 Overall Progress: 0%
Overall Progress: 0%

Chapter 1. Diabetes
1.1 Introduction
Diabetes
Glucose Metabolism Case Study
Popup content goes in this box
New box content

Aaron Sykes, the newest intern in the urgent care department at Cook County General Hospital, is seeing patients with the attending pediatrics physician, Dr. Erika Gonzalez. Their first patient of the day is Cecile, a 10-year-old girl brought in by her parents when she complained of blurred vision, fatigue, and excessive thirst.
"Good morning, Cecile, how are you feeling today?" Dr. Gonzalez asks as she enters the examination room.
"Better," Cecile replies, "although I am still a little thirsty."
"Okay, we will get you some water to help with your thirst," Dr. Gonzalez replies. She turns and greets the parents and then says, "The nurse will be here in a few minutes to draw some blood because we need to run a few tests. Try to relax and we will be back this afternoon with a better picture of what is going on."
Drs. Sykes and Gonzalez step into the hallway and review the patient history on the medical forms.
Dr. Sykes states, "Cecile has been experiencing several symptoms, including excessive thirst, frequent urination, blurred vision, and fatigue. Her parents had noticed changes over the past month or so, but when she complained about her vision they decided to bring her to the hospital."
Dr. Gonzalez says, "These are classic signs of diabetes. To confirm this diagnosis, and also to establish whether it is type 1 or type 2, we need to do bloodwork and determine what her glucose levels are."
1.2 Glucose Metabolism
Glucose Metabolism
Glucose is an important energy source that is used in both prokaryotes and eukaryotes. In mammalian cells, glucose enters the glycolytic pathway, and products are formed that will be used in the tricarboxylic acid (TCA) cycle within the mitochondria. Ultimately, products of the TCA cycle will feed into the electron transport chain to produce ATP for energy. In the context of diabetes, if the cells of the body are unable to efficiently extract glucose from the blood, then these downstream biochemical pathways will be negatively affected. In addition, high glucose levels in the blood, stemming from an inability to be taken up by cells, can have deleterious effects as well. Learn more about the pathophysiology of diabetes at: http://www.news-medical.net/health/Diabetes-Pathophysiology.aspx
Figure 12-3 shows the glycolytic pathway.
1.
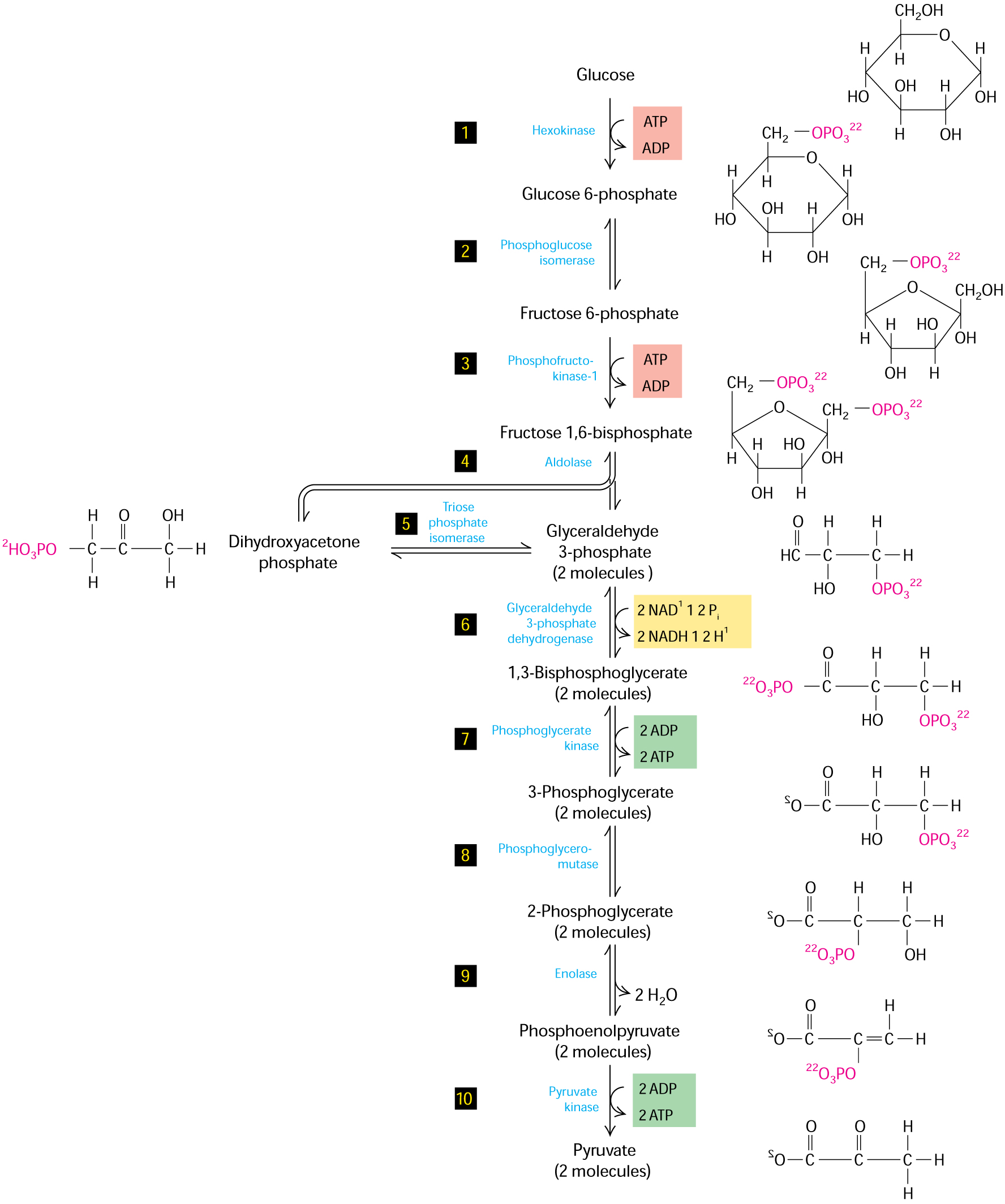
During glycolysis, glucose is converted to pyruvate in the cytosol. What important product(s) is/are generated in these metabolic reactions would be reduced in patients with diabetes?
| A. |
| B. |
| C. |
| D. |
| E. |
2.
In the glycolytic pathway shown above, which two molecules represent high-energy phosphate donors that are used for substrate-level phosphorylation reactions?
| A. |
| B. |
| C. |
| D. |
| E. |
3.
The pancreas is responsible for glucose metabolism and contains various types of secretory cells devoted to this goal. The α cells express glucagon to stimulate an increase in glucose production; β cells secrete insulin to promote uptake of glucose in skeletal muscle and fat cells; δ cells secrete somatostatin to regulate α and β cells; and γ cells secrete pancreatic polypeptide. Certain tissues are responsive to glucose uptake dependent on insulin levels, while others can take up glucose independent of insulin. Glucose enters all cells by facilitated diffusion mediated by the GLUT family of proteins.
β cells of the pancreas need to sample the glucose levels of the blood in order to regulate insulin secretion. Which GLUT protein is predominantly found on the surface of β cells to allow for the facilitated diffusion of glucose into the cell?
| A. |
| B. |
| C. |
| D. |
| E. |
4.
After a meal is ingested and blood glucose levels rise above 5 mmol/L, the β cells of the pancreas responds by secreting insulin. What event stimulates the fusion of insulin-containing secretory vesicles with the plasma membrane of the β cells?
| A. |
| B. |
| C. |
| D. |
| E. |
GLUT2 is expressed primarily on β cells and liver cells and is not dependent on insulin for mobilization to the cell surface. Influx of glucose leads to production of ATP through ATP synthase in the mitochondria, and ultimately to the influx of calcium which causes the release of insulin into the blood.
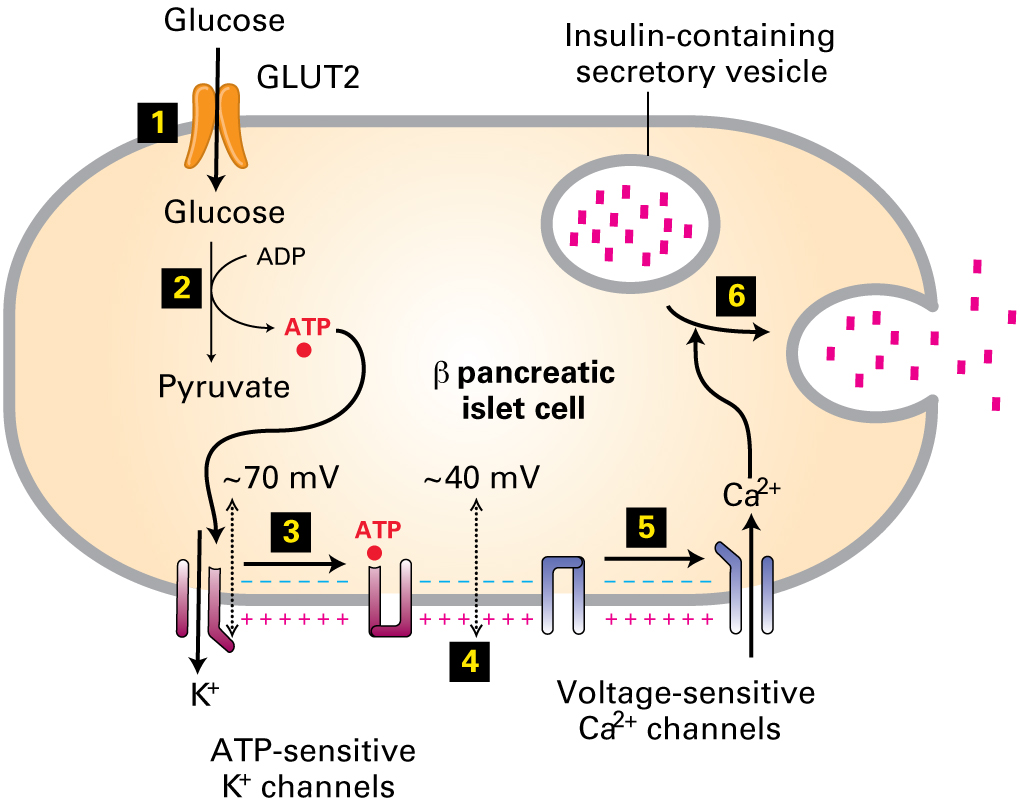
GLUT insulin-dependent proteins (e.g., GLUT4 in skeletal muscle and adipocytes) are normally localized in vesicles under the plasma membrane. Insulin signaling through the insulin receptor then causes activation of a cell-signaling pathway through Akt and subsequent mobilization of the vesicles to the cell surface. Once the GLUT proteins are situated in the plasma membrane, glucose can move with the concentration gradient into the cell.
5.
How would the cellular phenotype from a loss-of-function mutation in GLUT2 compare with diabetes?
Two of the clinical signs of diabetes include excessive thirst and increased urination. This stems from the fact that glucose is normally filtered in the Bowman’s capsule in the kidney, but completely resorbed from the kidney tubules by the blood. When excess glucose occurs from diabetes, there is retention of glucose in the urine, which in turn tends to pull in excess water because of osmosis and increases urination. The loss of water in the blood due to osmosis causes the patient becomes thirsty to replace the lost water.
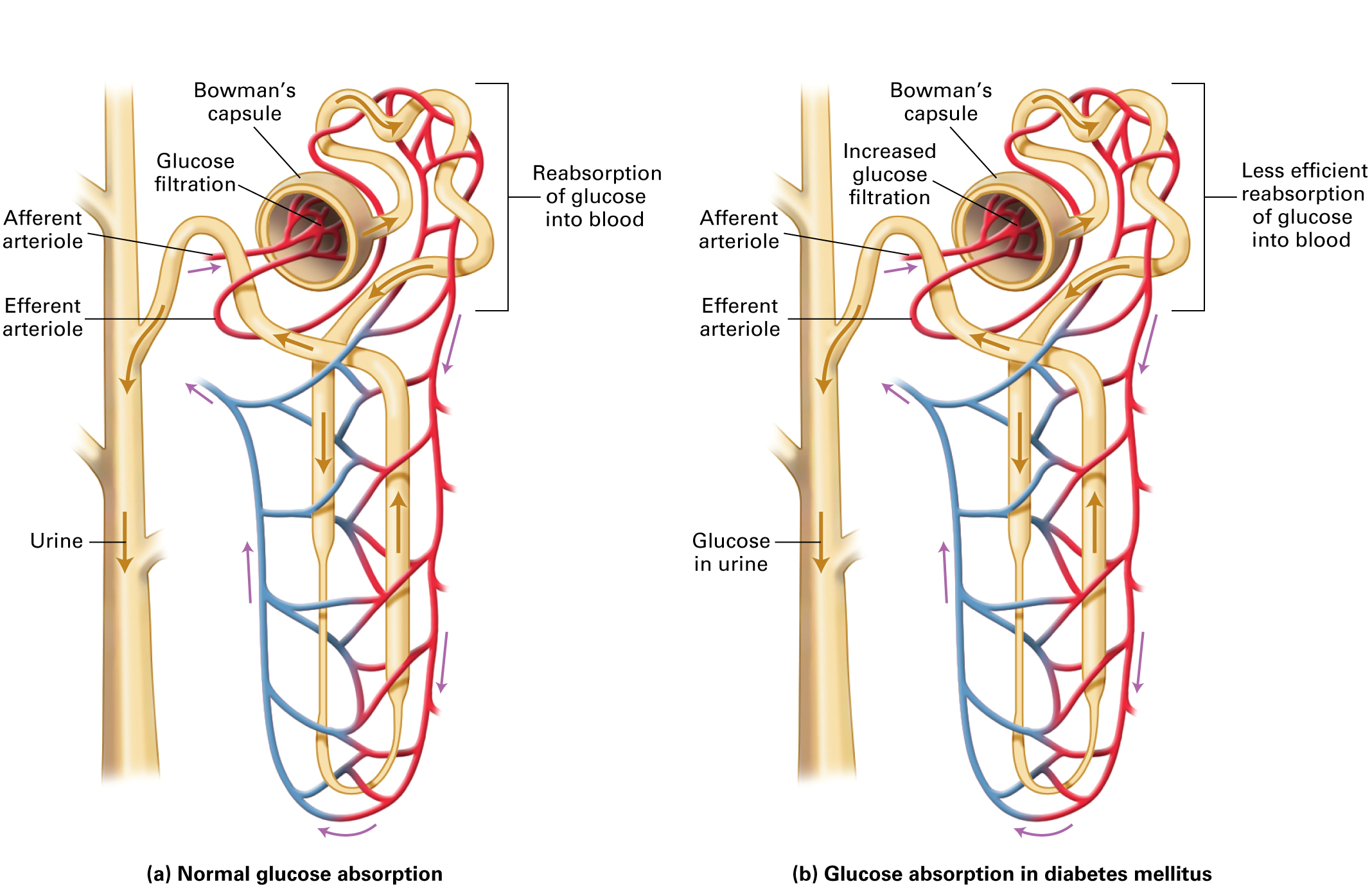
1.3 Blood Laboratory
Blood Laboratory
Dr. Sykes considered the four standard blood tests to screen for diabetes: (1) the glycated hemoglobin (A1C) test, (2) the fasting plasma glucose test (FPG), (3) the oral glucose tolerance test (OGTT), and (4) the random plasma glucose test (RPG). Read about these tests and their pros and cons at: http://www.niddk.nih.gov/health-information/health-topics/diagnostic-tests/comparing-tests-diabetes-prediabetes/Pages/index.aspx
In type 1 diabetes the patient is not able to produce sufficient levels of insulin; in type 2 diabetes the patient produces sufficient insulin, but the cells do not respond to appropriately to insulin in the blood.
6.
If the doctors are trying to discriminate between type 1 diabetes (loss of insulin production) and type 2 diabetes (resistance to insulin) which test would NOT be advisable to use for the diagnosis?
| A. |
| B. |
| C. |
| D. |
| E. |
7.
Dr. Sykes decides to order the OGTT test, which requires Cecile to fast the night before, then drink a glucose-based liquid. Blood samples are drawn immediately before drinking (zero time point), then at 30-minute intervals for 2 hours. The glucose levels are then determined from the blood samples.
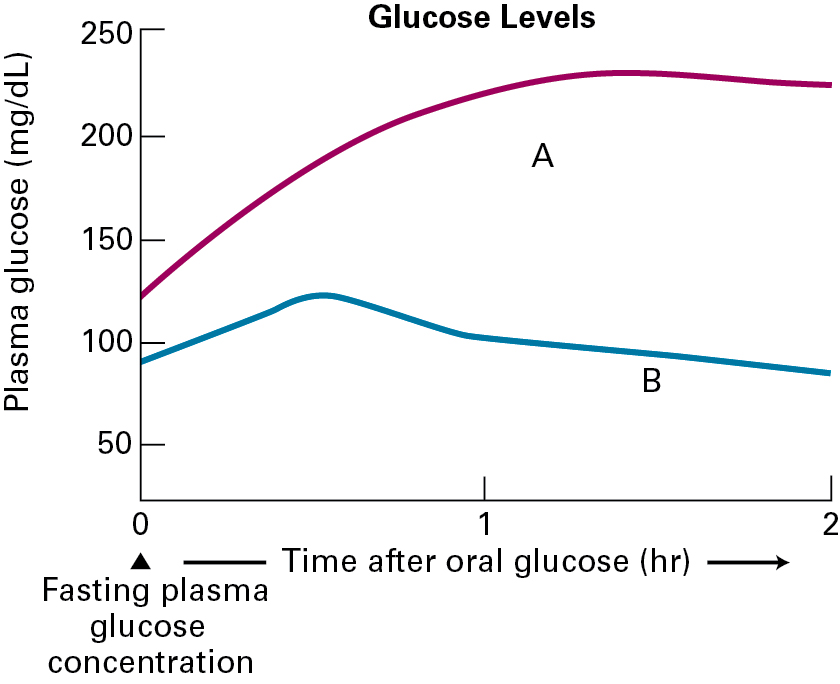
The above graph shows the OGTT assay for glucose levels of a control patient without diabetes and for a patient with diabetes. Which tracing represents the diabetic patient’s glucose levels?
| A. |
| B. |
For the OGTT assay, a glucose level above 200 mg/dL is an indicator for diabetes. Cecile’s OGTT assay shows a glucose level peaking at about 225 mg/dL. Dr. Sykes decides to test insulin levels from the same blood samples that were isolated at the time points described for the glucose test. The results of this assay are shown below.
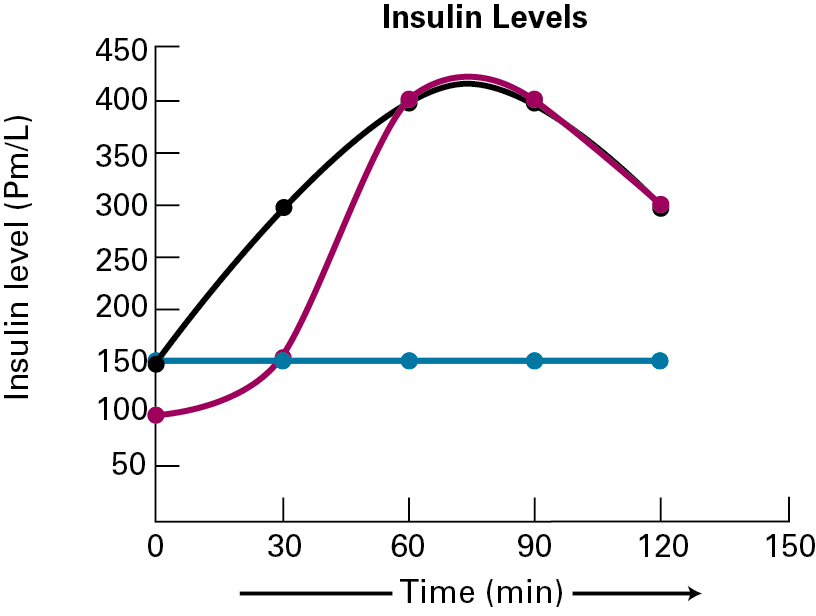
8.
Given that the black and red plots above represent the insulin levels in patients without diabetes, and the blue line is from Cecile’s blood work, what type of diabetes does she have?
| A. |
| B. |
1.4 Treatment
Treatment
Drs. Sykes and Gonzalez find that the results of the tests confirm type 1 diabetes and they discuss the diagnosis with Cecile and her parents. They mention that the most effective treatment strategy involves: (1) taking insulin, (2) counting carbohydrates, (3) frequently monitoring blood sugar levels, (4) eating healthy foods, and (5) exercising regularly and maintaining a healthy weight.
Dr. Gonzalez states, "Exercise and diet play a role, but monitoring sugar levels and taking insulin are very important in making sure that you don't have diabetes-related health problems in the future, Cecile. Insulin can be directly injected, or administered through a pump."
Dr. Sykes added, "Soon it will be possible to use an artificial pancreas that links a continuous glucose monitor to an insulin pump. Clinical trials have actually started on these devices."
The family decides to go with the insulin pump, and move to the artificial pancreas once it has been more thoroughly tested. Dr. Gonzalez also mentions the future possibility of stem cells placed into a scaffold, which would develop into β-pancreatic cells. The cells could respond to glucose levels and secrete insulin when needed.
9.
Autoimmmune diseases stem from the failure of the immune system to eliminate T cells that react to proteins on the body’s own cells. In type 1 diabetes, CD4+ and CD8+ T cells can attack β-pancreatic cells, and B cells can produce antibodies against β-pancreatic cell proteins. Although new β-pancreatic cells can be made, more cells are destroyed than produced. After about an 80% reduction in the β-pancreatic cells, insulin production is insufficient to meet the demands of the body, and type 1 diabetes symptoms result.
CD4+ T cells express FasL, which can interact with Fas on β-pancreatic cells to trigger apoptosis. What initiator caspase is recruited to adaptors at the cytoplasmic domain of activated Fas?
Answer options:
| A. |
| B. |
| C. |
| D. |
| E. |
10.
As Dr. Gonzalez mentioned, one potential avenue for a cure for type 1 diabetes is the development of a scaffold for stem cells that would differentiate into β-pancreatic cells. These replacement cells would essentially serve as a replacement pancreas.
What property would be useful in a stem cell scaffold designed to treat type 1 diabetes?
| A. |
| B. |
| C. |
| D. |
| E. |
11.
Secreted insulin will activate insulin receptors on different cells, like skeletal muscle cells and adipocytes. What is the primary cytoplasmic kinase activated by insulin signaling within these target cells to facilitate uptake of glucose?
| A. |
| B. |
| C. |
| D. |
| E. |
Once PKB (Akt) is activated in skeletal muscle or fat cells in response to insulin signaling, this protein phosphorylates a downstream protein that causes the rapid fusion of intracellular vesicles containing GLUT4 with the plasma membrane. These cells are then able to take up glucose from the blood by facilitated transport with the concentration gradient (higher glucose levels in blood than in cells).
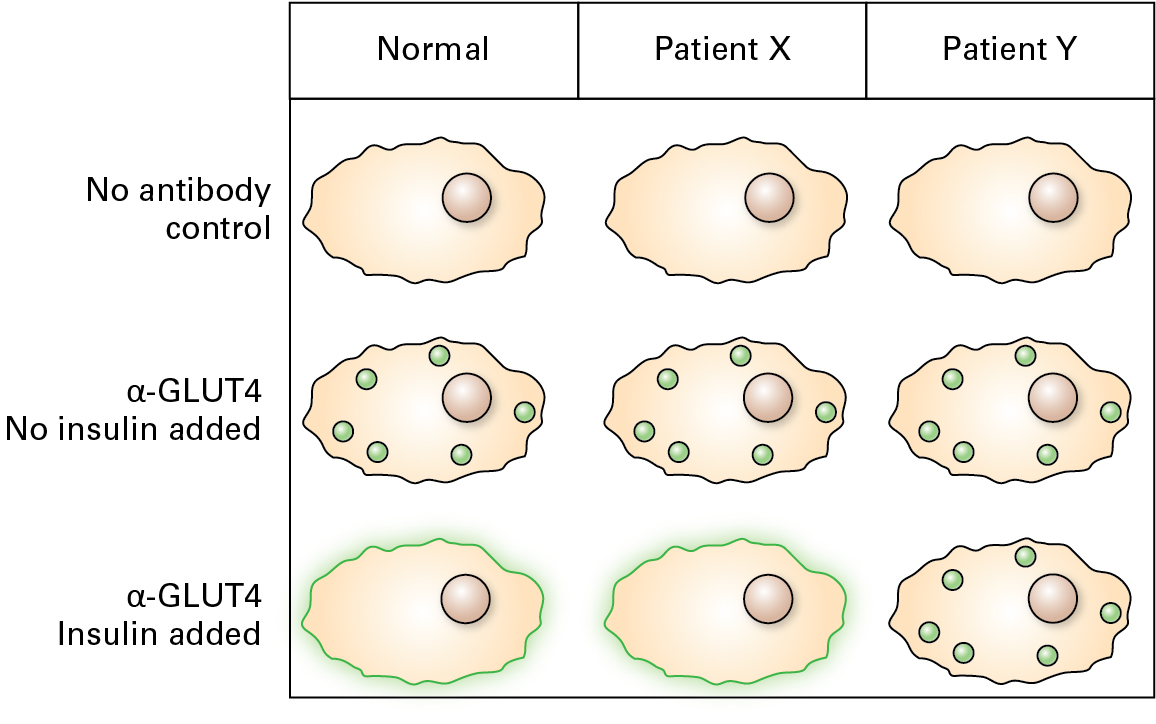
12.
Given the localization profile of GLUT4 above for skeletal muscle biopsied from Patient X and Patient Y, which would represent the expected phenotype from a patient with type 1 diabetes? With type 2 diabetes?
| Type 1: | |
| Type 2: |
1.5 Additional Information
Additional Information: Type 1 Diabetes
Diagnosis
Type 1 diabetes signs and symptoms can come on quickly and may include:
- Increased thirst
- Frequent urination
- Bedwetting in children who previously didn't wet the bed during the night
- Extreme hunger
- Unintended weight loss
- Irritability and other mood changes
- Fatigue and weakness
- Blurred vision
http://www.mayoclinic.org/diseases-conditions/type-1-diabetes/basics/symptoms/con-20019573
Risk Factors:
- Family history. Anyone with a parent or sibling with type 1 diabetes has a slightly increased risk of developing the condition.
- Genetics. The presence of certain genes indicates an increased risk of developing type 1 diabetes.
- Geography. The incidence of type 1 diabetes tends to increase as you travel away from the equator. People living in Finland and Sardinia have the highest incidence of type 1 diabetes — about two to three times higher than rates in the United States and 400 times the incidence among people living in Venezuela.
- Age. Although type 1 diabetes can appear at any age, it appears at two noticeable peaks. The first peak occurs in children between 4 and 7 years old, and the second is in children between 10 and 14 years old.
http://www.mayoclinic.org/diseases-conditions/type-1-diabetes/basics/risk-factors/con-20019573
Complications:
- Heart and blood vessel disease. Diabetes dramatically increases your risk of various cardiovascular problems, including coronary artery disease with chest pain (angina), heart attack, stroke, narrowing of the arteries (atherosclerosis) and high blood pressure.
- Nerve damage (neuropathy). Excess sugar can injure the walls of the tiny blood vessels (capillaries) that nourish your nerves, especially in the legs. This can cause tingling, numbness, burning, or pain that usually begins at the tips of the toes or fingers and gradually spreads upward. Poorly controlled blood sugar could cause you to eventually lose all sense of feeling in the affected limbs. Damage to the nerves that affect the gastrointestinal tract can cause problems with nausea, vomiting, diarrhea, or constipation. For men, erectile dysfunction may be an issue.
- Kidney damage (nephropathy). The kidneys contain millions of tiny blood vessel clusters that filter waste from your blood. Diabetes can damage this delicate filtering system. Severe damage can lead to kidney failure or irreversible end-stage kidney disease, which requires dialysis or a kidney transplant.
- Eye damage. Diabetes can damage the blood vessels of the retina (diabetic retinopathy), potentially leading to blindness. Diabetes also increases the risk of other serious vision conditions, such as cataracts and glaucoma.
- Foot damage. Nerve damage in the feet or poor blood flow to the feet increases the risk of various foot complications. Left untreated, cuts and blisters can become serious infections, which often heal poorly and may ultimately require toe, foot, or leg amputation.
- Skin and mouth conditions. Diabetes may leave you more susceptible to skin problems, including bacterial and fungal infections.
- Pregnancy complications. High blood sugar levels can be dangerous for both the mother and the baby. The risk of miscarriage, stillbirth, and birth defects are increased when diabetes isn't well-controlled. For the mother, diabetes increases the risk of diabetic ketoacidosis, diabetic eye problems (retinopathy), pregnancy-induced high blood pressure, and preeclampsia.
http://www.mayoclinic.org/diseases-conditions/type-1-diabetes/basics/complications/con-20019573
Tests and diagnosis:
- Glycated hemoglobin (A1C) test. This blood test indicates your average blood sugar level for the past two to three months. It works by measuring the percentage of blood sugar attached to hemoglobin, the oxygen-carrying protein in red blood cells. The higher your blood sugar levels, the more hemoglobin you'll have with sugar attached. An A1C level of 6.5 percent or higher on two separate tests indicates you have diabetes.
- Random blood sugar test. A blood sample will be taken at a random time. Blood sugar values are expressed in milligrams per deciliter (mg/dL) or millimoles per liter (mmol/L). Regardless of when you last ate, a random blood sugar level of 200 mg/dL (11.1 mmol/L) or higher suggests diabetes, especially when coupled with any of the signs and symptoms of diabetes, such as frequent urination and extreme thirst.
- Fasting blood sugar test. A blood sample will be taken after an overnight fast. A fasting blood sugar level of less than 100 mg/dL (5.6 mmol/L) is normal. A fasting blood sugar level from 100 to 125 mg/dL (5.6 to 6.9 mmol/L) is considered prediabetes. If it's 126 mg/dL (7 mmol/L) or higher on two separate tests, you have diabetes.
http://www.mayoclinic.org/diseases-conditions/type-1-diabetes/basics/tests-diagnosis/con-20019573
Treatment:
- Taking insulin
- Counting carbohydrates
- Frequently monitoring blood sugar
- Eating healthy foods
- Exercising regularly and maintaining a healthy weight
Insulin can't be taken orally to lower blood sugar because stomach enzymes interfere with insulin's action. Therefore, it must be given either through injections or an insulin pump.
Injections: You can use a fine needle and syringe or an insulin pen to inject insulin under your skin. Insulin pens look similar to ink pens, and are available in disposable or refillable varieties. Needles are available in a variety of sizes, so you can find one that's most comfortable for you.
An insulin pump: A device about the size of a cellphone worn on the outside of your body. A tube connects a reservoir of insulin to a catheter that's inserted under the skin of your abdomen. This type of pump can be worn in a variety of ways, such as on your waistband, in your pocket, or with specially designed pump belts.
Artificial pancreas: An emerging treatment approach, not yet available, is closed-loop insulin delivery, also known as the artificial pancreas. It links a continuous glucose monitor to an insulin pump. The device automatically delivers the correct amount of insulin when the monitor indicates the need for it. There are a number of different versions of the artificial pancreas, and clinical trials have had encouraging results. Studies on the device found that it could prevent low blood sugar levels overnight without significantly increasing morning blood sugar levels. More research needs to be done before a fully functional artificial pancreas can receive regulatory approval. The first step toward an artificial pancreas was approved in 2013.
http://www.mayoclinic.org/diseases-conditions/type-1-diabetes/basics/treatment/con-20019573
Investigational treatments:
- Pancreas transplant. With a successful pancreas transplant, you would no longer need insulin. But pancreas transplants aren't always successful — and the procedure poses serious risks. You would need a lifetime of potent immune-suppressing drugs to prevent organ rejection. These drugs can have serious side effects, including a high risk of infection and organ injury. Because the side effects can be more dangerous than the diabetes itself, pancreas transplants are generally reserved for those with very difficult-to-manage diabetes, or for people who also need a kidney transplant.
- Islet cell transplant. Researchers are experimenting with islet cell transplantation, which provides new insulin-producing cells from a donor pancreas. Although this experimental procedure has met with problems in the past, new techniques and better drugs to prevent islet cell rejection may improve its future chance for success.
Islet cell transplantation still requires the use of immune-suppressing medications. And just as it did with its own natural islet cells, the body often destroys transplanted islet cells, making the time off injected insulin short-lived. Additionally, a sufficient supply of islet cells isn't available for this treatment to become more widespread.
Researchers are working on ways to expand the number of available islets, as well as find ways to protect islets from the immune system. Some ideas that may soon be in clinical trials include encapsulating the individual islet cells, or housing the islet cells in a device that would shield them from the immune cells, but still allow in oxygen and a blood supply.
- Stem cell transplant. In a 2007 study, a small number of people with a new diagnosis of type 1 diabetes were able to stop using insulin for up to five years after being treated with stem cells made from their own blood. Although stem cell transplants — which involve shutting down the immune system and then building it up again — can be risky, the technique may one day provide an additional treatment option for patients with type 1 diabetes.
http://www.mayoclinic.org/diseases-conditions/type-1-diabetes/basics/treatment/con-20019573

Your grade of 0% has been submitted.