Frog Oocytes and Early Embryos Facilitate Biochemical Characterization of the Cell Cycle Machinery
Biochemical studies require the preparation of cell extracts from many cells. For biochemical studies of the cell cycle, the eggs and early embryos of amphibians and marine invertebrates are particularly suitable. These organisms typically have large eggs, and fertilization is followed by multiple synchronous cell cycles. By isolating large numbers of eggs from females and fertilizing them simultaneously by addition of sperm (or by treating them in ways that mimic fertilization), researchers can obtain extracts from cells at specific points in the cell cycle for analysis of proteins and enzymatic activities.
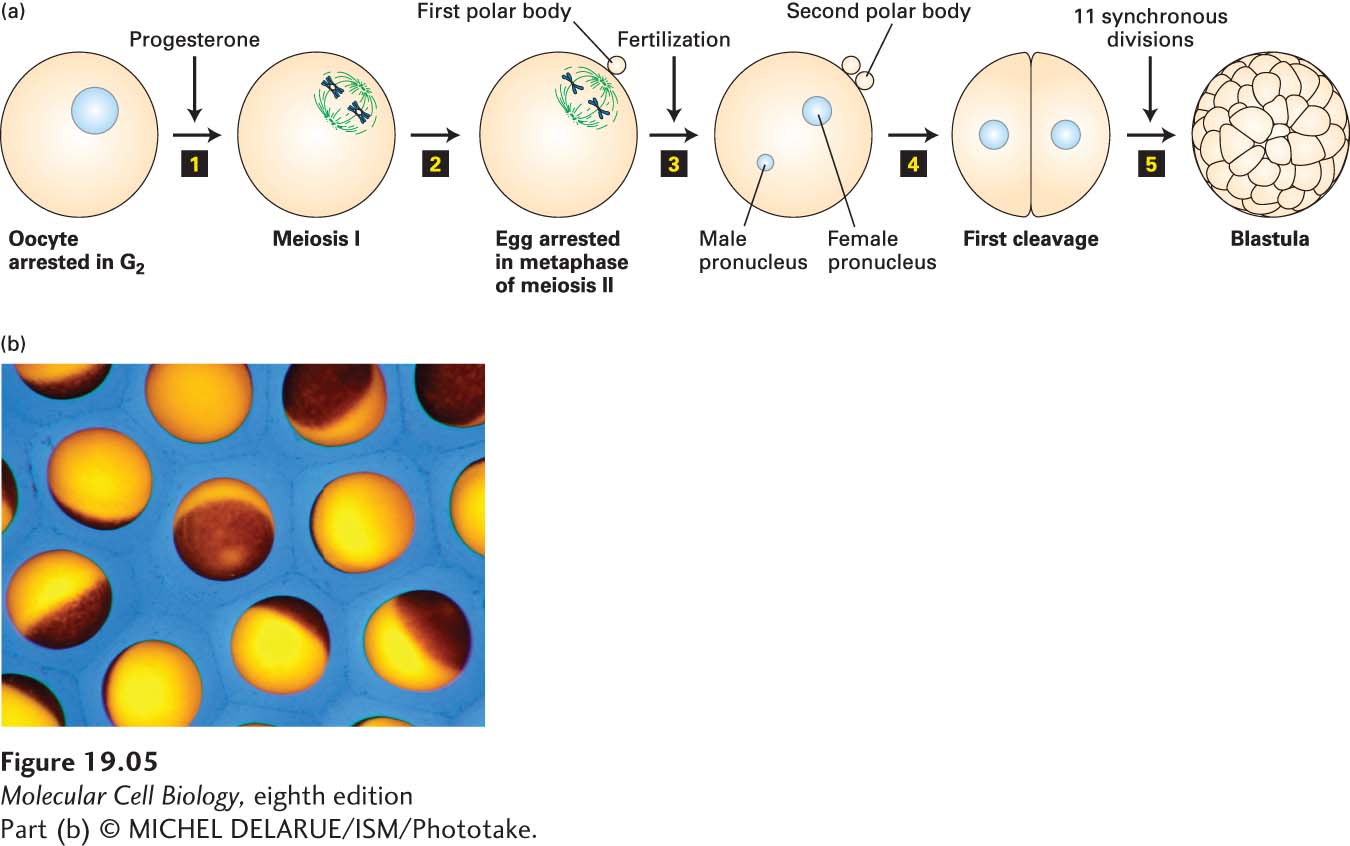
To understand how X. laevis oocytes and eggs can be used for the analysis of cell cycle progression, we must first lay out the events of oocyte maturation, which can be recapitulated in vitro. So far, we have discussed mitotic division. Oocytes, however, undergo a meiotic division (see Figure 19-35 for an overview of meiosis). As oocytes develop in the frog ovary, they replicate their DNA and become arrested in G2 for 8 months, during which time they grow in size to a diameter of 1 mm, stockpiling all the materials needed for the multiple cell divisions of the early embryo. When stimulated by a male, an adult female’s ovarian cells secrete the steroid hormone progesterone, which induces the G2-arrested oocytes to mature and enter meiosis. As we will see in Section 19.8, meiosis consists of two consecutive chromosome segregation phases known as meiosis I and meiosis II. Progesterone triggers oocytes to undergo meiosis I and progress to the second meiotic metaphase, where they arrest and await fertilization (Figure 19-5). At this stage the cells are called eggs. When fertilized by sperm, the egg nucleus is released from its metaphase II arrest and completes meiosis. The resulting haploid egg nucleus then fuses with the haploid sperm nucleus, producing a diploid zygote nucleus. DNA replication follows, and the first mitotic division of embryogenesis begins. The resulting embryonic cells then proceed through 11 more rapid, synchronous cell cycles, generating a hollow sphere of cells called the blastula. Cell division then slows, and subsequent divisions are non-
Page 879
The advantage of using X. laevis to study factors involved in mitosis is that large numbers of oocytes and eggs can be prepared that are all proceeding synchronously through the cell cycle events that follow progesterone treatment and fertilization. This makes it possible to prepare sufficient amounts of extract for biochemical experiments from cells that were all at the same point in the cell cycle. It was in this system that the cyclin-