Temperature limits the occurrence of aquatic life
At the beginning of this chapter, we saw that whales possess a number of adaptations that allow them to deal with the cold ocean temperatures, including thick layers of fat, high metabolism, and countercurrent circulation that exchanges heat between the warm arteries and cold veins. For aquatic organisms, most physiological processes occur only within the range of temperatures at which water is liquid. Relatively few organisms can survive temperatures above 45°C, which is the upper limit of the physiological range for most eukaryotic organisms. In this section we will look at how organisms have adapted to hot and cold temperatures.
Heat and Biological Molecules
Temperature influences physiological processes because of the way in which heat affects organic molecules. Heat imparts kinetic energy to living systems, causing biological molecules to change shape. Heat also accelerates chemical reactions by increasing the rate of molecule movement. In fact, the rate of most biological processes increases two to four times for each 10°C rise in temperature. We can see this when we examine the data for a fish known as the miiuy croaker (Miichthys miiuy), shown in Figure 2.18. The fish’s rate of oxygen consumption approximately doubles when the environmental temperature increases from 15°C to 25°C. To understand the relationship between temperature and physiological process, we calculate the ratio of the physiological rate at one temperature to the physiological rate when the temperature is 10°C cooler, a ratio that is referred to as the Q10 of the physiological process. By knowing the Q10 values of different physiological processes, we can better understand which processes are more sensitive to a change in temperature.
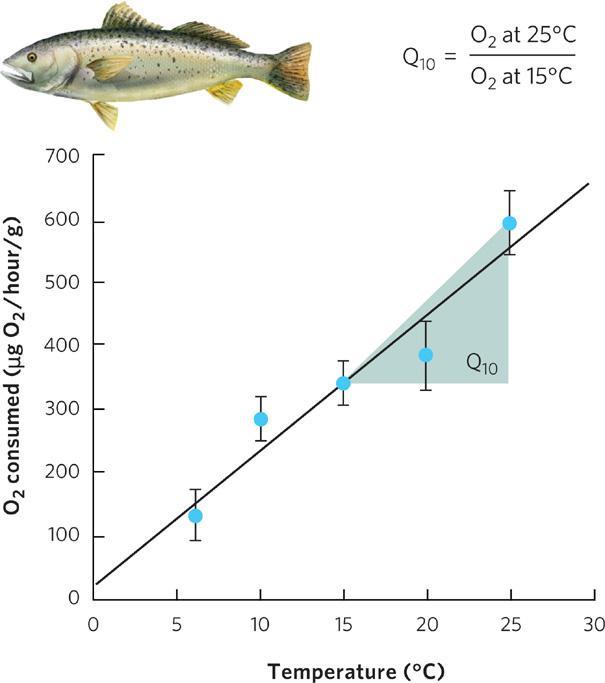
Q10 The ratio of the rate of a physiological process at one temperature to its rate at a temperature 10°C cooler.
Higher temperatures allow organisms to do many things more rapidly. They can swim, run, and fly faster. They can also digest and assimilate more food and, as a result, grow and develop faster. Beyond a certain point, however, high temperatures can depress life processes. In particular, proteins and other biological molecules become less stable at higher temperatures, and may not function properly or may lose their structure. The molecular motion caused by heat tends to open up, or denature, the structure of these molecules.
Thermophilic Heat-loving.
Given that proteins denature at high temperatures, scientists have been intrigued by the fact that some organisms, such as thermophilic (heat-loving) bacteria, can live at very high temperatures. For example, some photosynthetic bacteria can tolerate temperatures as high as 75°C and some archaebacteria can live in hot springs at temperatures up to 110°C (Figure 2.19). The chemosynthetic bacteria that live near deep-sea vents are a group of thermophiles that we discussed in Chapter 1. Researchers have discovered that the proteins of thermophilic bacteria have higher proportions of particular amino acids that form stronger bonds than the proteins of other, heat-intolerant species. These strong forces of attraction within and between molecules prevent them from being shaken apart under high temperatures, so the proteins do not denature.

50
Temperature also affects other biological compounds. For instance, the physical properties of fats and oils, which are major components of cell membranes and constitute the energy reserves of animals, depend on temperature. When fats are cold, they become stiff; when they are warm, they become fluid.
Thermal pollution Discharging water that is too hot to sustain aquatic species.
Organisms are negatively affected when exposed to temperatures that range above or below those to which they are adapted. Water used to keep nuclear power plants cool provides an example of this problem. Many nuclear power plants extract water from nearby lakes, rivers, or oceans for cooling the power plants and then return the water—which has become much warmer in the process—to its source. But the organisms in these bodies of water have no evolutionary history of being exposed to such high water temperatures and many of them die. In Ohio, for example, a coal-burning power plant raised the temperature of an adjacent stream up to 42°C, which is much warmer than would naturally occur, and warmer than the maximum of 32°C set by the U.S. EPA. Surveys found numerous dead fish in the stream and low numbers of fish downstream where the stream emptied into the Ohio River. Discharging water that is too hot to sustain aquatic species is known as thermal pollution. Understanding the thermal optima for aquatic species has led to regulations that restrict how much a discharging power plant can raise the temperature of a lake or river.
Cold Temperatures and Freezing
Temperatures on Earth’s surface rarely exceed 50°C, except in hot springs and at the soil surface in hot deserts. However, temperatures below the freezing point of water are common, particularly on land and in small ponds, which may freeze solid during winter. When living cells freeze, the crystal structure of ice disrupts most life processes and may damage delicate cell structures, eventually causing death. Many organisms successfully cope with freezing temperatures either by maintaining their body temperatures above the freezing point of water, or by activating chemical pathways that enable them to resist freezing or to tolerate its effects.
It might surprise you to learn that marine vertebrates are susceptible to freezing in cold seawater. How can blood and body tissues freeze solid while immersed in liquid? The answer is that dissolved substances lower the temperature at which water freezes. While pure water freezes at 0°C, seawater, which contains about 3.5 percent dissolved salts, freezes at −1.9°C. Because the blood and body tissues of most vertebrates contain about half the salt content of seawater, the animals can freeze solid before the surrounding seawater does.
Glycerol A chemical that prevents the hydrogen bonds of water from coming together to form ice unless the temperatures are well below freezing.
Marine animals have evolved a number of adaptations to combat the problem of freezing in water. We know that high salt concentrations interfere with many biochemical processes, so raising the concentration of solutes in blood and tissues is not a viable option. Instead, some Antarctic fish prevent freezing by raising their blood and tissue concentrations of nonsalt compounds such as glycerol. Glycerol is a chemical that prevents the hydrogen bonds of water from coming together to form ice unless the temperatures are well below freezing. A 10 percent glycerol solution in the body lowers the freezing point of water to about −2.3°C without severely disrupting biochemical processes. This is enough to lower the freezing point of the body below the freezing point of seawater. Glycoproteins are another group of compounds that can be used to lower the freezing temperature of water. Glycerol and glycoproteins act as antifreeze compounds, similar to the antifreeze used in automobiles, and allow fish such as the Arctic cod (Boreogadus saida) to remain active in seawater that is cold enough to cause most fish to freeze solid (Figure 2.20). Some terrestrial invertebrates also use the antifreeze approach; their body fluids may contain up to 30 percent glycerol as winter approaches.
Glycoproteins A group of compounds that can be used to lower the freezing temperature of water.

51
Supercooling A process in which glycoproteins in the blood impede ice formation by coating any ice crystals that begin to form.
Supercooling provides a second physical solution to the problem of freezing. Under certain circumstances, liquids can cool below the freezing point without developing ice crystals. Ice generally forms around an object, called a seed, which can be a small ice crystal or other particle. In supercooling, however, glycoproteins in the blood impede ice formation by coating any ice crystals that begin to form. In the absence of ice seeds, pure water may cool to more than –20°C without freezing. Such supercooling has been recorded down to –8°C in reptiles and –18°C in invertebrates.
Thermal Optima
Optimum The narrow range of environmental conditions to which an organism is best suited.
Every organism is best suited to a narrow range of environmental conditions; this range defines the optimum environmental conditions. In terms of temperature, most organisms have a thermal optimum, meaning the range of temperatures in which they perform best. The thermal optimum is determined by the properties of enzymes and lipids, the structures of cells and tissues, body form, and other characteristics that influence the ability of an organism to function well under the particular conditions of its environment. Returning to the example of fish in the ocean waters of the Antarctic, many species swim actively and consume oxygen at a rate comparable to fish living in much warmer regions near the equator. If you put a tropical fish in cold water, it becomes sluggish and soon dies; conversely, Antarctic fish cannot tolerate temperatures warmer than 5°C to 10°C.
Thermal optimum The range of temperatures within which organisms perform best.
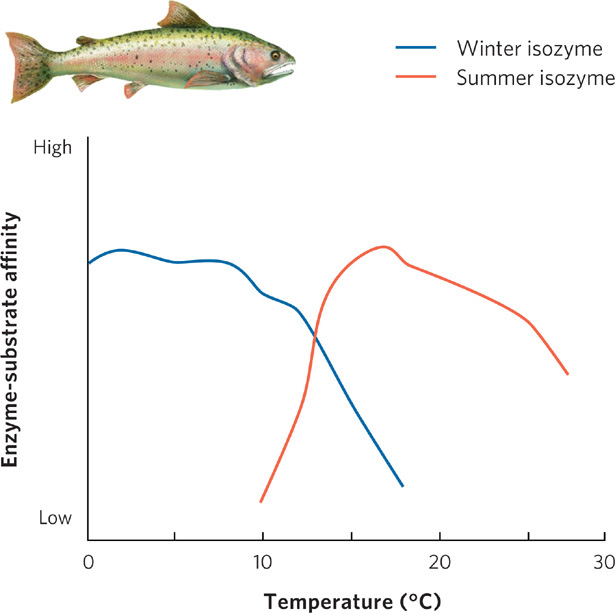
Isozymes Different forms of an enzyme that catalyze a given reaction.
Certain adaptations allow fish in cold oceans to swim as actively as fish in warm oceans. Swimming involves a series of biochemical reactions, most of which depend on enzymes. These reactions generally proceed more rapidly at high temperatures, so cold-adapted organisms must either have more of the substrate for a biochemical reaction, more of the enzyme that catalyzes the reaction, or a different version of the enzyme that operates better under colder temperatures. Different forms of an enzyme that catalyze a given reaction are called isozymes.
Consider the case of the rainbow trout (Oncorhynchus mykiss), a fish that lives in cold streams throughout much of North America. Seasonal changes in temperature are predictable for the trout: during winter, water temperatures may drop close to the freezing point, while summer temperatures can become very warm. In response to these seasonal temperature changes, the trout has evolved the ability to produce different isozymes in winter and in summer. One of these enzymes is acetylcholinesterase, which plays an important role in ensuring proper functioning of the nervous system by binding with the neurotransmitter acetylcholine. To understand how well different isozymes function at different temperatures, we can examine the rate of the chemical reaction between acetylcholine and acetylcholinesterase, a measure known as enzyme-substrate affinity. The winter isozyme, shown as the blue line in Figure 2.21, best catalyzes the reaction at 0°C to 10°C. This affinity drops rapidly at higher temperatures. In contrast, the summer isozyme, shown as the orange line, has a weak affinity for acetylcholine at 10°C and catalyzes reactions the best at 10°C to 20°C, though the affinity drops slowly at higher temperatures. As you might predict, the particular isozyme that a trout produces depends on the temperature of the water it lives in. When trout live at 2°C, they produce the winter isozyme, whereas when they live at 17°C, they produce the summer isozyme.
52
ECOLOGY TODAY CONNECTING THE CONCEPTS
THE DECLINE OF CORAL REEFS

Coral reefs are some of the most beautiful places on Earth and are home to an incredible diversity of species. Coral reefs are also one of the most diverse places on Earth, rivaling even the highly diverse tropical rainforests. For instance, the Great Barrier Reef off the eastern coast of Australia contains more than 400 species of coral, 200 species of birds, and 1,500 species of fish. For these reasons, many people become concerned when the biodiversity of coral reefs is threatened by human activity. We have long understood that overfishing and pollution have affected the species that inhabit coral reefs. In the past 15 years, however, scientists have discovered that changes in the abiotic aquatic environment—including changes in temperature, pH, and salinity—are damaging coral reef ecosystems.
53
Corals are a group of tiny animals that secrete hard exoskeletons made of limestone (calcium carbonate). They can be found worldwide in relatively shallow ocean waters that are low in nutrients and food. Although each individual coral is small—only a few millimeters in size—the limestone bodies of dead corals accumulate over hundreds or thousands of years to form massive coral reefs that can exceed 300,000 km2. The corals survive in these low nutrient waters by living in symbiotic relationships with several species of photosynthetic algae known as zooxanthallae. Corals have tubular bodies with tentacles that stick out and catch bits of food and detritus that go by. Their digestion produces CO2, which can be used by symbiotic algae during photosynthesis. As we learned earlier in this chapter, CO2 can often be difficult for aquatic producers to obtain. In exchange, the algae produce O2 and sugars, some of which can be passed to the coral. In short, the algae get a safe place to live and a steady supply of CO2 for photosynthesis, while the corals get a source of energy in the form of sugars and a steady supply of O2 for respiration.
Coral bleaching Loss of color in corals as a result of the corals expelling their symbiotic algae.
During the past two decades, scientists have learned that the symbiotic relationship between the corals and algae is very sensitive to environmental changes. When corals are experiencing stress in their environment, they expel the symbiotic algae from their bodies. Because corals obtain their bright colors from the symbiotic algae, corals that expel their algae often look white and are said to experience coral bleaching.
Coral bleaching is associated with unusually high ocean temperatures. As we discussed in this chapter, while increases in water temperature can increase the rate of chemical reactions, temperatures that exceed the thermal optima can be detrimental. Bleaching can begin if summer ocean temperatures are even just 1°C higher than the average maximum. If the temperature rise is brief—a few days or weeks—the algae can recolonize the corals. However, the corals will experience slower growth and reduced reproduction. However, if the temperatures are 2°C to 3°C higher than the average maximum, corals can die. During the past two decades, scientists have witnessed major bleaching events worldwide in 1998, 2003, 2005, and 2010. During the 1998 event, 42 percent of corals in the Great Barrier Reef experienced bleaching. With continuing increases in global temperatures, a topic we will cover in detail in later chapters, temperature-induced coral bleaching is expected to continue.
Changes in salt concentration are also an issue for corals. High ocean temperatures increase the evaporation of water from the ocean, which increases the ocean’s salt concentration. In the case of corals, the stress of increased salt concentrations combined with the stress of high temperatures makes them increasingly vulnerable to coral bleaching and coral death. The rise in ocean temperatures also appears to stress corals in a way that makes them more susceptible to pathogens that can kill them.
Another source of decline in corals is a decrease in the pH of seawater. Because atmospheric CO2 is in equilibrium with dissolved CO2 in the ocean, the recent increase in atmospheric CO2 has caused an increase in dissolved CO2. This, in turn, has caused an increase in carbonic acid, and a decline in the pH of the oceans. As we have seen, carbonic acid (H2CO3) disassociates to bicarbonate ions (HCO3−) and hydrogen ions (H+). The hydrogen ions can then combine with other carbonate ions to make bicarbonate, making carbonate ions less available for the corals to produce their exoskeleton of calcium carbonate.
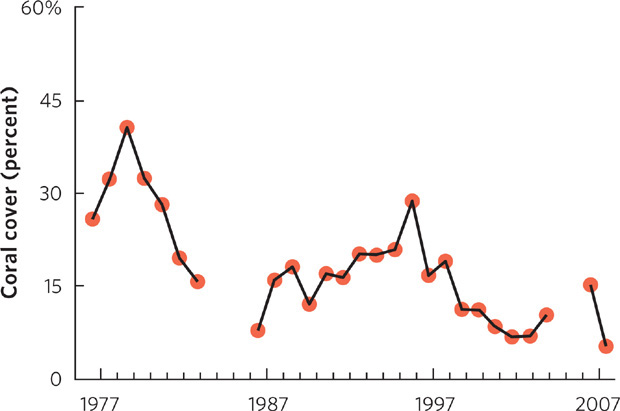
The decline in coral reefs has become a serious problem. In the Caribbean, for example, the percent of live corals has declined from more than 40 percent in 1977 to less than 10 percent in 2007. Only 20 years ago, scientists debated whether abiotic changes were playing a role in the decline of corals. As more data have been collected, there is now general agreement. Given that the changes in temperature, pH, and salinity are expected to rise even more in future decades, scientists currently predict that many species of coral will continue to decline. However, some species of coral may have sufficient genetic variation to allow evolutionary changes that permit them to adapt to the changing environmental conditions and persist.
SOURCES: Carpenter, K. E., et al. 2008. One-third of reef-building corals face elevated extinction risk from climate change and local impacts. Science 321: 560-563.
Hoegh-Guldberg, O., et al. 2007. Coral reefs under rapid climate change and ocean acidification. Science 318: 1737-1742.
54