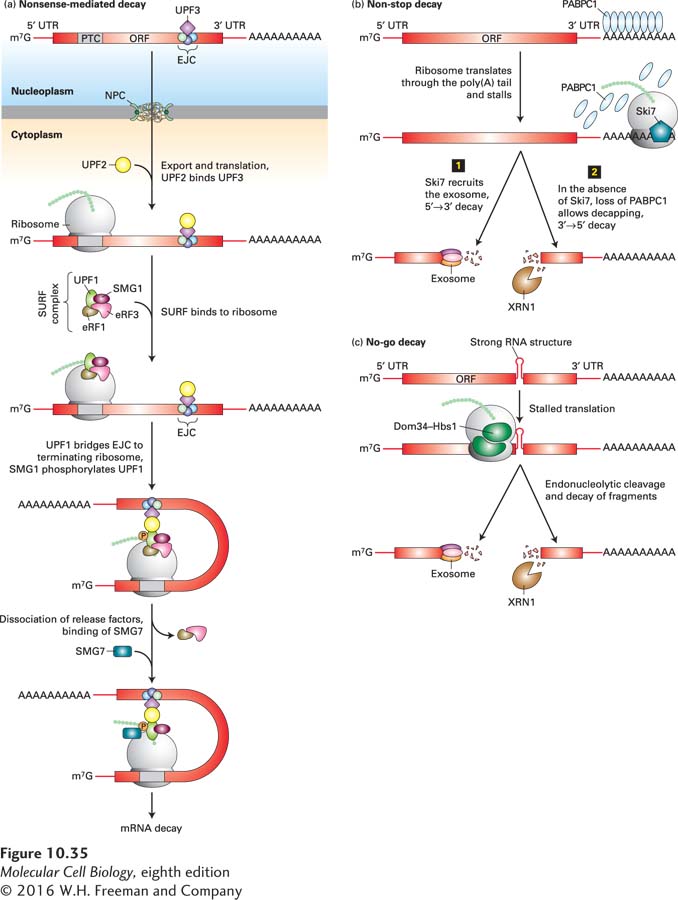
FIGURE 10- e- 2- n- C- n- 5- o- m- 4-