
32.5 Nucleotide Biosynthesis Is Regulated by Feedback Inhibition
✓ 6 Explain how nucleotide synthesis is regulated.
Nucleotide biosynthesis is regulated by feedback inhibition in a manner similar to the regulation of amino acid biosynthesis. These regulatory pathways ensure that the various nucleotides are produced in the required quantities.
Pyrimidine Biosynthesis Is Regulated by Aspartate Transcarbamoylase
Aspartate transcarbamoylase (ATCase) is a key enzyme for the regulation of pyrimidine biosynthesis in bacteria. ATCase is inhibited by CTP, the final product of pyrimidine biosynthesis, and stimulated by ATP. This coupling of inhibition by a pyrimidine nucleotide with stimulation by a purine nucleotide serves to balance the two nucleotide pools.

Carbamoyl phosphate synthetase also is a site of feedback inhibition in both bacteria and eukaryotes.
The Synthesis of Purine Nucleotides Is Controlled by Feedback Inhibition at Several Sites
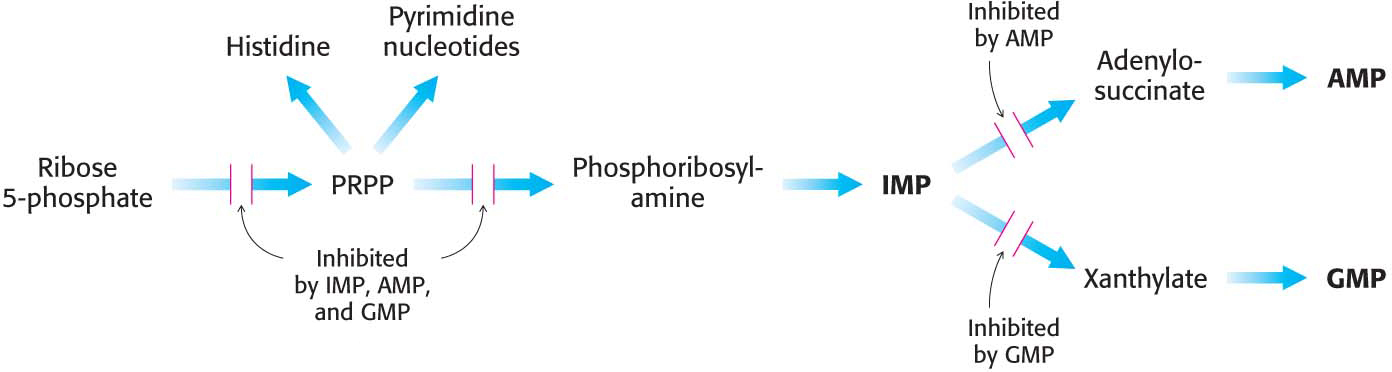
The regulatory scheme for purine nucleotides is more complex than that for pyrimidine nucleotides (Figure 32.10).
The committed step in purine nucleotide biosynthesis is the conversion of PRPP into phosphoribosylamine by glutamine phosphoribosyl amidotransferase. This important enzyme is feedback-
inhibited by many purine ribonucleotides. It is noteworthy that AMP and GMP, the final products of the pathway, synergistically inhibit the amidotransferase. Inosinate is the branch point in the synthesis of AMP and GMP. The reactions leading away from inosinate are sites of feedback inhibition. AMP inhibits the conversion of inosinate into adenylosuccinate, its immediate precursor. Similarly, GMP inhibits the conversion of inosinate into xanthylate, its immediate precursor.
As already noted, GTP is a substrate in the synthesis of AMP, whereas ATP is a substrate in the synthesis of GMP. This reciprocal substrate relation tends to balance the synthesis of adenine and guanine ribonucleotides.
 CLINICAL INSIGHT
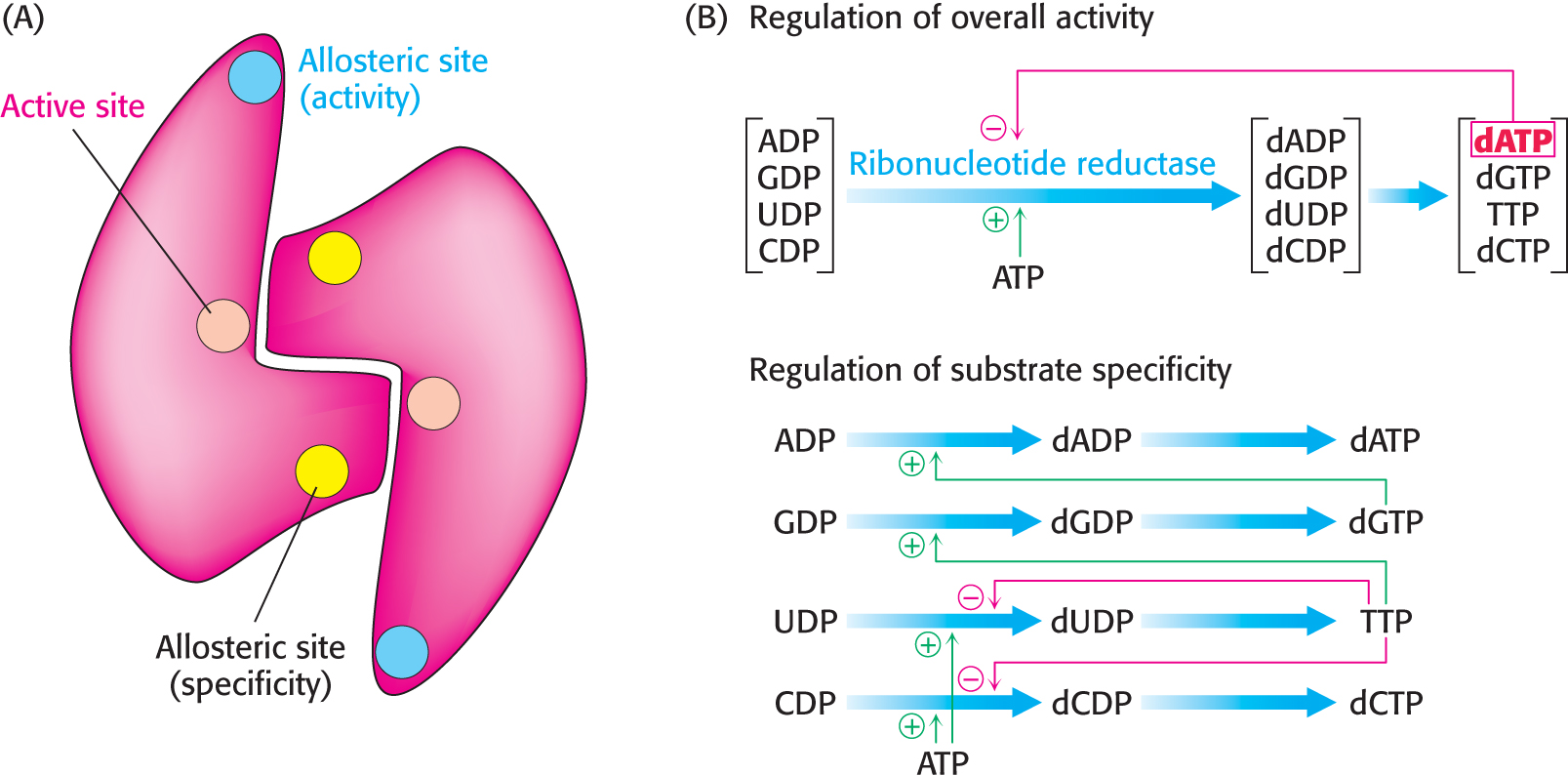
CLINICAL INSIGHTThe Synthesis of Deoxyribonucleotides Is Controlled by the Regulation of Ribonucleotide Reductase
The reduction of ribonucleotides to deoxyribonucleotides is precisely controlled by allosteric interactions. E. coli ribonucleotide reductase is a homodimeric enzyme. Each subunit contains two allosteric sites: one of them controls the overall activity of the enzyme, whereas the other regulates substrate specificity (Figure 32.11). The overall catalytic activity of ribonucleotide reductase is diminished by the binding of dATP, which signals an abundance of deoxyribonucleotides. The binding of ATP reverses this feedback inhibition. The binding of dATP or ATP to the substrate-
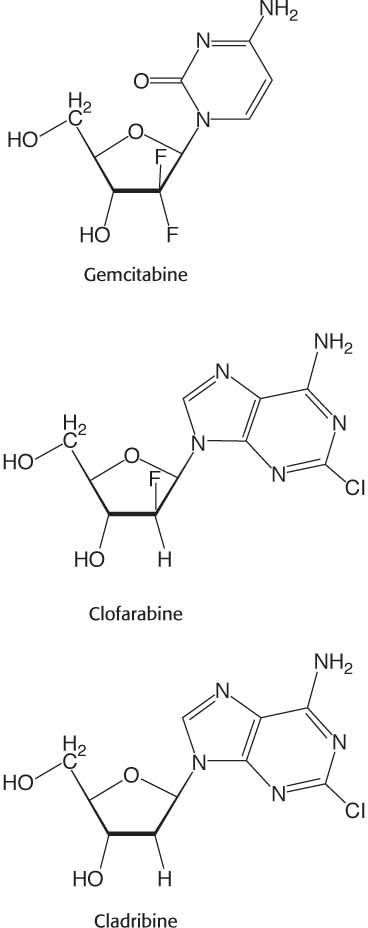
Ribonucleotide reductase is an attractive target for cancer therapy, and a number of clinically approved anticancer drugs mimic substrates and regulators of the enzyme. The pyrmidine analog gemcitabine, once converted to the diphosphate form in vivo, becomes a suicide inhibitor of the reductase used to treat advanced pancreatic cancer. The purine analogs clofarabine and cladribine, upon conversion to their triphosphate forms in vivo, are dATP analogs and act as allosteric inhibitors of the enzyme. Clofarabine is used to treat pediatric acute myeloid leukemia while cladribine is effective against some forms of chronic lymphoid leukemia.