ENGINEERING ORGANS
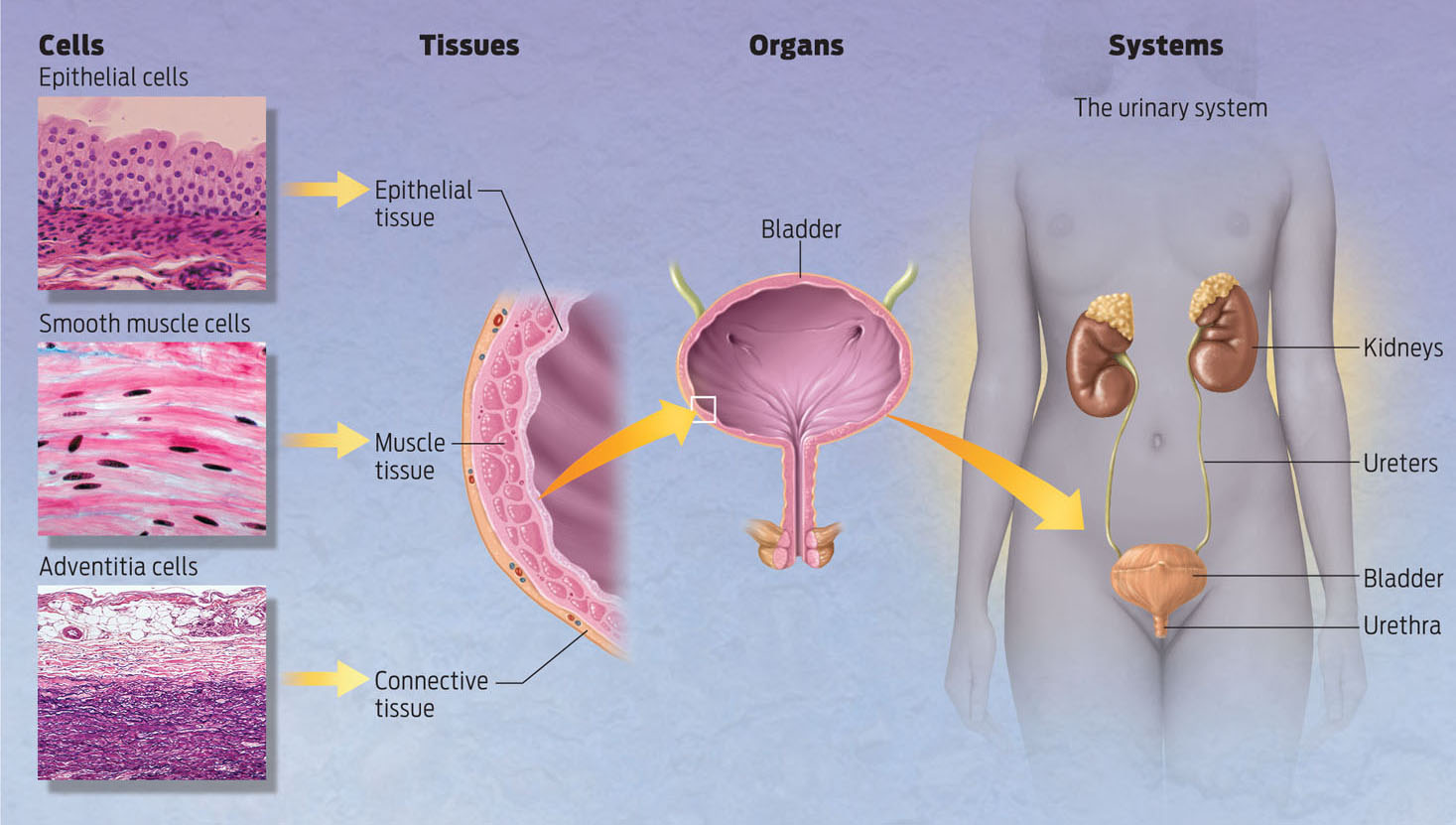
TISSUE An organized group of different cell types that work together to carry out a particular function.
The effort to engineer human organs for transplants dates back more than 40 years and is based on the knowledge that organs made from a patient’s own cells would have advantages over ones obtained from human or nonhuman donors. Not only would they be genetically compatible, they also wouldn’t rely on a supply of donors. Important strides in the field were made in the late 1980s, when Joseph Vacanti of Boston Children’s Hospital teamed up with Robert Langer at MIT to engineer tissues. The pair wanted to design synthetic biodegradable scaffolds that could be molded into particular shapes–a human ear, for example–and then coated with cells that would grow into a tissue. The scaffold itself would never need to be removed–it would in time dissolve.
Atala, who had collaborated with Vacanti and Langer, applied this research on biodegradable scaffolds to his own work on engineered bladders. Atala, a surgeon, sought to help his patients whose bladders were not functioning normally because of cancer, injury, or a birth defect. For more than a century, doctors have treated such patients by using pieces of their intestine or bowel to reconstruct their bladders. But because intestine and bowel are built to do different things than the bladder, this treatment is not ideal–it can lead to many problems later, including leaks and even cancer. Growing a piece of new bladder to repair the organ, Atala reasoned, would be much better.
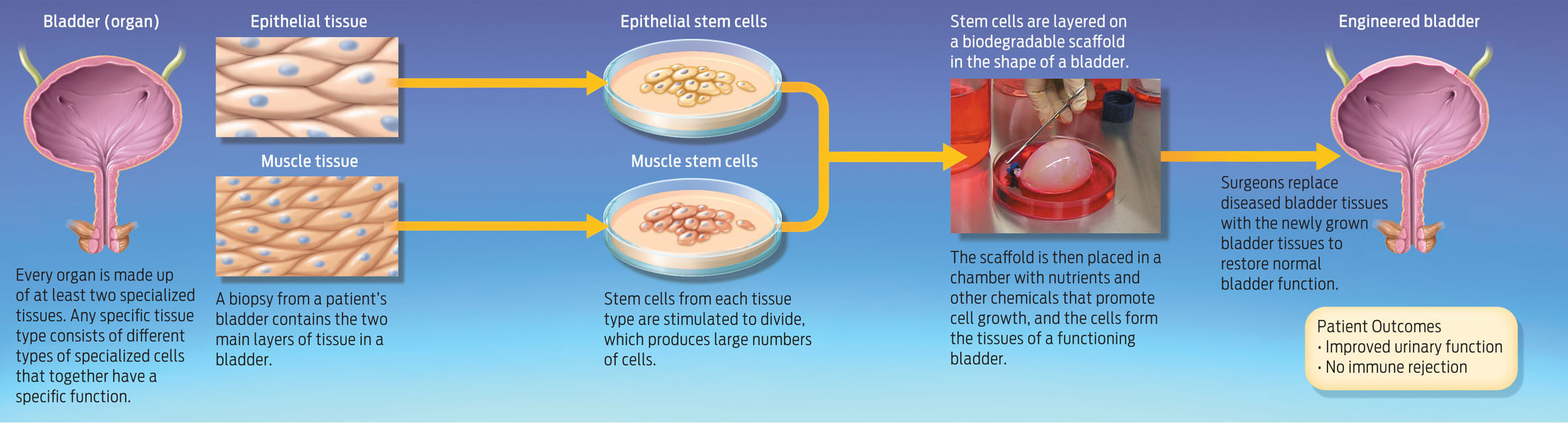
Although it has been possible for decades to grow human skin outside the body to treat burn victims, growing more-complex organs like bladders has been challenging. Skin is a thin, flat organ made up of mostly one cell type. A bladder, by contrast, is a round, hollow organ made up of multiple cell types working together. To grow a bladder, scientists must grow several layers of tissue, including muscle and epithelial tissue. (Epithelial cells are flat cells that line many organs.) Moreover, the thicker the tissue, the more blood vessels required to nourish it (INFOGRAPHIC 13.2).
Tissues are integrated groups of specialized cells working together. Multiple tissues combine to form organs, which in turn cooperate as part of a single functioning organ system.
It took Atala 17 years of research to achieve success. He spent the most of those years figuring out how to get bladder cells to divide outside the body, and then how to grow them on a biodegradable scaffold in the shape of a bladder. Next, he had to test the bladders in laboratory animals to make sure they worked. Finally, in 1999, he was ready to test the bladders in patients.
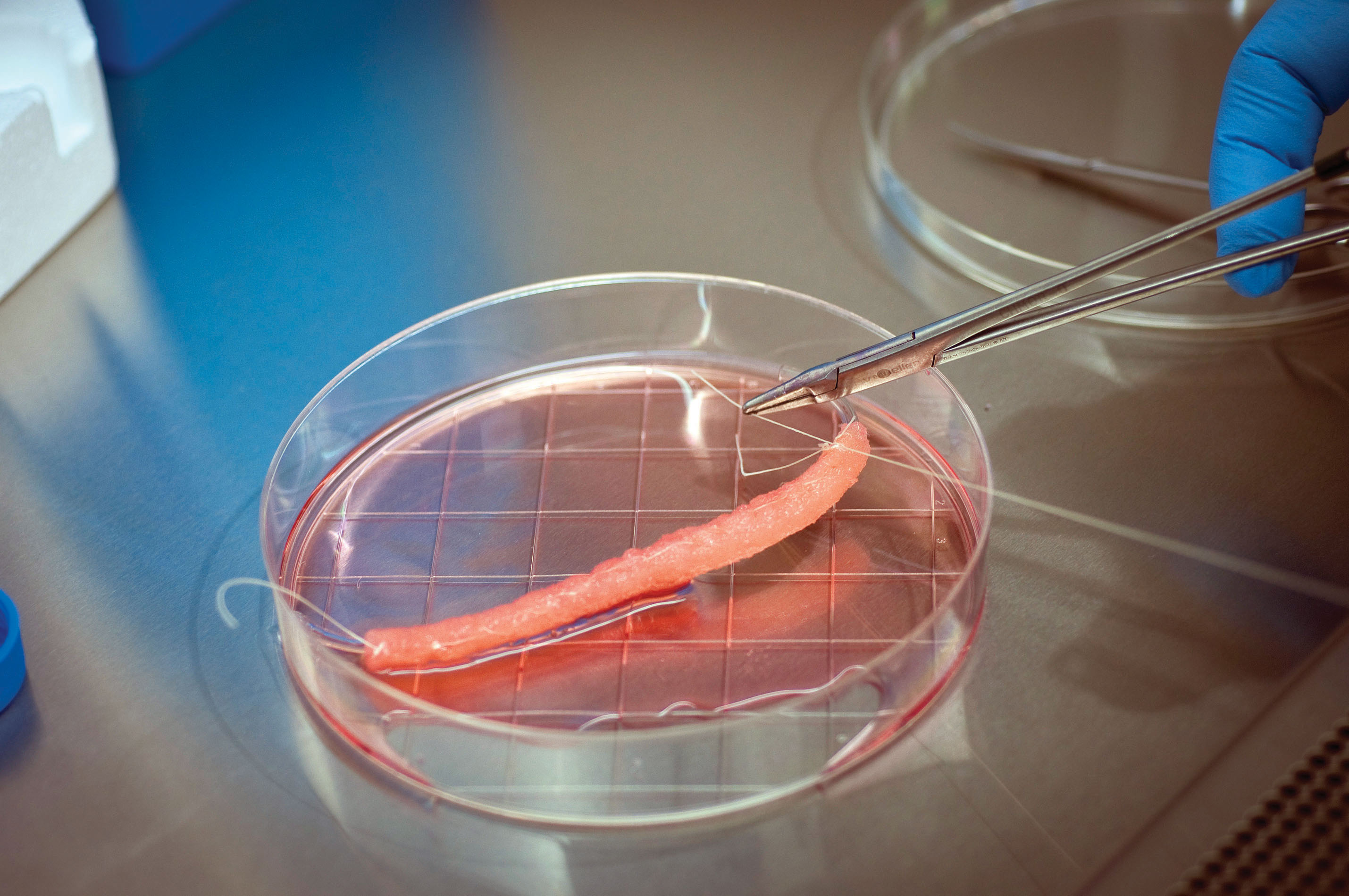
For each patient, Atala cut a piece of tissue smaller than a postage stamp from inside the bladder and extracted two types of cells–muscle stem cells and bladder epithelial stem cells. He then mixed these stem cells with chemicals that promoted cell division, producing a stockpile of millions of cells. Next, he layered these stem cells onto the biodegradable scaffold, which he had sculpted to resemble a human bladder. He bathed the scaffold in nutrients to stimulate the cells to divide, and then placed the scaffold with nutrients and other growth factors in an incubator to simulate conditions inside the human body. The cells went through several cell divisions, attached to the scaffold, and grew into the tissue layers that make up a bladder. Two months later, surgeons reconstructed the patient’s bladders using the new bladder tissue (INFOGRAPHIC 13.3).

The treatment, so far, seems remarkably successful. Not only does the technique improve bladder function, it avoids the complications that result from using tissues from other organs–like the bowel–for bladder repair. “Doing bowel-for-bladder replacements in children really got to me. It’s one thing to put them into an adult, but putting them in a child with a 70-plus life expectancy didn’t make sense when you knew there would be trouble down the line,” Atala told the New York Times in 2006, soon after he had published his results.
Atala’s technique also avoids the immune rejection that can result from using bladder tissue transplanted from a donor. Just as a food allergy can cause shock when the offending food is ingested, a person’s immune system can react similarly to a donated organ and reject it. By contrast, tissue grown from a person’s own cells poses no such risk of rejection because the tissue is genetically identical to the source.
As of 2012, a total of about 30 patients have received the experimental bladder treatment, which is being evaluated in an ongoing clinical trial. Thanks to the engineered bladder he received in 2001, Luke Masella is now a healthy young man with functioning bladder and kidneys. The 21-year-old is currently a senior at the University of Connecticut, studying media, television, and communications.
Atala is using his organ-growing technique to make other organs for transplant as well. He has used it to grow replacement urethras for children whose urethras were damaged by trauma to their pelvis, for example. The urethras are working normally in all five boys who received them, Atala reported in The Lancet in 2011. And these organs are just the beginning. Atala’s group at Wake Forest is working on developing a whole host of organs for transplant, including heart, kidney, and liver.