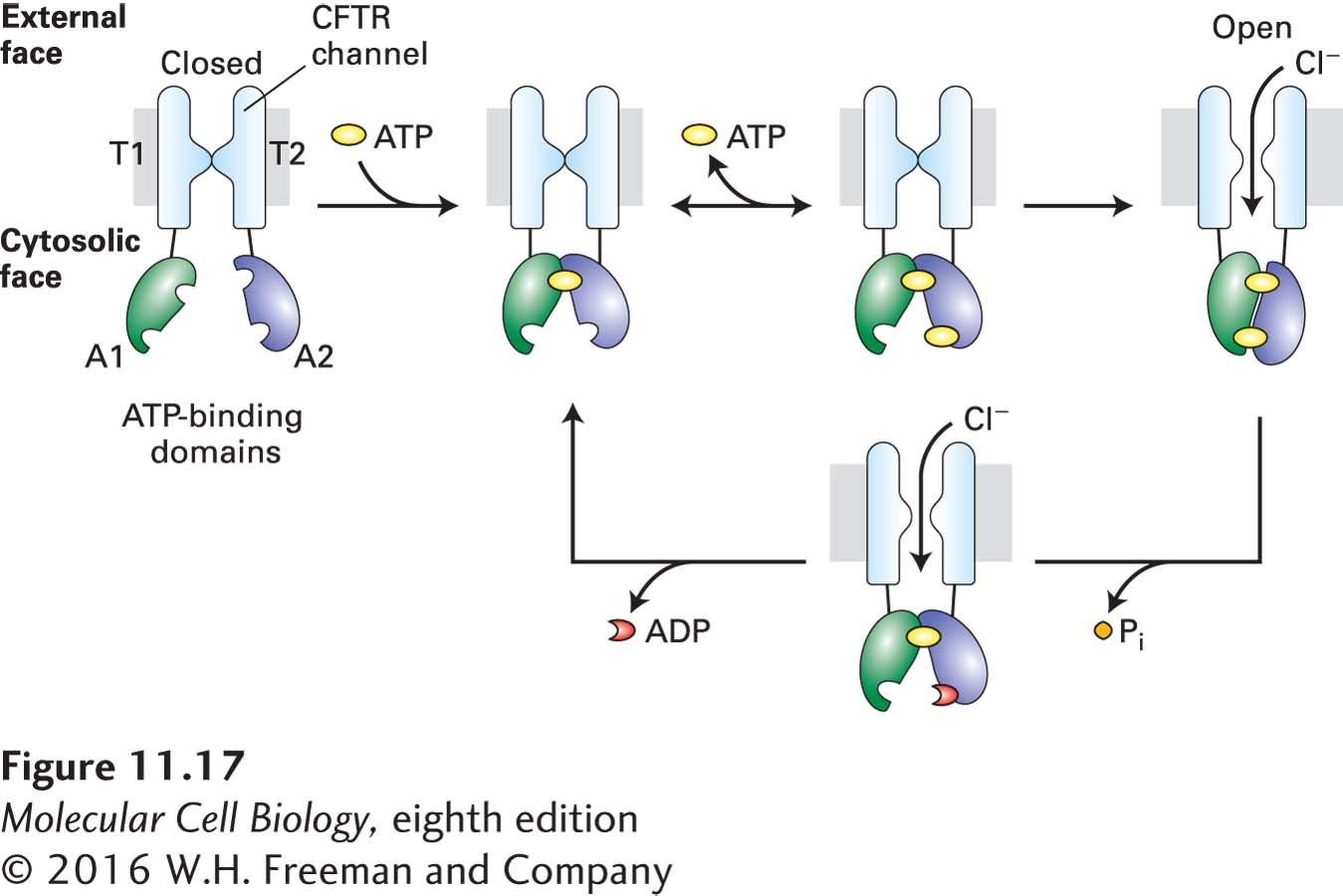
FIGURE 11- 1– 1– P-