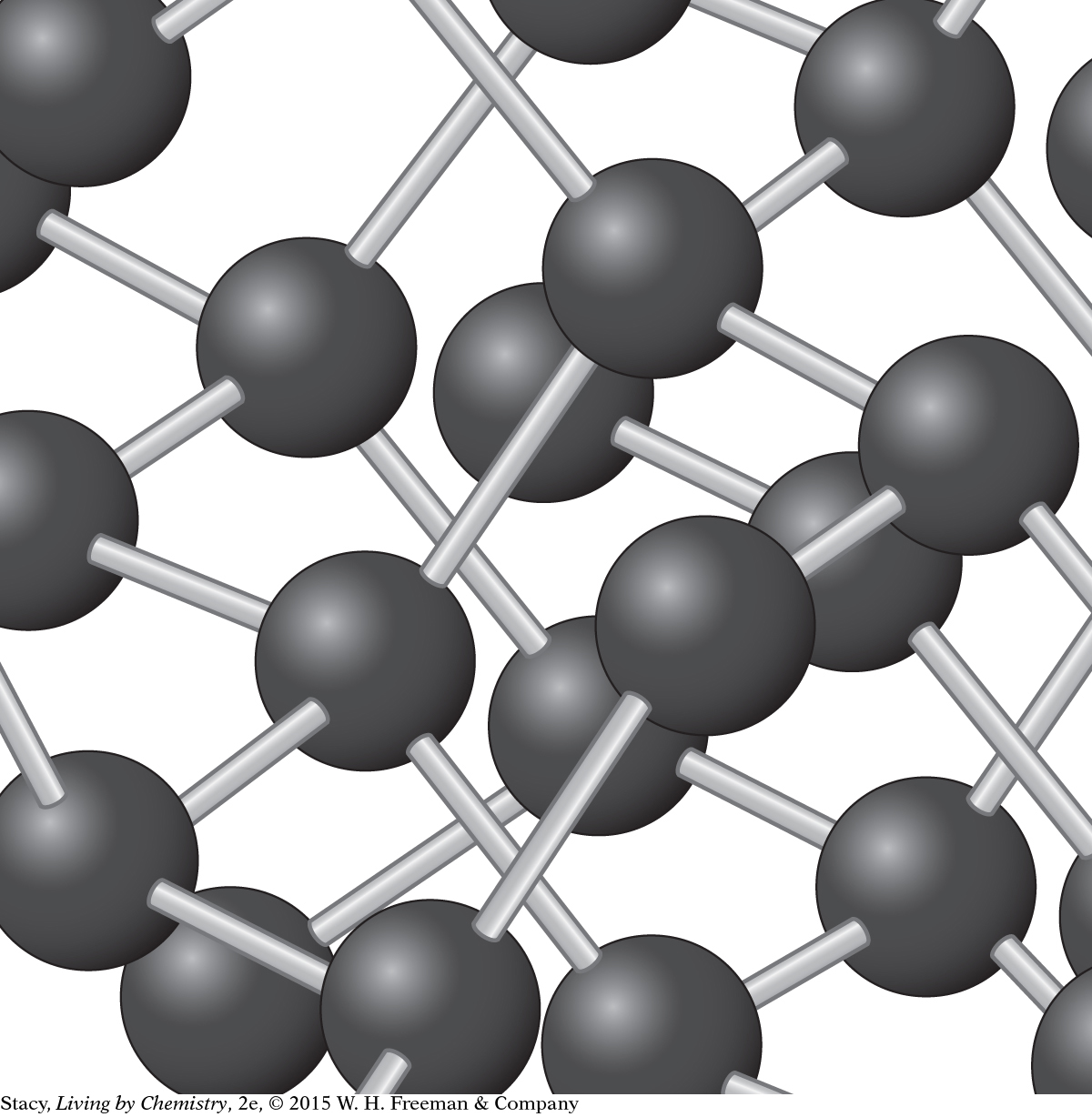
Diamond consists of carbon atoms that are covalently bonded in a network.
|
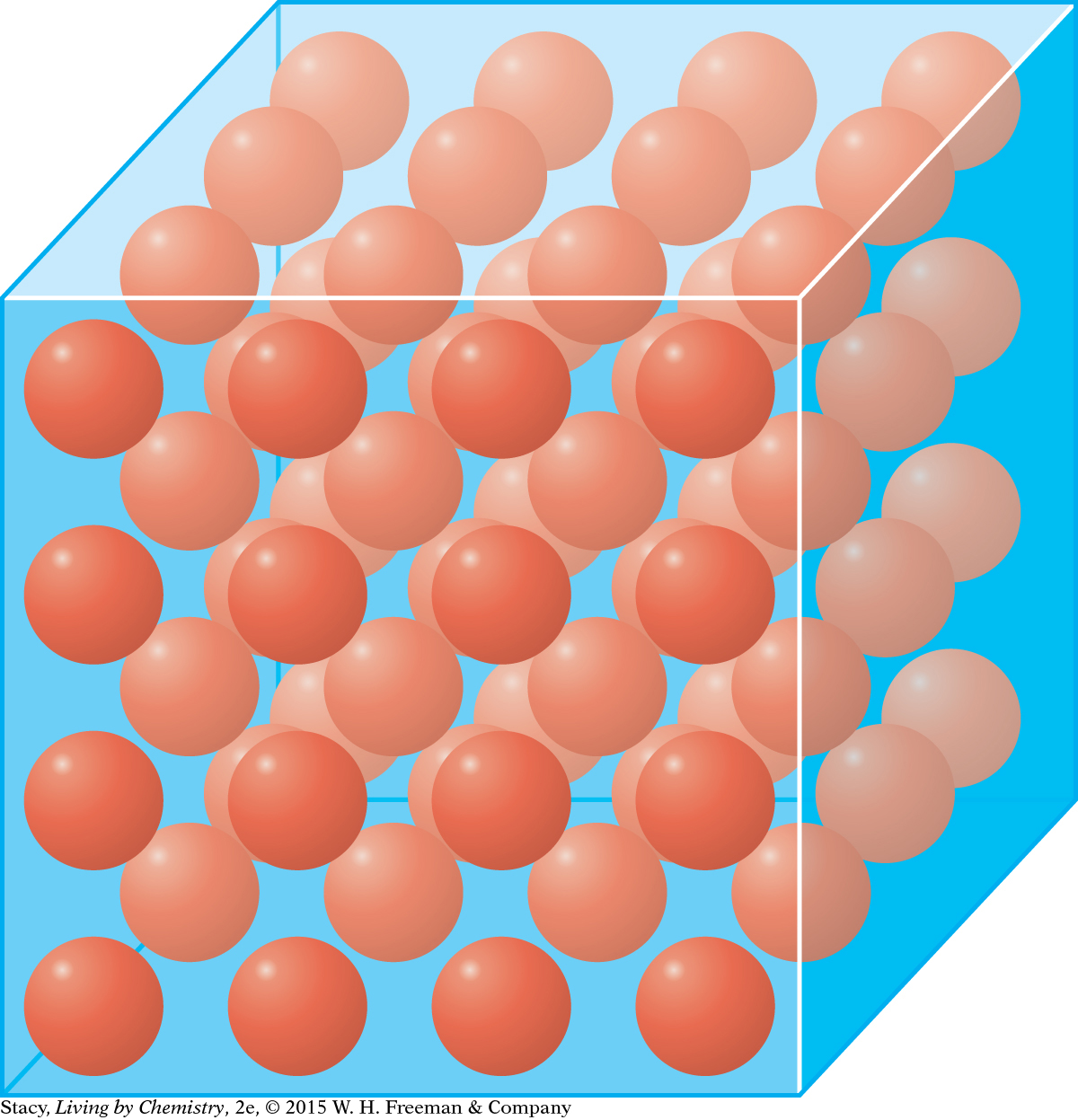
In a metal, positively charged atoms and a negatively charged “sea” of atoms create bonds.
|
Relating Bonds and Properties
Relating Bonds and Properties
Some properties of substances, such as solubility and conductivity, are directly related to the type of bonds the atoms in the substances have. Therefore, it is possible to match the bond type to the physical properties observed in different types of substances. Examine what happens to each type of substance when it is struck by a hypothetical hammer.
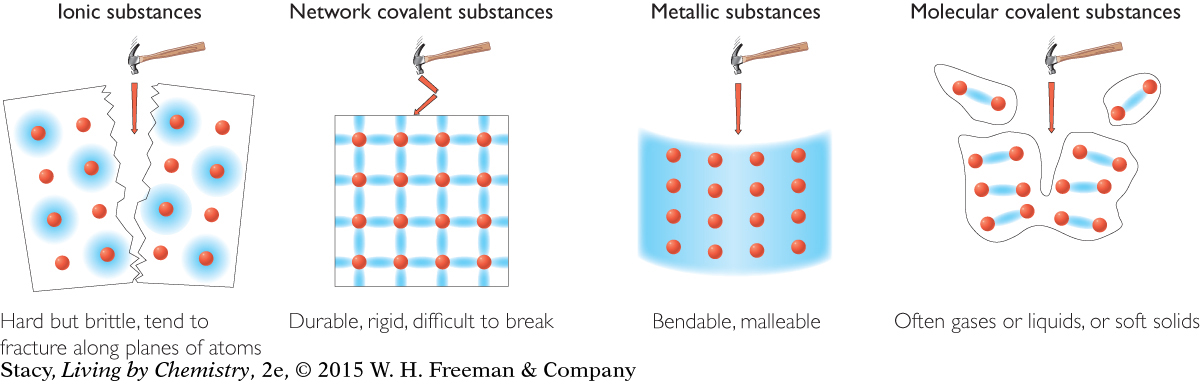
Notice that the hardest substance is a solid with network covalent bonding. This is because bonding in these substances is in an organized network.
INDUSTRY CONNECTION
INDUSTRY
CONNECTION
Metals can be combined into alloys. For example, CuZn is brass, a copper-zinc alloy. Chrome-moly is an alloy of steel, chrome, and molybdenum, used to make bicycle frames that are stronger and lighter than those made of steel. Steel itself is an alloy of iron and carbon.

Bonding can also help to explain the properties of dissolving and conductivity. Examine the illustration below representing dissolving. Water is represented by the lighter blue areas. Ionic solids and molecular covalent substances dissolve in water. Metallic solids and network covalent solids do not.
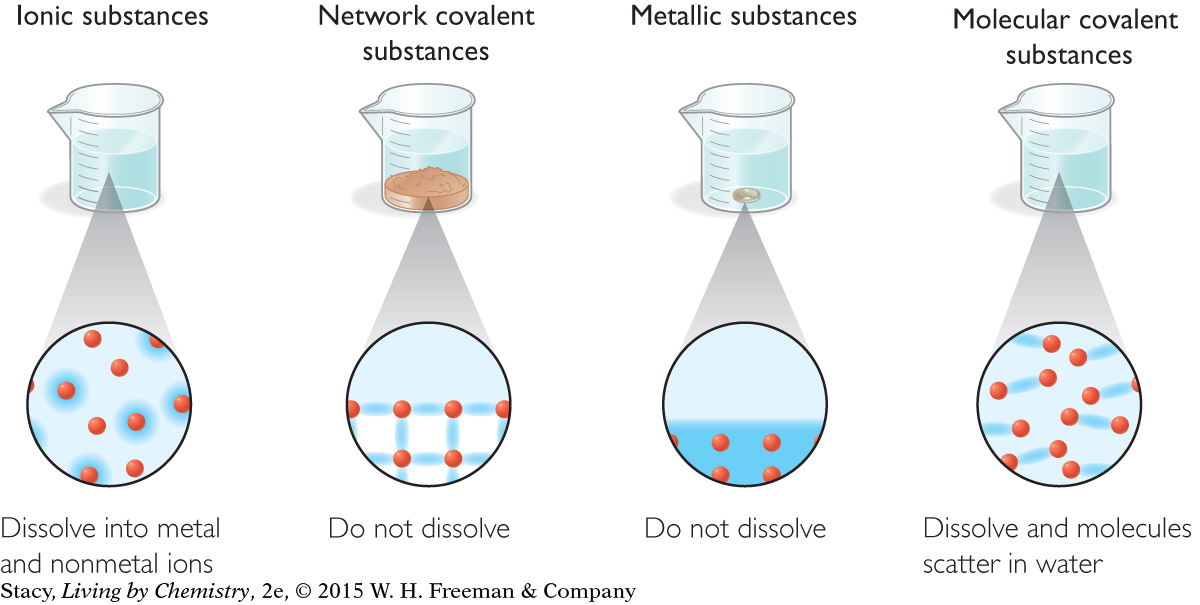
Conduction requires the movement of a charged particle, either an ion or an electron. Metals conduct electricity because the valence electrons are free to move throughout the solid. Ionic compounds that have been dissolved in water conduct electricity because the cations and anions are free to move in the solution. Network covalent solids and molecular covalent substances do not conduct electricity. The charge cannot move in these substances because the electrons are “stuck” between the atoms and are not available to move.
The periodic table is a valuable tool in figuring out bonding. You can use the table to determine if the elements in a compound are metals, nonmetals, or both.
Ionic compounds, such as salts, are made from metal and nonmetal elements.
Metallic compounds, such as brass, are made only of metal atoms.
Network covalent compounds, such as diamonds, and molecular covalent compounds, such as methane, are made from nonmetals.
Example
Identifying the Type of Bonding
Determine the bonding in each of the following substances. What general physical properties can you expect of each substance?
magnesium chloride, MgCl2
rubbing alcohol, C3H8O
Solution
Magnesium chloride, MgCl2, is an ionic compound with ionic bonding because it is made of a metal and a nonmetal element. It is probably brittle, dissolves in water, and conducts electricity when dissolved.
Rubbing alcohol, C3H8O, is a molecular covalent substance with molecular covalent bonding because it is made entirely of nonmetal atoms and is a liquid.
LESSON SUMMARY
LESSON SUMMARY
How are atoms connected to one another?
KEY TERMS
chemical bond
ionic bonding
molecular covalent bonding
metallic bonding
network covalent bonding
covalent bonding
molecule
Atoms in substances are held together by chemical bonds. Chemists have identified four main models of bonding within substances: ionic, network covalent, molecular covalent, and metallic. Many properties of a substance correspond to the type of bonding between the atoms of that substance.
Exercises
Reading Questions
Explain why substances do not simply crumble into piles of atoms.
Name the four models of bonding and explain them in your own words. Be specific about the location of the valence electrons.
Reason and Apply
Determine the type of bonding in each substance.
zinc, Zn(s)
propane, C3H8(l)
calcium carbonate, CaCO3(s)
Based on physical properties, which of these substances is an ionic compound? Explain your reasoning.
hair gel
silver bracelet
motor oil
baking soda
In the copper cycle from Lesson 7, you saw that nitrogen dioxide, NO2(g), formed when copper, Cu(s), was dissolved in nitric acid. How would you classify the bonding in NO2(g)? Explain.
Which statement is true?
Aqueous solutions of calcium chloride, CaCl2, conduct electricity.
Glass, made of silicon dioxide, SiO2, does not dissolve in water.
Ethanol, C2H6O, dissolves in water but does not conduct electricity.
Brass, also called copper zinc, CuZn, conducts electricity.
All of the above are true.
Suppose you have a mixture of sodium chloride, NaCl, and carbon, C. Explain how you can use water to separate the two substances.
Explain why copper is used as wire, but copper chloride is not.
Explain why carbon is a solid and not a gas.
Will each of these substances dissolve in water? Explain your thinking.
Ca
NaNO3
Si
CH4
CuSO4