Review the Concepts
1. The gecko is a reptile with an amazing ability to climb smooth surfaces, including glass. Geckos appear to stick to smooth surfaces via van der Waals interactions between septa on their feet and the smooth surface. How is this method of stickiness advantageous over covalent interactions? Given that van der Waals forces are among the weakest molecular interactions, how can the gecko’s feet stick so effectively?
2. The K+ channel is an example of a transmembrane protein (a protein that spans the phospholipid bilayer of the plasma membrane). What types of amino acids are likely to be found (a) lining the channel through which K+ passes, (b) in contact with the hydrophobic core of the phospholipid bilayer containing fatty acyl groups, (c) in the cytosolic domain of the protein, and (d) in the extracellular domain of the protein?
3. V-
4. Disulfide bonds help to stabilize the three-
5. In the 1960s, the drug thalidomide was prescribed to pregnant women to treat morning sickness. However, thalidomide caused severe limb defects in the children of some women who took the drug, and its use for morning sickness was discontinued. It is now known that thalidomide was administered as a mixture of two stereoisomeric compounds, one of which relieved morning sickness and the other of which was responsible for the birth defects. What are stereoisomers? Why might two such closely related compounds have such different physiological effects?
6. Name the compound shown below.
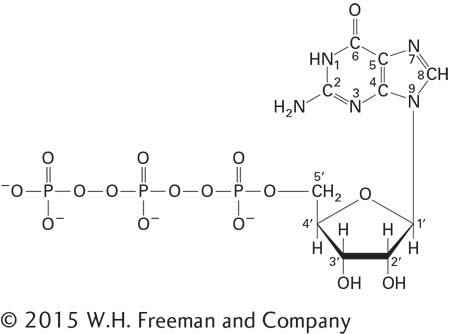
Page 66
Is this nucleotide a component of DNA, RNA, or both? Name one other function of this compound.
7. The chemical basis of blood-
8. Calculate the pH of 1 L of pure water at equilibrium. How will the pH change after 0.008 moles of the strong base sodium hydroxide (NaOH) are dissolved in the water? Now, calculate the pH of a 50 mM aqueous solution of the weak acid 3-
9. Ammonia (NH3) is a weak base that under acidic conditions becomes protonated to the ammonium ion in the following reaction:
NH3 + H+ → NH4+
NH3 freely permeates biological membranes, including those of lysosomes. The lysosome is a subcellular organelle with a pH of about 4.5–
10. Consider the binding reaction L + R → LR, where L is a ligand and R is its receptor. When 1 × 10–3 M of L is added to a solution containing 5 × 10–2 M of R, 90 percent of the L binds to form LR. What is the Keq of this reaction? How will the Keq be affected by the addition of a protein that facilitates (catalyzes) this binding reaction? What is the dissociation equilibrium constant Kd?
11. What is the ionization state of phosphoric acid in the cytoplasm? Why is phosphoric acid such a physiologically important compound?
12. The ΔG°′ for the reaction X + Y → XY is –1000 cal/mol. What is the ΔG at 25 °C (298 °Kelvin) starting with 0.01 M each of X, Y, and XY? Suggest two ways one could make this reaction energetically favorable.
13. According to health experts, saturated fatty acids, which come from animal fats, are a major factor contributing to coronary heart disease. What distinguishes a saturated fatty acid from an unsaturated fatty acid, and to what does the term saturated refer? Recently, trans unsaturated fatty acids, or trans fats, which raise total cholesterol levels in the body, have also been implicated in heart disease. How does the cis stereoisomer differ from the trans configuration, and what effect does the cis configuration have on the structure of the fatty acid chain?
14. Chemical modifications of amino acids contribute to the diversity and function of proteins. For instance, γ-carboxylation of specific amino acids is required to make some proteins biologically active. What particular amino acid undergoes this modification, and what is its biological relevance? Warfarin, a derivative of coumarin, which is present in many plants, inhibits γ-carboxylation of this amino acid and was used in the past as a rat poison. At present, it is also used clinically in humans. What patients might be prescribed warfarin and why?