22.5 The Study of Development Reveals Patterns and Processes of Evolution
“Ontogeny recapitulates phylogeny” is a familiar phrase that was coined in the 1860s by German zoologist Ernst Haeckel to describe his belief—now considered an oversimplication—that during their development (ontogeny) organisms repeat their evolutionary history (phylogeny). According to Haeckel’s belief, a human embryo passes through fish, amphibian, reptilian, and mammalian stages before developing human traits. Scientists have long recognized that organisms do not pass through the adult stages of their ancestors during their development, but the embryos of these related organisms often display similarities.
Common Genes in Developmental Pathways
Although ontogeny does not precisely recapitulate phylogeny, many evolutionary biologists today are turning to the study of development for a better understanding of the processes and patterns of evolution. Sometimes called “evo-devo,” the study of evolution through the analysis of development is revealing that the same genes often shape developmental pathways in distantly related organisms. Biologists once thought that segmentation in vertebrates and invertebrates was only superficially similar, but we now know that in both Drosophila and the primative chordate Branchiostoma, the engrailed gene divides the embryo into specific segments. A gene called distalless, which creates the legs of a fruit fly, plays a role in the development of crustacean branched appendages. This same gene also stimulates body outgrowths of many other organisms, from polycheate worms to starfish. Another example is the role of Pitx1 in controlling the presence and absence of pelvic spines in sticklebacks, discussed in the introduction to this chapter. This same gene is found in mice, where it also affects hind limb development, and in humans, where mutations of the gene have been associated with the development of clubfoot.
648
An amazing example of how the same genes in distantly related organisms can shape similar developmental pathways is seen in the development of eyes in fruit flies, mice, and humans. Walter Gehring and his collaborators examined the effect of the eyeless gene in Drosophila, which is required for proper development of the fruit-fly eye. Gehring and his coworkers genetically engineered cells that expressed eyeless in parts of the fly where the gene is not normally expressed. When these flies hatched, they had eyes on their wings, antennae, and legs (Figure 22.18). These structures were not just tissue that resembled eyes, but complete eyes with a cornea, cone cells, and photoreceptors that responded to light, although the flies could not use these eyes to see, because they lacked a connection to the nervous system.
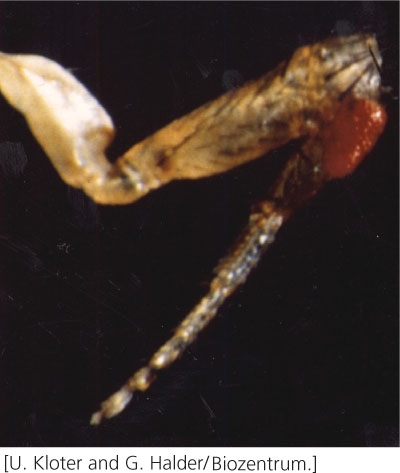
The eyeless gene has counterparts in mice and humans that affect the development of mammalian eyes. There is a striking similarity between the eyeless gene of Drosophila and the Small eye gene that exists in mice. In mice, a mutation in one copy of Small eye causes small eyes; a mouse that is homozygous for the Small eye mutation has no eyes. There is also a similarity between the eyeless gene in Drosophila and the Aniridia gene in humans; a mutation in Aniridia produces a severely malformed human eye. Similarities in the sequences of eyeless, Small eye, and Aniridia suggest that all three genes evolved from a common ancestral sequence and a common pathway underlies eye development in flies, mice, and humans. This possibility is surprising, because the eyes of insects and mammals were thought to have evolved independently.
Similar genes may be part of a developmental pathway common to two different species but have quite different effects. For example, a Hox gene called AbdB helps define the posterior end of a Drosophila embryo; a similar group of genes in birds divides the wing into three segments. In another example, the sog gene in fruit flies stimulates cells to assume a ventral orientation in the embryo, but the expression of a similar gene called chordin in vertebrates causes cells to assume dorsal orientation, exactly the opposite of the situation in fruit flies. In vertebrates, toll genes encode proteins called Toll-like receptors that bind to molecules on pathogens and stimulate the immune system. In fruit flies, the toll gene similarly functions in immunity, but it also encodes a protein that helps determine the dorsal–ventral axis, as mentioned earlier. The theme emerging from these studies is that a small, common set of genes may underlie many basic developmental processes in many different organisms.
Evolution Through Change in Gene Expression
Another concept revealed by studies in evo-devo is that many major evolutionary adaptations are not accomplished through changes in the types of proteins produced but through changes in the expression of genes that encode proteins that regulate development. This is illustrated in the introduction to this chapter, where deletion of an enhancer that stimulates the Pitx1 gene in sticklebacks is responsible for the evolution of pelvic spine reduction in freshwater populations of the fish.
This concept is also seen in the evolution of blind cavefish. The Mexican tetra, Astyanax mexicanus, normally occurs in surface waters of streams and rivers in Texas and northern Mexico. Some 10,000 years ago, a few tetras migrated into caves. In the total darkness of their cave environment, vision was not needed and, with the passage of time, these tetras lost their eyes. Today, some 30 distinct populations of Mexican tetras are totally blind and eyeless (Figure 22.19), whereas surface-dwelling populations of the same species have retained normal eye development.

649
How did Mexican tetras lose their eyes? Mexican tetra zygotes begin to develop eyes, just like their surface-dwelling cousins, but about 24 hours after fertilization, eye development aborts, and the cells that were destined to become the lens spontaneously die. The absence of the lens prevents other components of the eye, such as the cornea and iris, from developing. The optic cup and retina form, but their growth is retarded and photoreceptor cells never differentiate. The degenerate eye sinks into the orbit and is eventually covered by a flap of skin.
Blind Mexican tetras have the same genes as surface-dwelling Mexican tetras; how they differ is in gene expression. Two genes named sonic hedgehog (shh) and tiggy-winkle hedgehog (twhh) are more widely expressed in the eye primordium of blind cavefish than in surface fish. (The original hedgehog gene was named for a mutant phenotype in Drosophila, in which the mutant embryo is covered with denticles like a hedgehog. The sonic hedgehog gene is named after the video game character, and the tiggy-winkle hedgehog gene is named for Mrs. Tiggy-Winkle, a character from Beatrix Potter’s books.) The expanded expression of shh and twhh activates the transcription of other genes, which cause lens cells to undergo apoptosis, and the lens degenerates.
When geneticists injected twhh or shh mRNA into the embryos of surface fish, the development of the lens aborted, and adults that developed from these embryos were missing eyes. When drugs were used to inhibit the expression of twhh and shh in cavefish embryos, eye development in these fish was partly restored. These results demonstrate that eye development in Mexican tetras is regulated by the precise expression of shh and twhh. Overexpression of one or both of these genes in the cavefish induces the death of lens cells and aborts normal eye development. A small increase in the transcription of either gene during embryonic development results in a major anatomical change in adult fish, change that has allowed the fish to adapt to the darkness of the cave environment.  TRY PROBLEM 27
TRY PROBLEM 27
Another example of differences in gene expression bringing about evolutionary change is seen in Darwin’s finches, a group of 14 closely related birds found on the Galápagos Islands (see Figure 26.9). The birds differ primarily in the size and shape of their beaks: ground finches have deep and wide beaks, cactus finches have long and pointed beaks, and warbler finches have sharp and thin beaks. These differences are associated with diet, and evolutionary changes in beak shape and size have taken place in the past when climate changes brought about shifts in the abundance of food items.
To investigate the underlying genetic basis of these evolutionary changes, Arkaht Abzhanov and his colleagues used microarrays (see Chapter 20) to examine differences in transcription levels of several thousand genes in five species of Darwin’s finches. They found differences in the expression of a gene that encodes a protein called calmodulin (CaM); the gene that encodes CaM was more highly expressed in the long and pointed beak of cactus-finch embryos than in the beaks of the other species. CaM takes part in a process called calcium signaling, which is known to affect many aspects of development. When Abzhanov and his coworkers activated calcium signaling in developing chicken embryos, the chickens had longer beaks, like those of the cactus finch. Thus, these researchers were able to reproduce, at least in part, the evolutionary difference that distinguishes cactus finches. This experiment shows that changes in the expression of a single gene in the course of development can produce significant anatomical differences in adults.
In these studies and others, the combined efforts of developmental biologists, geneticists, and evolutionary biologists are sources of important insights into how evolution takes place. Although Haeckel’s euphonious phrase “ontogeny recapitulates phylogeny” was incorrect, evo-devo is proving that development can reveal much about the process of evolution.