
12.2 Membrane Fluidity Is Controlled by Fatty Acid Composition and Cholesterol Content
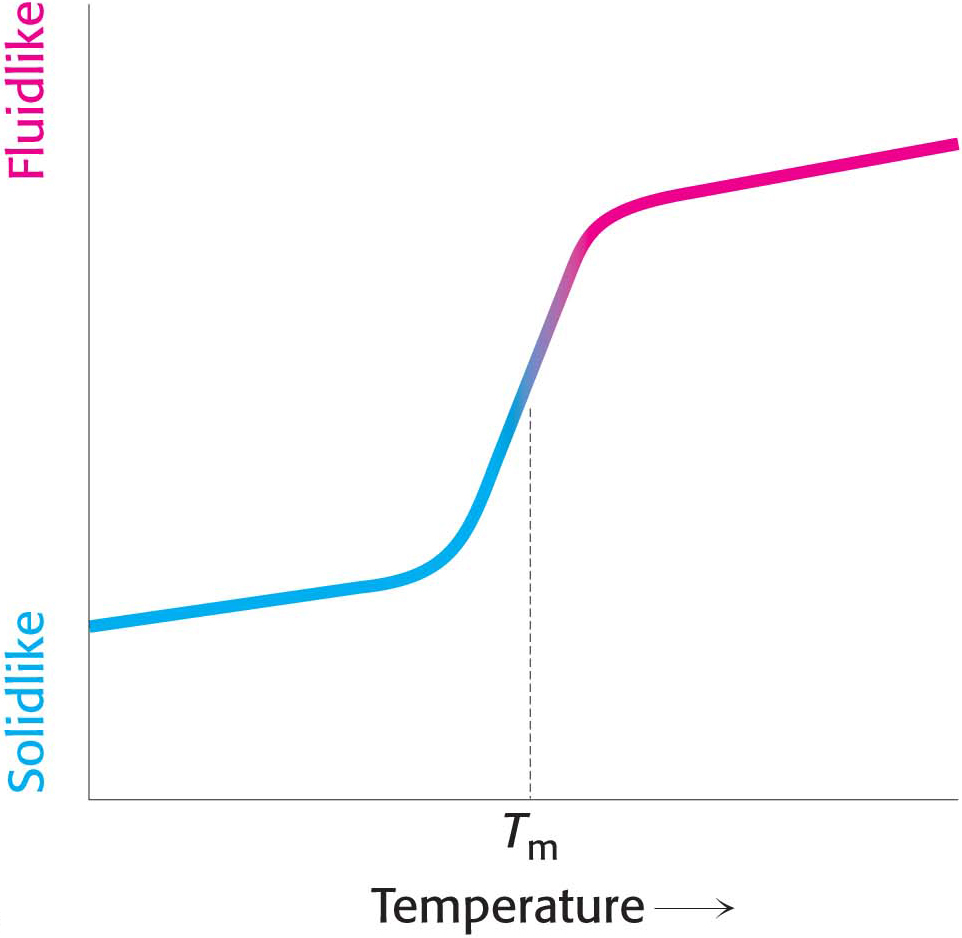
Many membrane processes, such as transport or signal transduction, depend on the fluidity of the membrane lipids, which is determined by the properties of fatty acid chains. Recall that the melting point of individual fatty acids depends on their length and the number of cis double bonds. These same chemical properties affect the fluidity of the membranes of which the fatty acids are a component. The fatty acid chains in membrane bilayers may be arranged in an ordered, rigid state or in a relatively disordered, fluid state. The transition from the rigid to the fluid state takes place rather abruptly as the temperature is raised above Tm, the melting temperature (Figure 12.5). This transition temperature depends on the length of the fatty acid chains and on their degree of unsaturation. Long saturated fatty acids interact more strongly because of the increased number of van der Waals interactions than do short ones and thus favor the rigid state (Figure 12.6). On the other hand, a cis double bond produces a bend in the hydrocarbon chain. This bend interferes with a highly ordered packing of fatty acid chains, and so Tm is lowered.
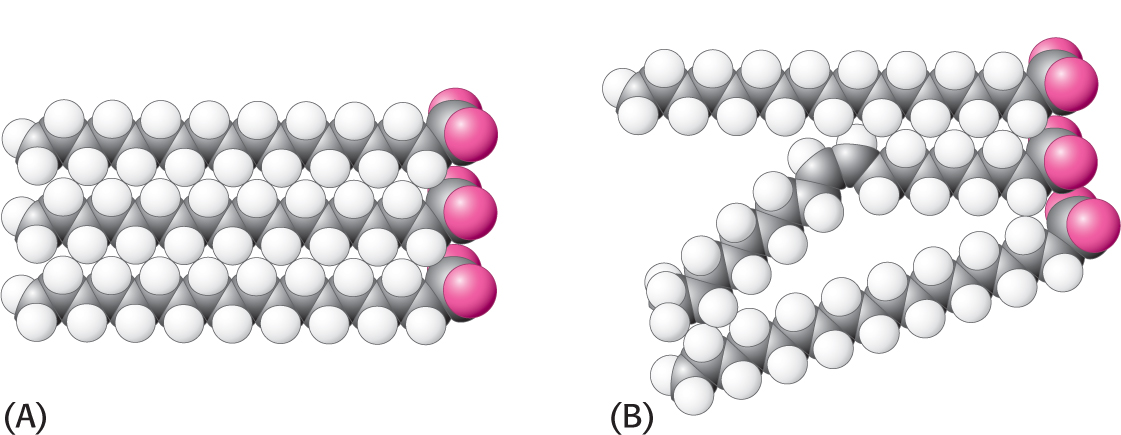
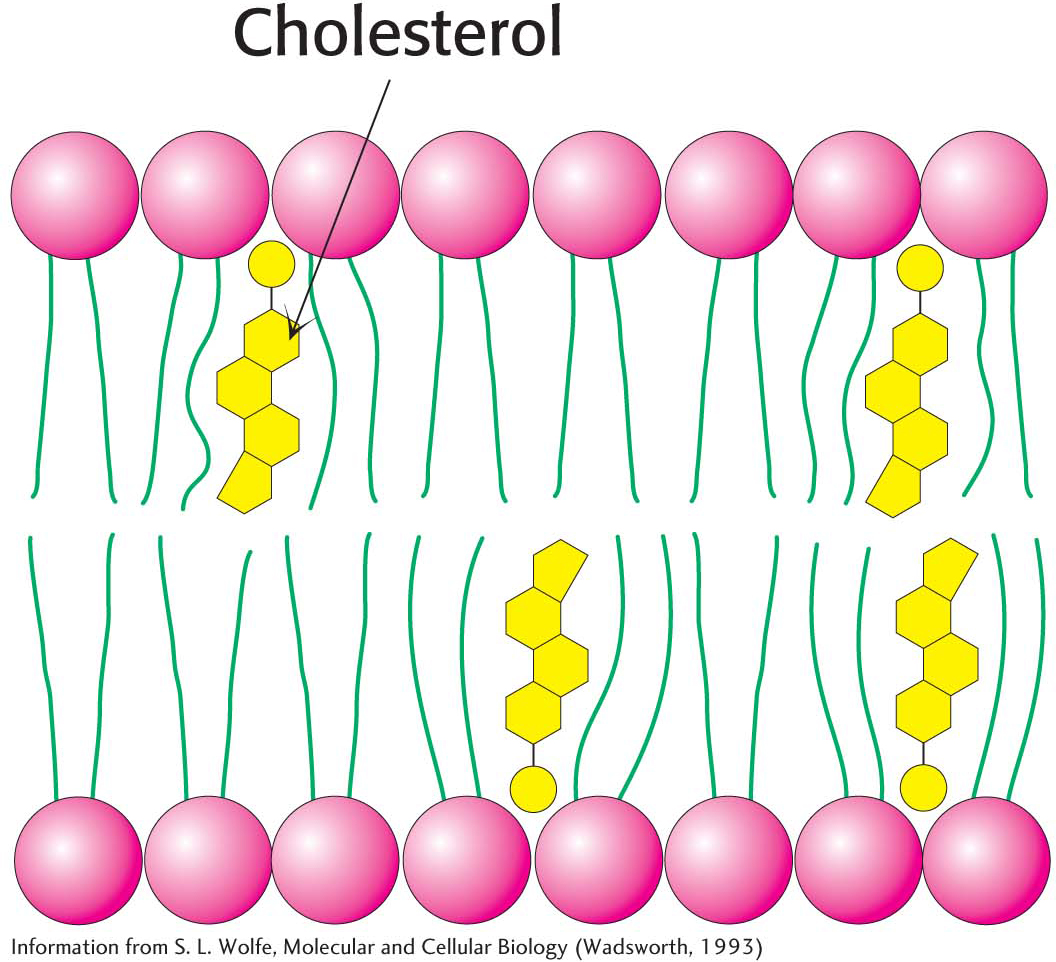
Bacteria regulate the fluidity of their membranes by varying the number of double bonds and the length of their fatty acid chains. In animals, cholesterol is the key modulator of membrane fluidity. Cholesterol contains a bulky steroid nucleus with a hydroxyl group at one end and a flexible hydrocarbon tail at the other end. The molecule inserts into bilayers with its long axis perpendicular to the plane of the membrane. Cholesterol’s hydroxyl group forms a hydrogen bond with a carbonyl oxygen atom of a phospholipid head group, whereas its hydrocarbon tail is located in the nonpolar core of the bilayer. The different shape of cholesterol compared with that of phospholipids disrupts the regular interactions between fatty acid chains and helps maintain membrane fluidity (Figure 12.7).
QUICK QUIZ 1
Predict the effect on membrane-
The increase in temperature will increase the fluidity of the membrane. To prevent the membrane from becoming too fluid, the bacteria will incorporate longer-
In addition to its nonspecific effects on membrane fluidity, cholesterol can form specific complexes with saturated fatty acid components of lipids and specific proteins. These complexes concentrate within small (10–