4.2: Energy has two forms: kinetic and potential.
“Batteries not included.” For a child, those are pretty depressing words. We know that many of the toys and electronic gadgets that make our lives fun or useful (or both) need energy—
Energy is the capacity to do work. And work is anything that involves moving matter against an opposing force. In the study of living things, we encounter two types of energy: kinetic and potential. Kinetic energy is the energy of motion. The kinetic energy of an object is the energy that it has due to its motion. Legs pushing bike pedals and birds flapping wings are examples of kinetic energy (FIGURE 4-3). Heat, which results from lots of molecules moving rapidly, is another form of kinetic energy. Because it comes from the movement of high-

138
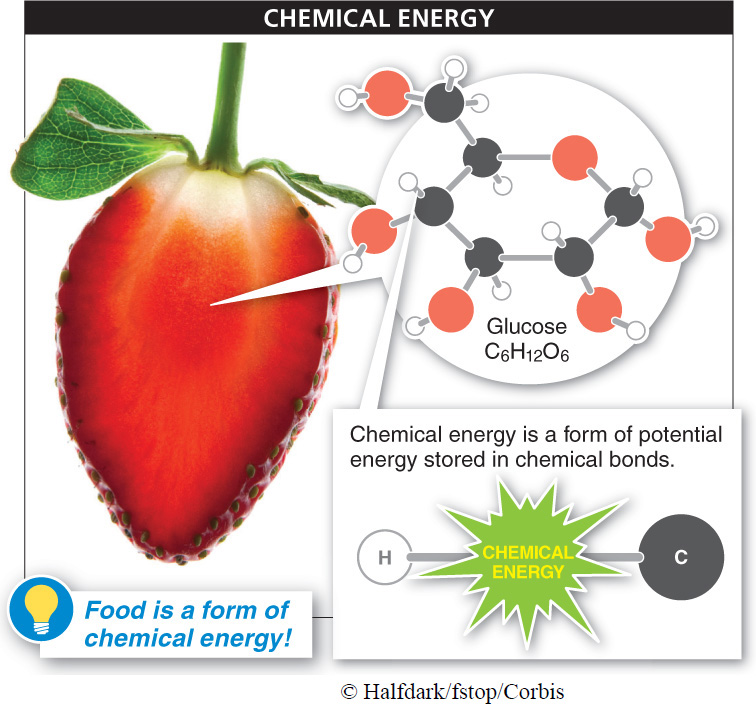
An object does not have to be moving to have the capacity to do work; it may have potential energy, which is stored energy that results from an object’s location or position. Water behind a dam, for example, has potential energy. If a hole is opened in the dam, the water can flow through, and perhaps spin a waterwheel or turbine. A concentration gradient, which we discussed in Chapter 3, also has potential energy: if the molecules in an area of high concentration move toward an area of lower concentration, the potential energy of the gradient is converted to the kinetic energy of molecular movement, and this kinetic energy can do work. Chemical energy, the storage of energy in chemical bonds, is also a type of potential energy.
Because potential energy doesn’t involve movement, it is a less obvious form of energy than kinetic energy. An apple has potential energy, as does any other type of food (FIGURE 4-4). Why? Because, during cellular respiration, the chemical energy stored in the chemical bonds making up the food can be released (when those bonds are broken and lower-
139
TAKE-HOME MESSAGE 4.2
Energy, the capacity to do work, comes in two forms. Kinetic energy is the energy of moving objects, while potential energy, such as chemical energy, is stored energy that results from the position or location of an object.
Give two examples of kinetic energy and potential energy.