Chapter 1. Pigment Lab 1
Introduction
REEL Pigment Lab 1
Part C
Experimentally, the color of solutions can be studied by UV-Visible (UV-Vis) absorbance spectroscopy. In this measurement, a light source generates radiation with wavelengths ranging from 300–900 nm, which includes UV (<400 nm), visible (400-750nm) and IR (>750nm) light. In an absorbance measurement, the light is passed through a sample to reach a detector that is sensitive to the wavelength of the light, and indicates which wavelengths of light have been absorbed.
If a sample is colored, it will absorb a portion of the visible spectrum. We can then use the color wheel to understand the color as perceived by the human eye. In the simplest cases, only one color is absorbed and the color that results is the complementary color. For example, consider the solution depicted in Figure C-1. All of the wavelengths of light generated pass through the sample to the detector, with the exception of red light, which is absorbed. As a result, our eyes see the complementary color, green.
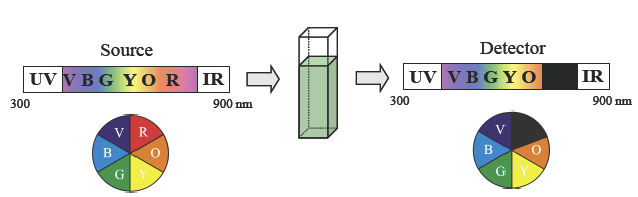
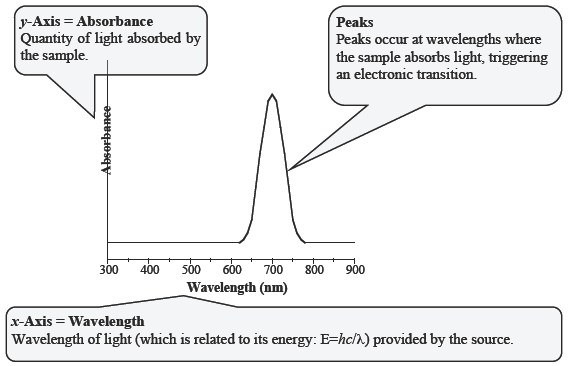
The output of a UV–Vis spectrometer is called a spectrum. The degree of absorbance is plotted on the y-axis and the wavelength of light is plotted on the x-axis. The UV–Vis spectrum of the green solution from Figure C-1 is shown below in Figure C-2. Most spectra are not this simple, but by considering which wavelengths of light are absorbed and which are transmitted, it is possible to predict the color of a substance.
When scientists started to synthesize and isolate transition metal coordination compounds, they noticed a large variety of colors depending on the metal-ligand combination used. After Hans Bethe and John Hasbrouck van Vleck introduced their crystal field theory, chemists began to relate the colors of transition metal complexes with the crystal field splitting energy.
From the previous exercise, we learned that when ligands surround a transition metal in a given geometry, some of the orbitals on the metal interact differently with the ligands than others. As we learned in Part B, this causes a splitting of the d-orbitals into two sets – one at higher energy, and one at lower energy. This energy splitting is known as the crystal field splitting energy (which you investigated in Part B) and the energy difference between the two levels often falls in the visible range of the spectrum.
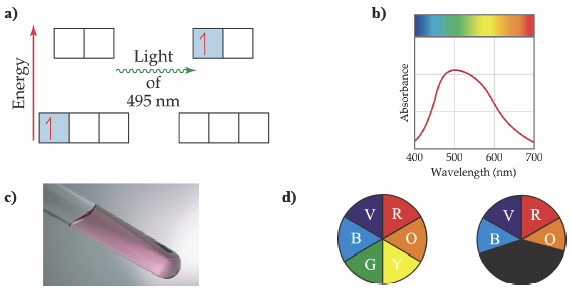
When a transition metal has partially filled d-orbitals, absorption of light can trigger an electronic transition from one d-orbital to another d-orbital higher in energy. This is called a d-to-d transition. To illustrate this point, let us consider the red-violet solution of the transition metal complex, [Ti(H2O)6]3+ (Figure C-3). The d-orbitals are split into two groups by the coordinating water molecules, and the energy difference between the two sets of orbitals is 4.02 x 10−19 J. A photon whose energy is 4.02 x 10−19 J is able to excite the electron from a lower energy d-orbital to one of the higher energy d-orbitals. Such a photon has a wavelength of 495 nm which falls in the green region of the spectrum. The UV-Vis spectrum shows a broad absorption with a maximum at 495 nm. Because the compound absorbs green light (and to a lesser extent yellow), it appears redviolet to our eyes (red and violet are the complementary colors of green, and yellow respectively) (Figure C-3).
By studying groups of compounds, and looking for trends, chemists discovered some key factors that influenced the crystal field splitting energy.
The metal oxidation state and position in the periodic table affect the crystal field splitting energy. Chemists noticed that the greater the oxidation state, the greater the crystal field splitting energy. In addition, the larger 4d and 5d transition metals exhibit greater crystal field splitting energy than their smaller 3d counterparts. Both of these trends can be explained by a simple principle: the greater the interaction or overlap between d-orbitals and ligands, the greater the crystal field splitting energy, Δ. The ligand also plays a role in determining the size of the crystal field splitting energy. The spectrochemical series arranges ligands from those that give small Δ (weak field ligands) to those that give large Δ (strong field ligands). We will explore the spectrochemical series in greater detail in the next lab.
Another important factor that influences the crystal field splitting energy is the coordination geometry of the central metal. The two most common geometries are octahedral (six ligands) and tetrahedral (four ligands). You developed crystal field theory for both tetrahedral and octahedral geometries in Part B, where you learned that the crystal field splitting energy is larger for octahedral complexes than tetrahedral complexes. In fact, for the same metal and ligand set, the crystal field splitting energy for a tetrahedral complex is approximately 4/9 the size of the equivalent octahedral complex.
In this part of the lab, you will be investigating the chemistry of two ions: [Co(H2O)6]2+ and [CoCl4]2− to probe the effect of the coordination geometry on the color of transition metal coordination compounds. You will be using UV-Vis spectroscopy as an experimental probe of color and crystal field splitting energy.
Click the Next button to start the activity
1.1 Activity Completed!
Activity results are being submitted...