
31.1 The Nitrogenase Complex Fixes Nitrogen
✓ 3 Explain the centrality of nitrogen fixation to life, and describe how atmospheric nitrogen is converted into a biologically useful form of nitrogen.
To meet the kinetic challenge of nitrogen fixation, nitrogen-
In principle, the reduction of N2 to NH3 is a six-
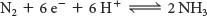
However, the biological reaction always generates at least 1 mol of H2 in addition to 2 mol of NH3 for each mole of N ≡ N. Hence, an input of two additional electrons is required:
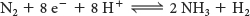
In most nitrogen-
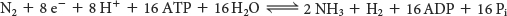
The product of the reduction, NH3, is a base in aqueous solutions, attracting a proton to form NH4+.
Note that O2 is required for oxidative phosphorylation to generate the ATP necessary for nitrogen fixation. However, the nitrogenase complex is exquisitely sensitive to inactivation by O2. To allow ATP synthesis and nitrogenase to function simultaneously, leguminous plants maintain a very low concentration of free O2 in their root nodules, the location of the nitrogenase. This is accomplished by binding O2 to leghemoglobin, a homolog of hemoglobin.
The Molybdenum–Iron Cofactor of Nitrogenase Binds and Reduces Atmospheric Nitrogen
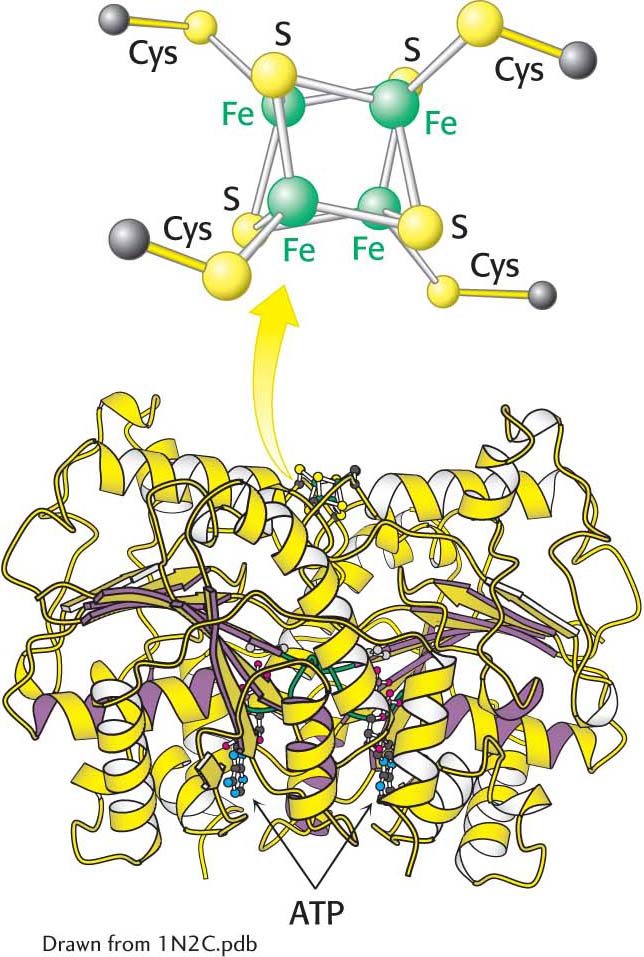
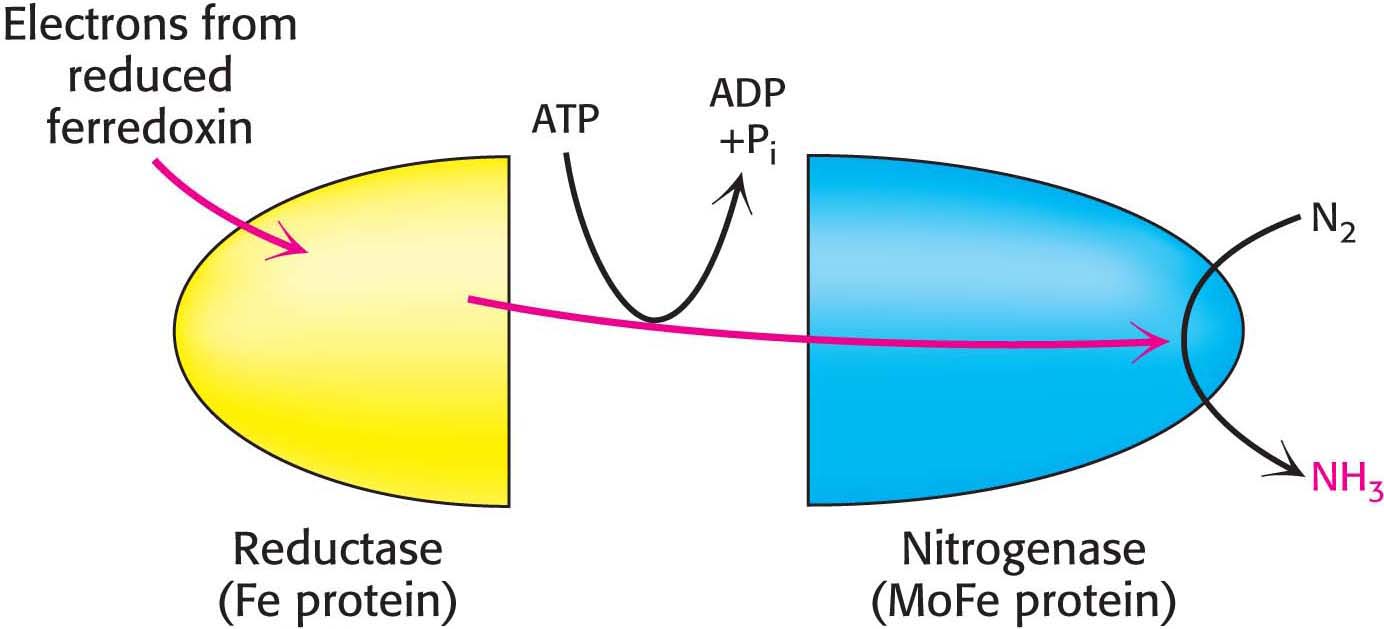
Both the reductase and the nitrogenase components of the complex are iron–
The nitrogenase component, an α2β2 tetramer (240 kDa), requires the FeMo cofactor, which consists of [Fe4-S3] and [Mo-
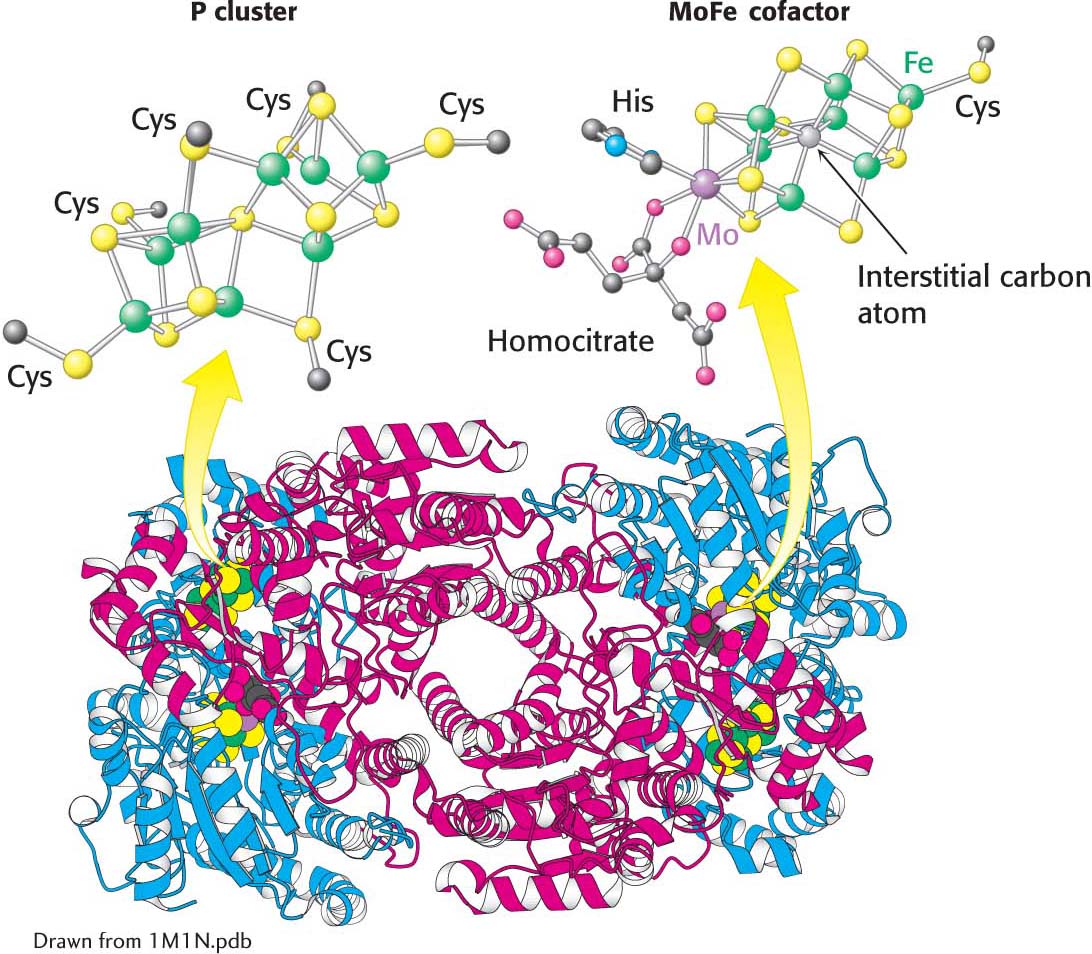
Electrons flow from the reductase to the nitrogenase at a part of the nitrogenase called the P cluster, an iron-
Ammonium Ion Is Incorporated into an Amino Acid Through Glutamate and Glutamine
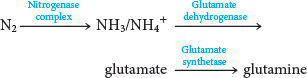
The next task in the assimilation of nitrogen into biomolecules is to incorporate ammonium ion (NH4+) into the biochemically versatile amino acids. α-Ketoglutarate and glutamate play pivotal roles as the acceptor of ammonium ion, forming glutamate and glutamine, respectively. Glutamate is synthesized from NH4+ and α-ketoglutarate, a citric acid cycle intermediate, by the action of glutamate dehydrogenase:
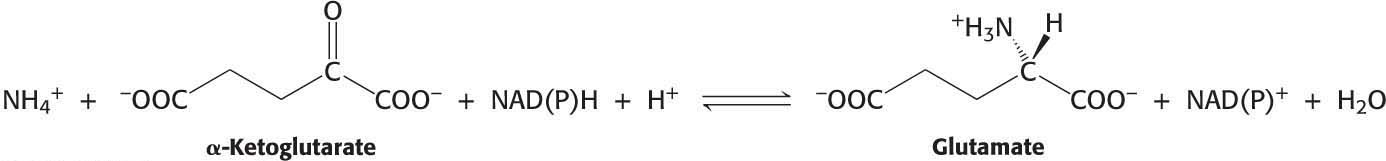
DID YOU KNOW?
Amidation describes a reaction in which an amine reacts with a carboxylic acid to yield an amide.
Recall that glutamate dehydrogenase also plays a key role in the removal of nitrogen from biological systems.
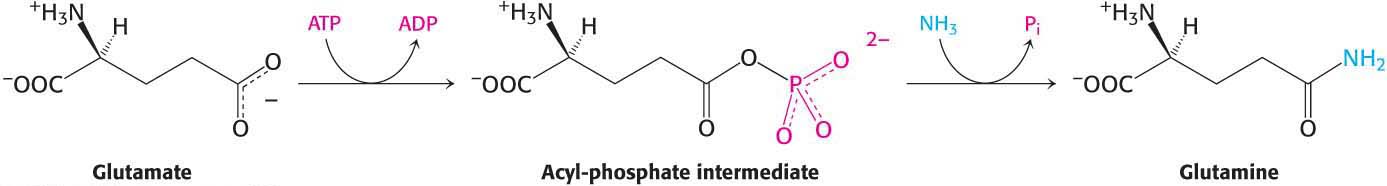
A second ammonium ion is incorporated into glutamate to form glutamine by the action of glutamine synthetase. This amidation is driven by the hydrolysis of ATP:
QUICK QUIZ 1
Trace the flow of nitrogen from atmospheric N2 to glutamine.
ATP participates directly in the reaction by phosphorylating the side chain of glutamate to form an acyl-
Glutamate subsequently donates its α-amino group to various ketoacids by transamination reactions to form most of the amino acids. Glutamine, the other major nitrogen donor, contributes its side-
