Operons and the Noisy Cell
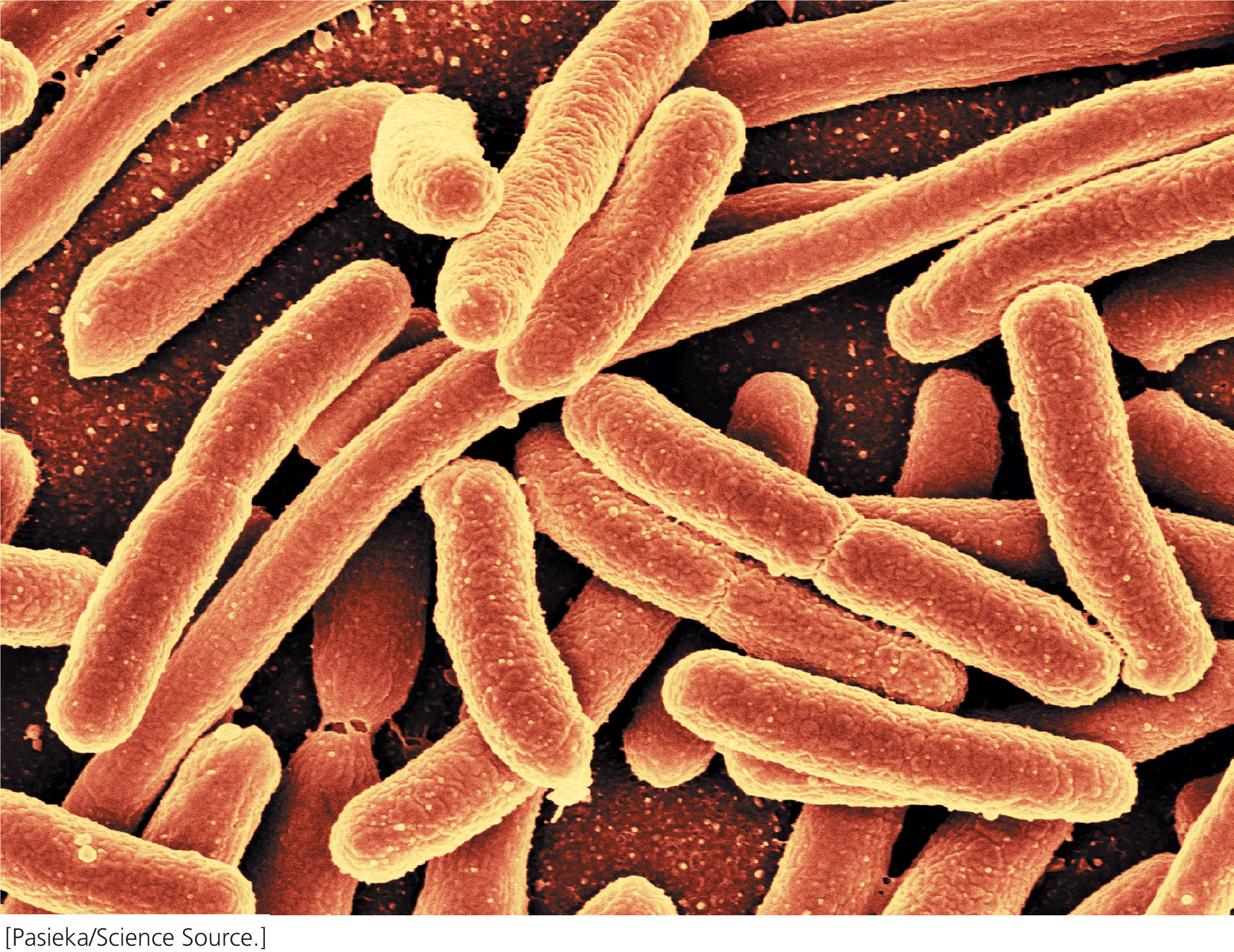
In 2011, geneticists from around the world celebrated the fiftieth anniversary of the operon. An operon is a group of genes that share a common promoter and are transcribed as a unit, producing a single mRNA that encodes several proteins. Typically, these proteins interact in some way; for example, the trp operon in the bacterium Escherichia coli encodes components of three enzymes that work together to synthesize the amino acid tryptophan. Operons control the expression of genes such as these by regulating their transcription. Genes in bacteria are often organized into operons, but operons are much less common in eukaryotes.
The operon was discovered through the elegant research of Francois Jacob and Jacques Monod, who worked at opposite ends of the attic floor of the Pasteur Institute in Paris. Jacob was studying bacteriophage lambda, a virus that infects E. coli; Monod was analyzing the properties of β-galactosidase, an enzyme E. coli uses to metabolize the sugar lactose. One summer evening in 1958, Jacob had a flash of inspiration—he saw a connection between the research taking place at each end of the attic. Jacob recognized that the genes that induced phage reproduction were controlled in the same way as the genes that control production of β-galactosidase in E. coli. This led to an important collaboration between Jacob and Monod; together they eventually uncovered the structure and function of the lac operon, and, in 1965, were awarded the Nobel Prize in Physiology or Medicine along with their collaborator Andre Lwoff.
Following Jacob and Monod’s discovery of the lac operon, other operons were discovered in bacteria and a great deal of research focused on the mechanism of operon function. Despite extensive research on how operons work, much less was known about why operons exist: why do prokaryotes have them while most eukaryotes don’t? Why are some genes included in operons and others not? These questions intrigued Oleg Igoshin at Rice University, and Christian Ray at the University of Texas MD Anderson Cancer Center. Igoshin and Ray are computational biologists, a new breed of scientists who use complex mathematics to study fundamental problems of biology. Igoshin and Ray took what might seem like an unlikely approach to the question of why operons exist. Instead of growing bacteria, inducing mutations, and examining DNA, they developed a series of mathematical models of gene networks that could be run on the computer. With these models they looked at how genes functioned when grouped into operons and when regulated separately.
Igoshin and Ray knew that random fluctuations occur naturally in the levels of transcription and translation. Because of these fluctuations—noise in the system—the amounts of different proteins can vary widely; the amount of protein produced may be more or less than is optimal for cell growth and survival. Igoshin and Ray hypothesized that coordinating the transcription of several genes through an operon structure might reduce noise in the system and permit more finely tuned control over gene expression.
To test their hypothesis, Igoshin and Ray ran computer models for six different types of interactions between the products of genes that are potentially found in operons. Their models showed that for some types of protein interactions, grouping genes together in an operon decreases the biochemical noise. For other types of protein interactions, grouping the genes in operons actually increases the noise. Thus, operon structure has the potential to increase or decrease noise depending on which genes are grouped together.
Igoshin and Ray then examined genes that are actually found in operons in E. coli, and discovered that operons with genes whose interactions decrease noise were more common than expected on a random basis. Conversely, operons whose gene interactions increase noise were less common than expected. They concluded that operons have evolved as a way for the cell to couple together transcription of genes to reduce biochemical noise in the cell, allowing the cell to more finely tune the relative proportions of proteins encoded by the operon. Igoshin and Ray speculated that operons are less common in eukaryotes because the larger cell volume reduces the effect of random fluctuations and, perhaps, because eukaryotes have other mechanisms (such as changes in chromatin structure) that couple the transcription of genes.
This is the first of two chapters about gene regulation, the mechanisms and systems that control the expression of genes. In this chapter, we consider systems of gene regulation in bacteria. We begin by considering the necessity for gene regulation, the levels at which gene expression is controlled, and the difference between genes and regulatory elements. We then examine the structure and function of operons, and consider gene regulation in some specific examples of operons, including the lac operon studied by Jacob and Monod. Finally, we will discuss several types of bacterial gene regulation that are facilitated by RNA molecules. In Chapter 17 we will discuss mechanisms of gene regulation in eukaryotic genomes.