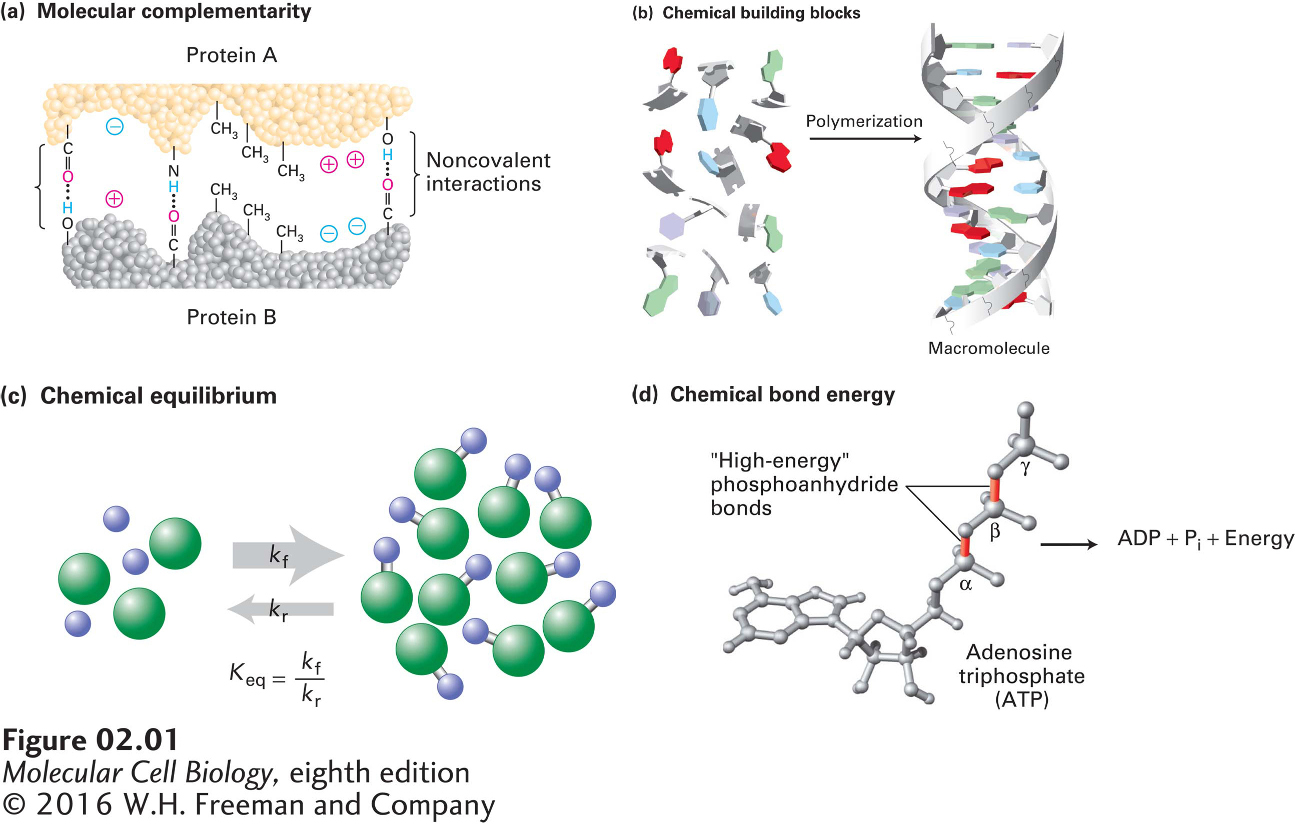
FIGURE 2- 1 Chemistry of life: four key concepts. (a) Molecular complementarity lies at the heart of all biomolecular interactions (see Section 2.1), as when two proteins with complementary shapes and chemical properties come together to form a tightly bound complex. (b) Small molecules serve as building blocks for larger structures (see Section 2.2). For example, to generate the information- carrying macromolecule DNA, four small nucleotide building blocks are covalently linked into long strings (polymers), which then wrap around each other to form the double helix. (c) Chemical reactions are reversible, and the distribution of the chemicals between starting reactants (left) and the products of the reactions (right) depends on the rate constants of the forward (kf, upper arrow) and reverse (kr, lower arrow) reactions. The ratio of these, Keq, provides an informative measure of the relative amounts of products and reactants that will be present at equilibrium (see Section 2.3). (d) In many cases, the source of energy for chemical reactions in cells is the hydrolysis of the molecule ATP (see Section 2.4). This energy is released when a high- energy phosphoanhydride bond linking the b and g phosphates in the ATP molecule (red) is broken by the addition of a water molecule, forming ADP and Pi.
[Leave] [Close]