
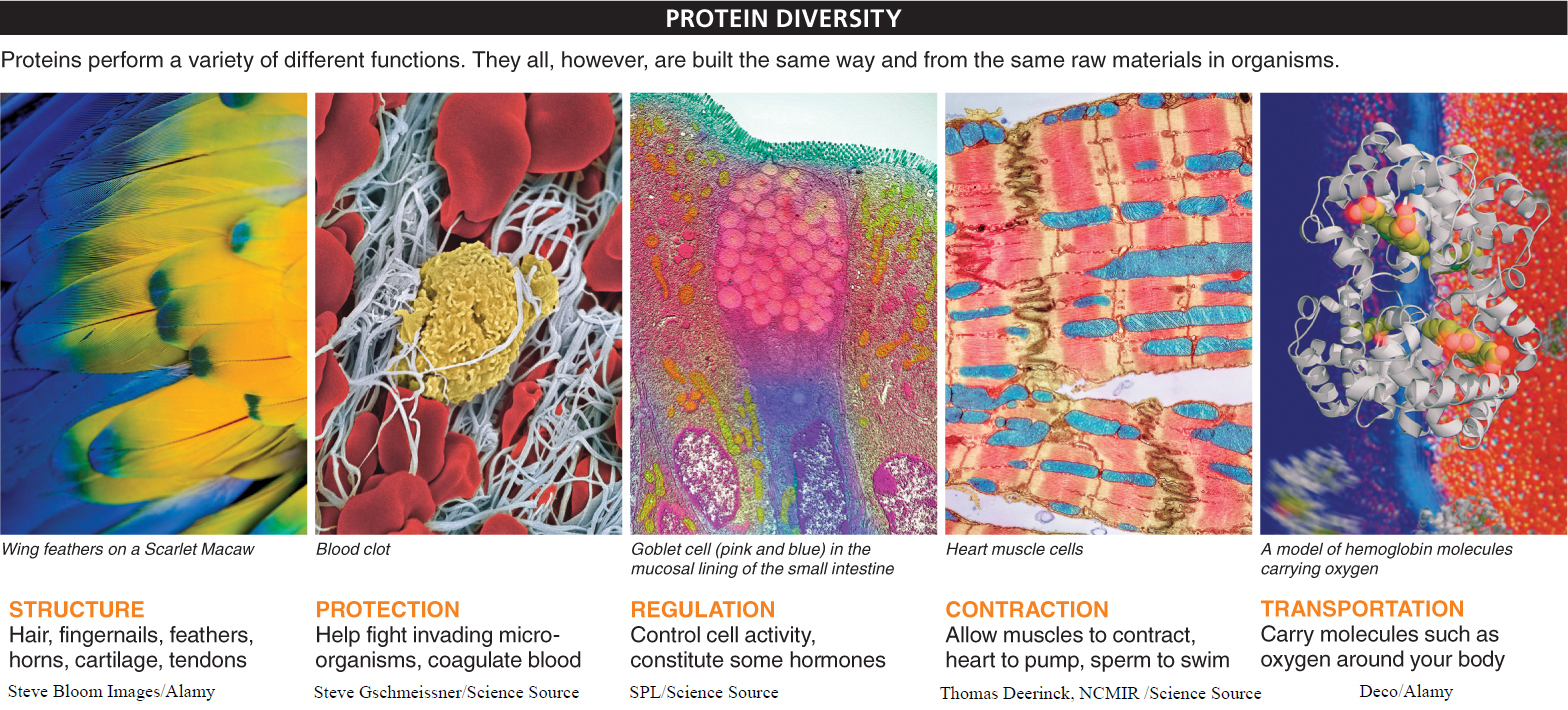
You can’t look at a living organism and not see proteins (FIGURE 2-36). Inside and out, proteins are the chief building blocks of all life. They make up skin and feathers and horns. They make up muscles and are a significant component of bone. In your bloodstream, proteins fight invading microorganisms and stop you from bleeding to death from a shaving cut. Proteins control the levels of sugar and other chemicals in your bloodstream and carry oxygen from one place in your body to another. And in just about every cell in every living organism, proteins called enzymes initiate and assist every chemical reaction that occurs.
68
Although proteins perform several very different types of functions, all are built in the same way and from the same raw materials in all organisms. In the English language, every sentence is made up of words and every word is formed from one or more of the 26 letters of the alphabet. With 26 letters we can write anything, from sonnets to cookbooks to biology textbooks. Proteins, too, are constructed from a sort of alphabet. Instead of 26 letters there are 20 molecules, known as amino acids. Unique combinations of these 20 amino acids are strung together, like beads on a string, and the resulting protein has a unique structure and chemical behavior.
Let’s look more closely at the structure of the amino acids in the protein alphabet. They all have the same basic two-
Amino acids contain the same familiar atoms as carbohydrates and lipids—
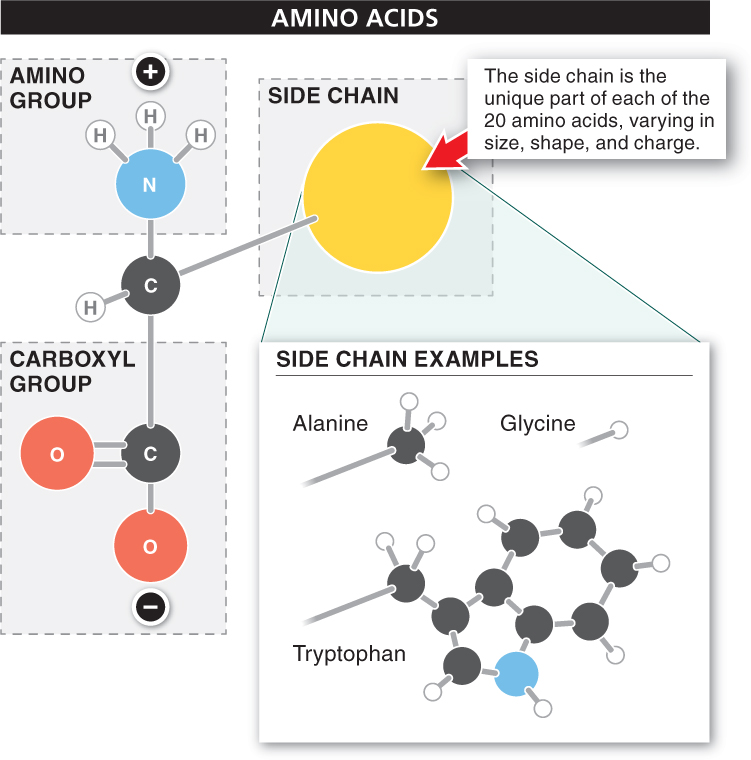
The fourth bond of the central carbon atom attaches to a side chain. This side chain is the unique part of each of the 20 amino acids. In the simplest amino acid, glycine, for example, the side chain is simply a hydrogen atom. In other amino acids, the side chain is a single CH3 group or three or four such groups. Most of the side chains include both hydrogen and carbon atoms, and a few include nitrogen or sulfur atoms. The side chain determines an amino acid’s chemical properties, such as whether the amino acid molecule is polar or nonpolar.
TAKE-HOME MESSAGE 2.15
Unique combinations of 20 amino acids give rise to proteins, the chief building blocks of the physical structures that make up all organisms. Proteins perform myriad functions, from assisting chemical reactions to causing blood clotting to building bones to fighting microorganisms.
List six different functions of proteins.
The textbook lists structural, protective, regulatory, contractile, transport, and enzyme roles for proteins. Structural proteins form hair, fingernails, feathers, and other structures in organisms. Protective proteins help fight invading microorganisms. Regulatory proteins, such as hormones, control cell activity. Contractile proteins allow muscles to contract and the heart to pump. Transport proteins carry molecules such as oxygen around the body.