
29.5 Cholesterol Is the Precursor of Steroid Hormones
Although cholesterol is well known in its own right as a contributor to the development of heart disease, metabolites of cholesterol—
 CLINICAL INSIGHT
CLINICAL INSIGHTBile Salts Facilitate Lipid Absorption
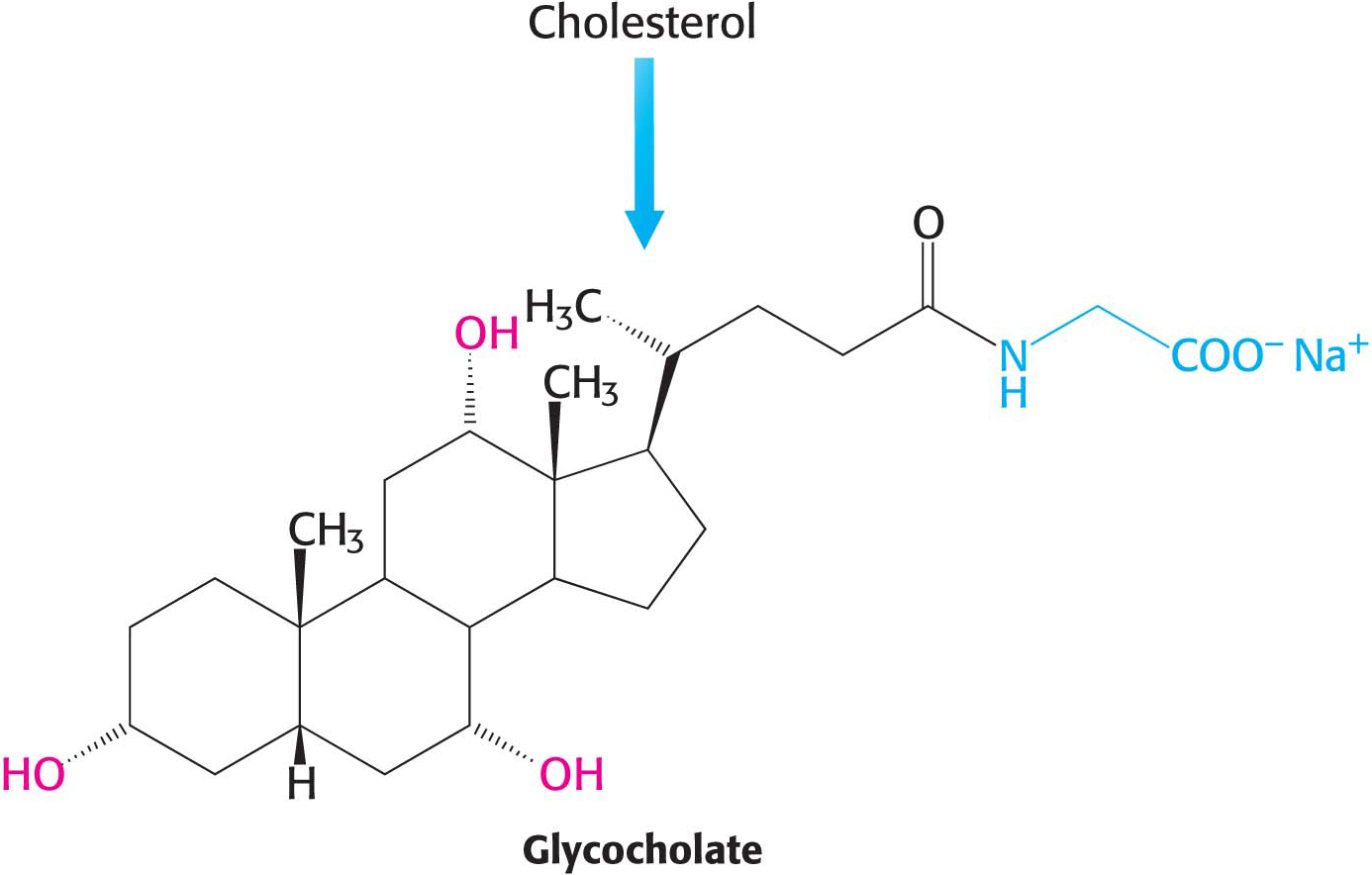
Bile salts, the major constituent of bile, are highly effective detergents because they contain both polar and nonpolar regions. They solubilize dietary lipids so that they are readily digested by lipases and absorbed by the intestine. Bile salts are synthesized in the liver from cholesterol, stored and concentrated in the gall bladder, and then released into the small intestine. Glycocholate is the major bile salt (Figure 29.19).
In addition to bile salts, bile is composed of cholesterol, phospholipids, and the breakdown products of heme. If too much cholesterol is present in the bile, it will precipitate to form gallbladder stones. These stones may block bile secretion and inflame the gall bladder, a condition called cholelithiasis. Symptoms include pain in the upper right abdomen and nausea. If need be, the gall bladder is removed, and bile flows from the liver through the bile duct directly into the intestine.
Steroid Hormones Are Crucial Signal Molecules
Cholesterol is the precursor of steroid hormones (Figure 29.20). These hormones are powerful signal molecules that regulate a host of organismal functions. There are five major classes of steroid hormones: progestagens, glucocorticoids, mineralocorticoids, androgens, and estrogens.
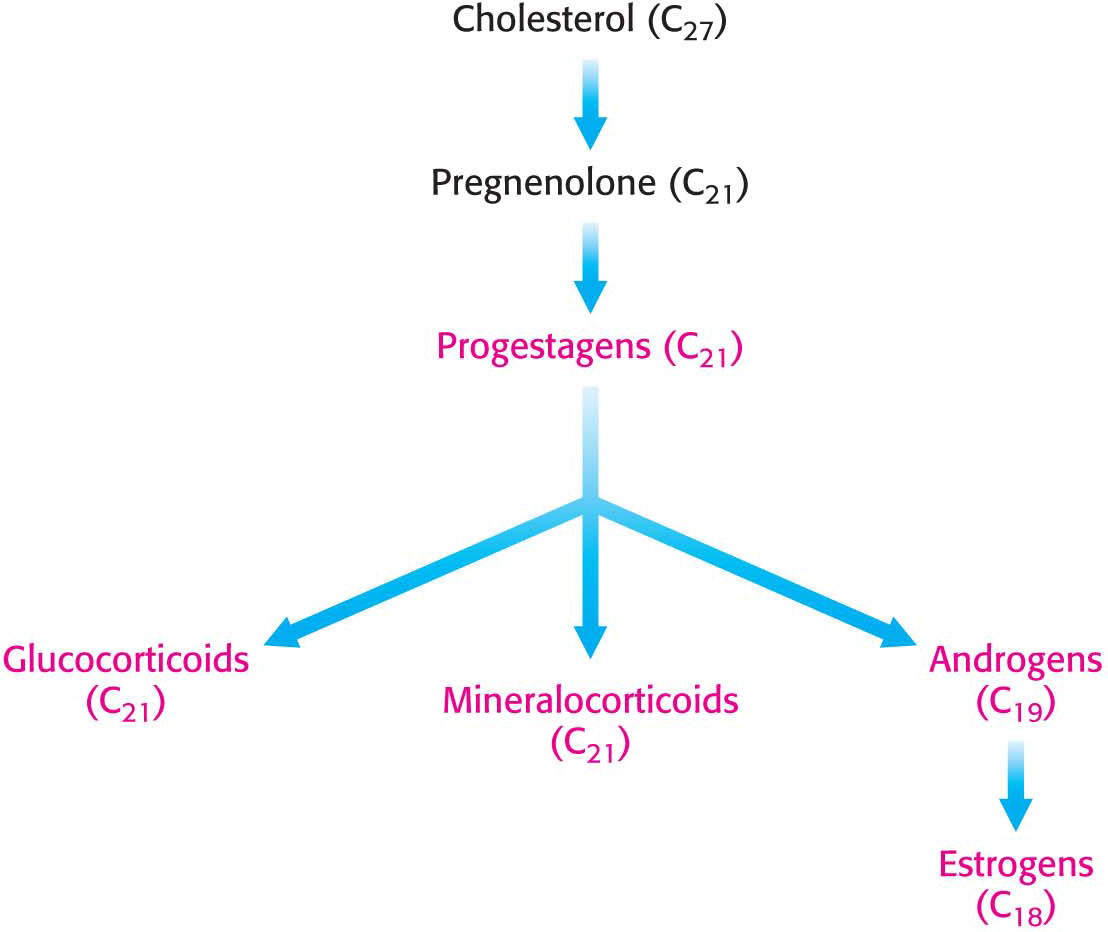
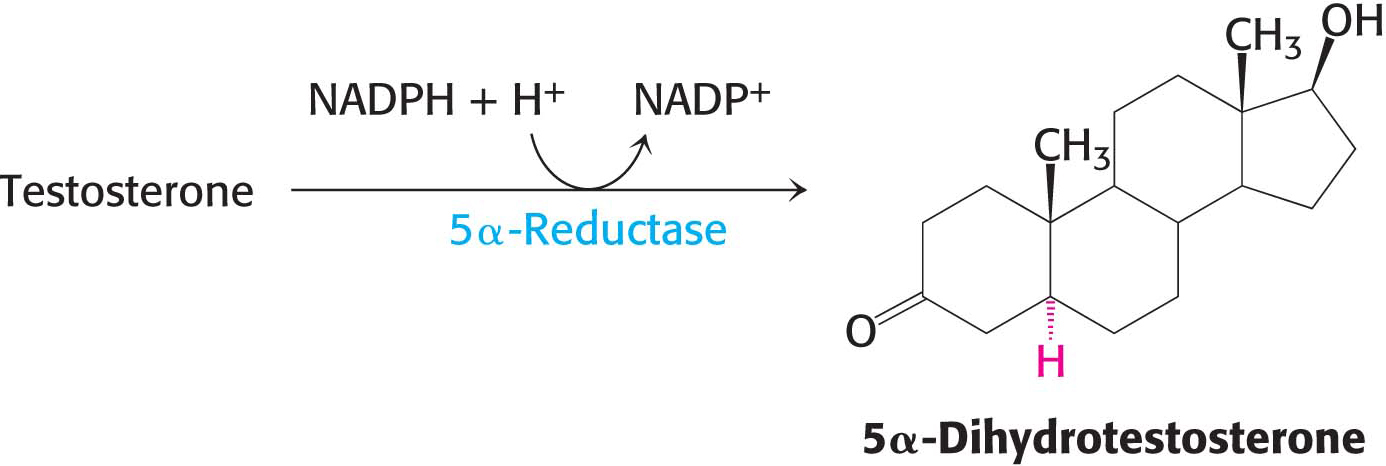
Progesterone, a progestagen, prepares the lining of the uterus for the implantation of an ovum and is essential for the maintenance of pregnancy. Androgens are responsible for the development of male secondary sex characteristics. Testosterone, an important androgen, leads to the development of male sexual behavior. It is also important for the maintenance of the testes and the development of muscle mass. Owing to the latter activity, testosterone is referred to as an anabolic steroid. Testosterone is reduced by 5 α-reductase to yield dihydrotestosterone (DHT), a powerful embryonic androgen that instigates the development and differentiation of the male phenotype:
Estrogens such as estradiol are required for the development of female secondary sex characteristics. Estrogens, along with progesterone, also participate in the ovarian cycle. Glucocorticoids (such as cortisol) promote gluconeogenesis and the formation of glycogen, enhance the degradation of fat and protein, and inhibit the inflammatory response. They enable animals to respond to stress—
All steroid hormones function in a similar manner. These powerful signal molecules bind to and activate receptor proteins that serve as transcription factors, proteins that regulate the expression of target genes (Chapter 37).
Vitamin D Is Derived from Cholesterol by the Energy of Sunlight
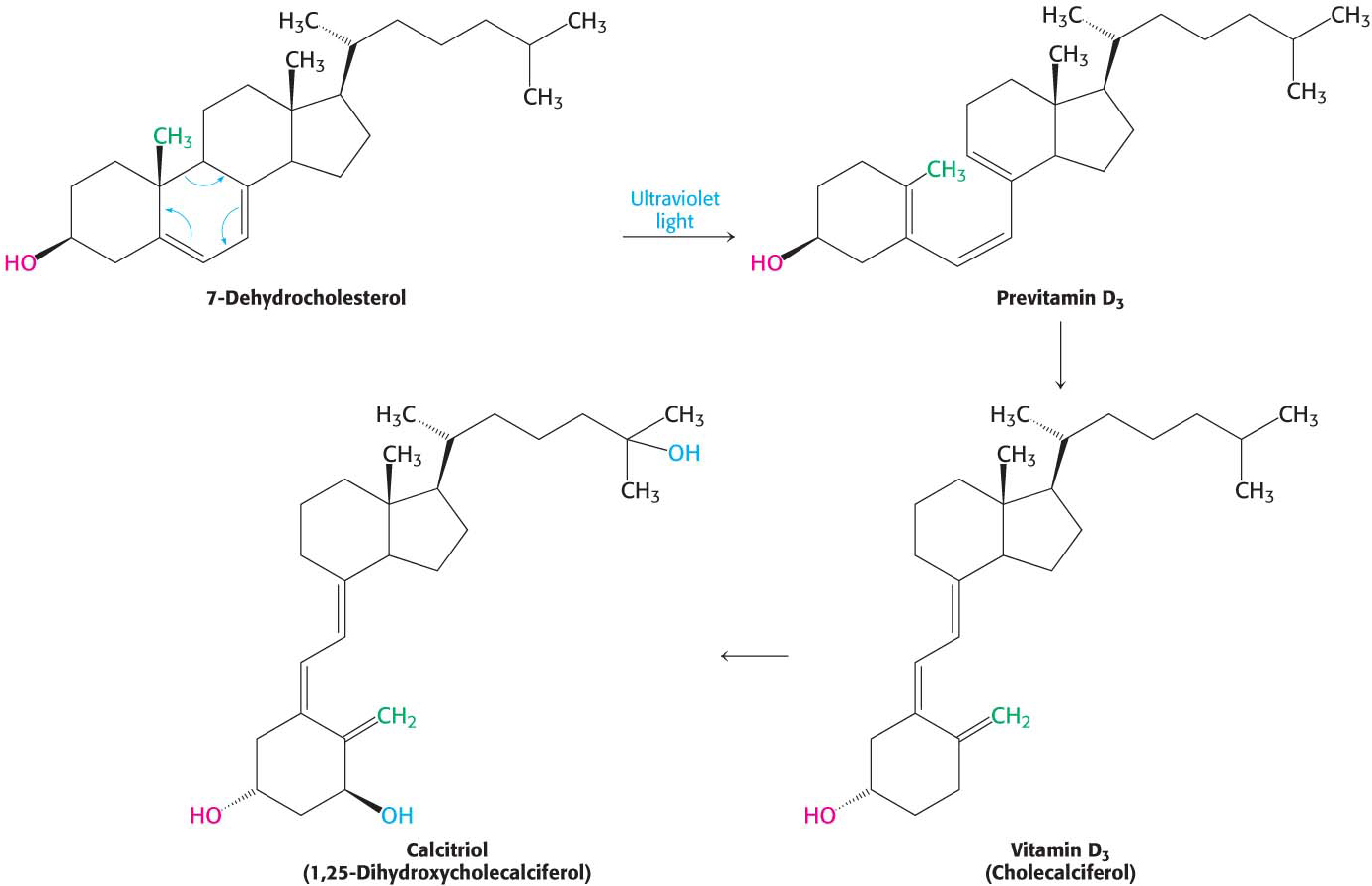
Cholesterol is also the precursor of vitamin D, which plays an essential role in the control of calcium and phosphorus metabolism. The ultraviolet light of sunlight breaks a bond in 7-
 CLINICAL INSIGHT
CLINICAL INSIGHTVitamin D Is Necessary for Bone Development
Vitamin D deficiency in childhood produces rickets, a disease characterized by inadequate calcification of cartilage and bone. Rickets was so common in seventeenth-
Research done in the past few years indicates that vitamin D may play a much larger biochemical role than simply the regulation of bone metabolism. Muscle seems to be a target of vitamin D action. In muscle, vitamin D appears to affect a number of biochemical processes, with the net effect being enhanced muscle performance. Studies also suggest that vitamin D prevents cardiovascular disease, reduces the incidence of a variety of cancers, and protects against autoimmune diseases including diabetes. Moreover, vitamin D deficiency appears to be more common than thought. People living in northern climes may not be exposed to enough sunlight during certain times of the year to synthesize adequate amounts of vitamin D. Since skin pigmentation blocks UV radiation, people with dark complexions living in such areas may be especially susceptible to vitamin D deficiency. For example, in the United States, some studies suggest that 75% of Blacks and many Hispanics and Asians have insufficient blood levels of vitamin D. This recent research on vitamin D shows again the dynamic nature of biochemical investigations. Vitamin D, a chemical whose biochemical role was believed to be well established, now offers new frontiers of biomedical research.

 CLINICAL INSIGHT
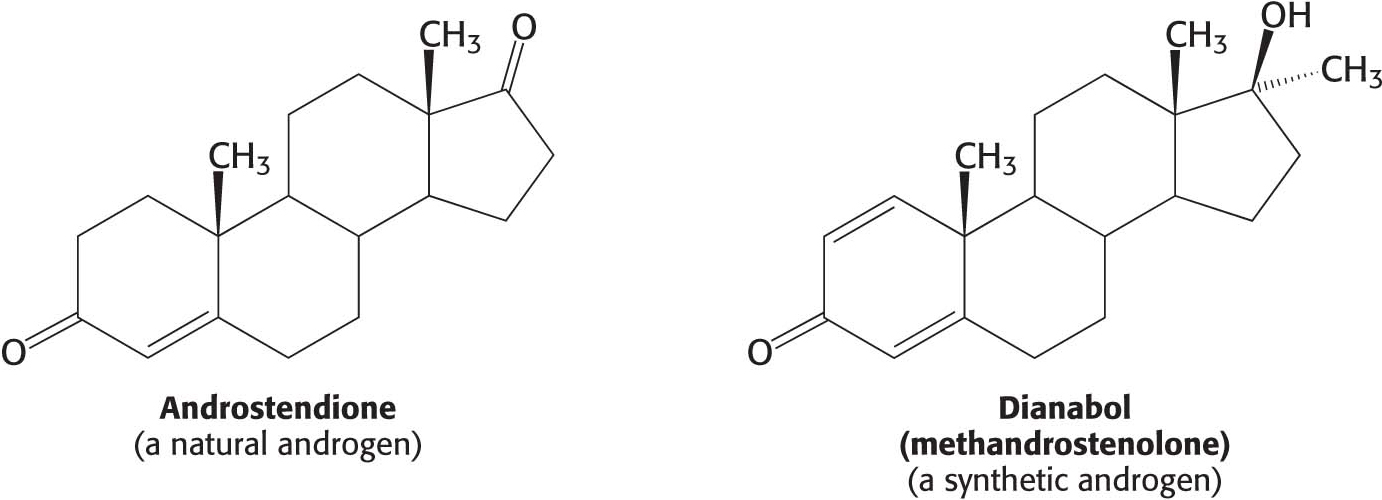
CLINICAL INSIGHTAndrogens Can Be Used to Artificially Enhance Athletic Performance
Some athletes take androgens because the anabolic effects of androgens increase lean muscle mass (Figure 29.22). Synthetic steroids, such as dianabol, have been developed in an attempt to separate the anabolic properties (the growth of lean muscle mass) from the androgenic properties (the development of male sexual characteristics) of endogenous androgens.
However, to clearly separate these two activities has proved to be impossible. The use of anabolic steroids in males leads to a decrease in the secretion of testosterone, testicular atrophy, and, sometimes, breast enlargement (gynecomastia) owing to the conversion of some of the excess androgen into estrogen. In women, excess testosterone causes a decrease in ovulation and estrogen secretion, breast regression, and the growth of facial hair.
Oxygen Atoms Are Added to Steroids by Cytochrome P450 Monooxygenases
One of the chemical features that distinguish the various steroids from one another is the number and location of oxygen atoms. Carbon 3 is the only carbon atom bound to oxygen in cholesterol. All other oxygen atoms in steroid hormones are introduced by hydroxylation reactions that require NADPH and O2. The enzymes catalyzing these reactions are called monooxygenases or mixed-
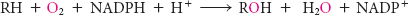
Hydroxylation requires the activation of oxygen. In the synthesis of steroid hormones, activation is accomplished by a cytochrome P450 monooxygenase, a family of membrane-
Metabolism in Context: Ethanol Also Is Processed by the Cytochrome P450 System
In Chapter 28, we considered the consequences of excess ethanol consumption. Many of the deleterious biochemical effects stem from the overproduction of NADH by alcohol dehydrogenase and acetaldehyde dehydrogenase. Ethanol can also be processed by an ethanol-
The adverse effects of ethanol are not limited to the metabolism of ethanol itself. Vitamin A (retinol) is converted into retinoic acid, an important signal molecule for growth and development in vertebrates, by the same dehydrogenases that metabolize ethanol. Consequently, this activation does not take place in the presence of ethanol, which acts as a competitive inhibitor. Moreover, the P450 enzymes induced by ethanol inactivate retinoic acid. These disruptions in the retinoic acid signaling pathway are believed to be responsible, at least in part, for fetal alcohol syndrome and for the development of a variety of cancers.