
33.2 Nucleic Acid Strands Can Form a Double-Helical Structure
The covalent structure of nucleic acids accounts for their ability to carry information in the form of a sequence of bases. Other features of nucleic acid structure facilitate the process of replication—that is, the generation of two copies of a nucleic acid from one. DNA replication is the basis of cell duplication, growth, and, ultimately, reproduction. Replication depends on the ability of the bases found in nucleic acids to form specific base pairs in such a way that a helical structure consisting of two strands is formed.
The Double Helix Is Stabilized by Hydrogen Bonds and the Hydrophobic Effect
The discovery of the DNA double helix has been chronicled in many articles, books, and even a movie. James Watson and Francis Crick, using data obtained by Maurice Wilkins and Rosalind Franklin and simple molecular models, inferred a structural model for DNA that was the source of some remarkable insights into the functional properties of nucleic acids (Figure 33.11).
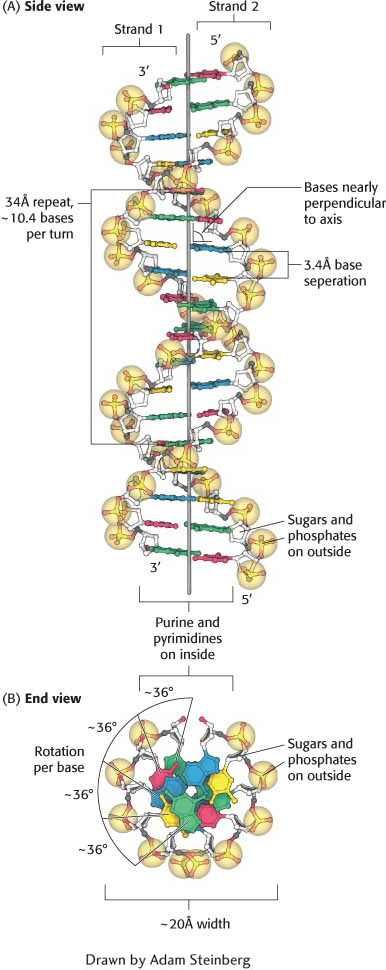
The features of the Watson–
Two helical DNA strands are coiled around a common axis, forming a right-
handed double helix. The strands run in opposite directions; that is, they have opposite directionality. One strand has a 5′-to- 3′ direction and pairs with the other strand, which has a 3′-to- 5′ orientation. The sugar–
phosphate backbones are on the outside of the helix; therefore, the purine and pyrimidine bases lie on the inside. Page 613The bases are nearly perpendicular to the helix axis, and adjacent bases are separated by approximately 3.4 Å. The helical structure repeats on the order of every 34 Å, with about 10.4 bases per turn of helix. There is a rotation of nearly 36 degrees per base (360 degrees per full turn/10.4 bases per turn).
The diameter of the helix is about 20 Å.
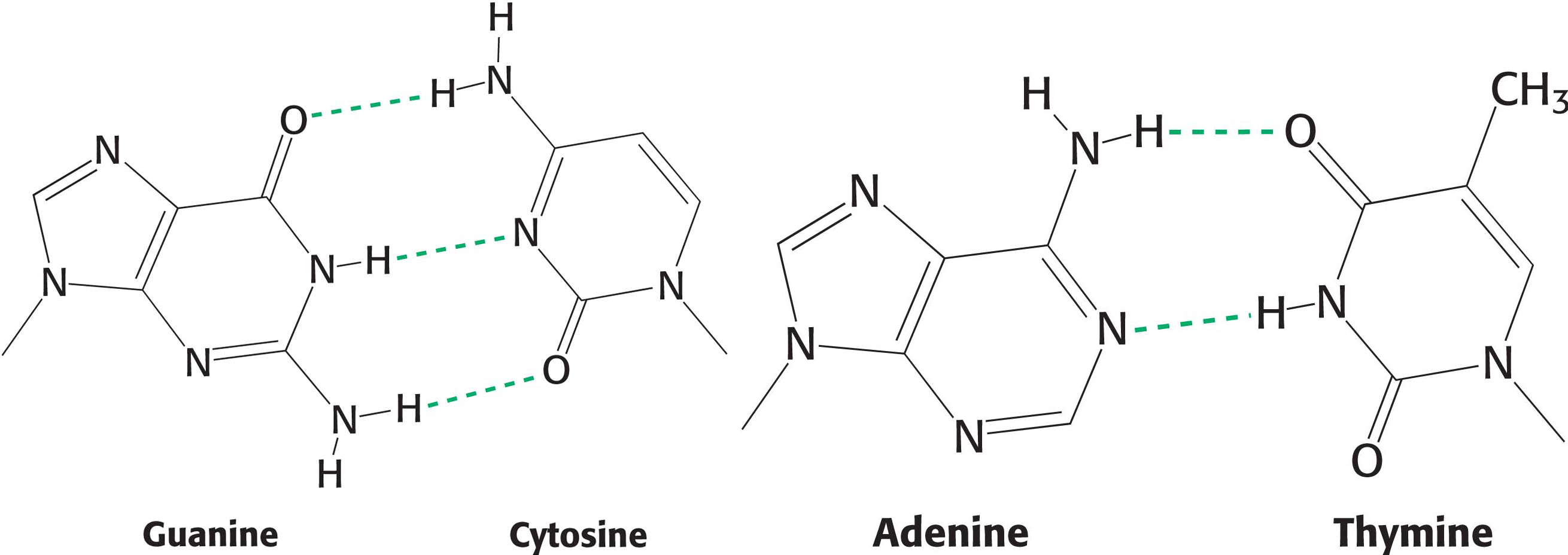
How is such a regular structure able to accommodate an arbitrary sequence of bases, given the different sizes and shapes of the purines and pyrimidines? Restricting the combinations in which bases on opposite strands can be paired—
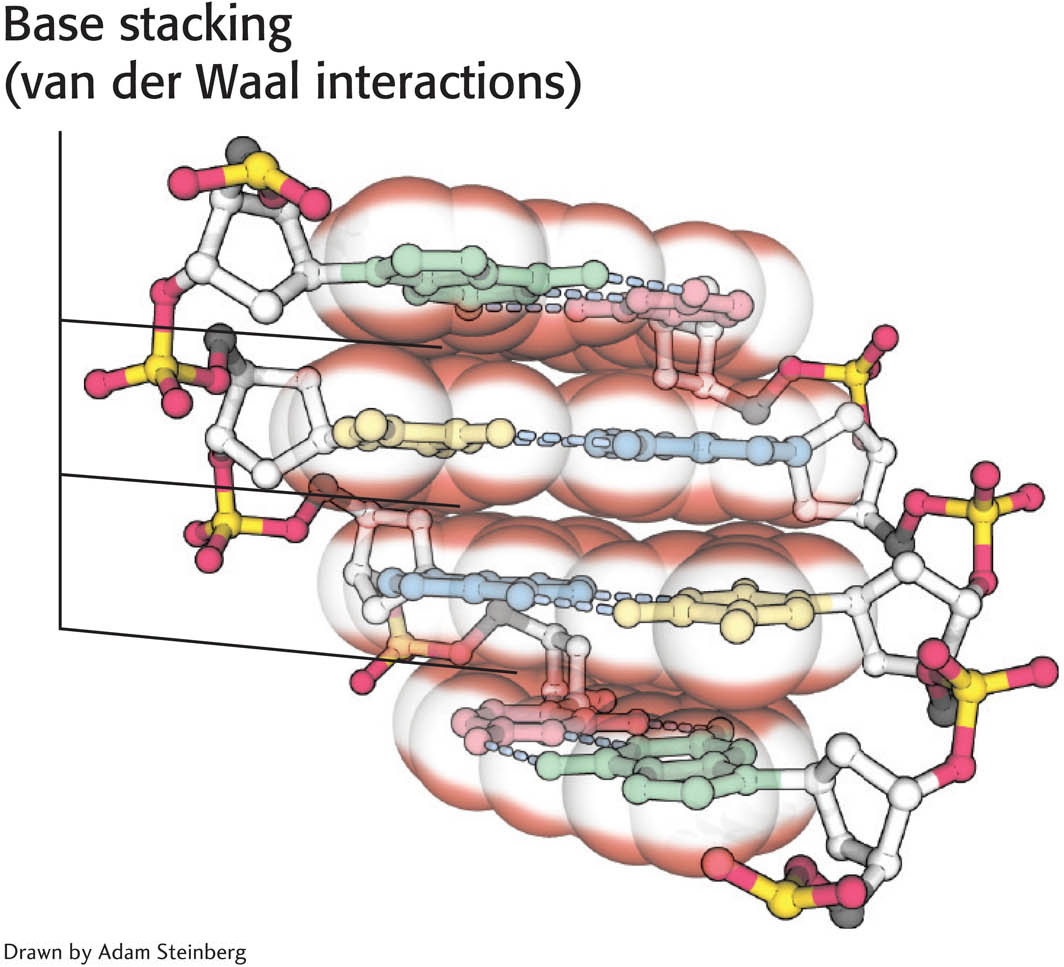
The stacking of bases one on top of another also contributes to the stability of the double helix in two ways (Figure 33.13). First, the double helix is stabilized by the hydrophobic effect: hydrophobic interactions between the bases drive the bases to the interior of the helix, resulting in the exposure of the more polar surfaces to the surrounding water. This arrangement is reminiscent of protein folding in which hydrophobic amino acids are interior in the protein and hydrophilic amino acids are exterior. Second, stacked bases attract one another through van der Waals forces, a phenomenon called base stacking. Energies associated with van der Waals interactions are quite small, such that typical interactions contribute from 2 to 4 kJ mol−1 (0.5 to 1.0 kcal mol−1) per atom pair. In the double helix, however, a large number of atoms are in van der Waals contact, and the net effect, summed over these atom pairs, is substantial. As stated earlier, the base sequence is the information content of nucleic acids. Note that, in DNA, this information is tucked into the relative safety of the interior of the double helix.
The Double Helix Facilitates the Accurate Transmission of Hereditary Information
The double-
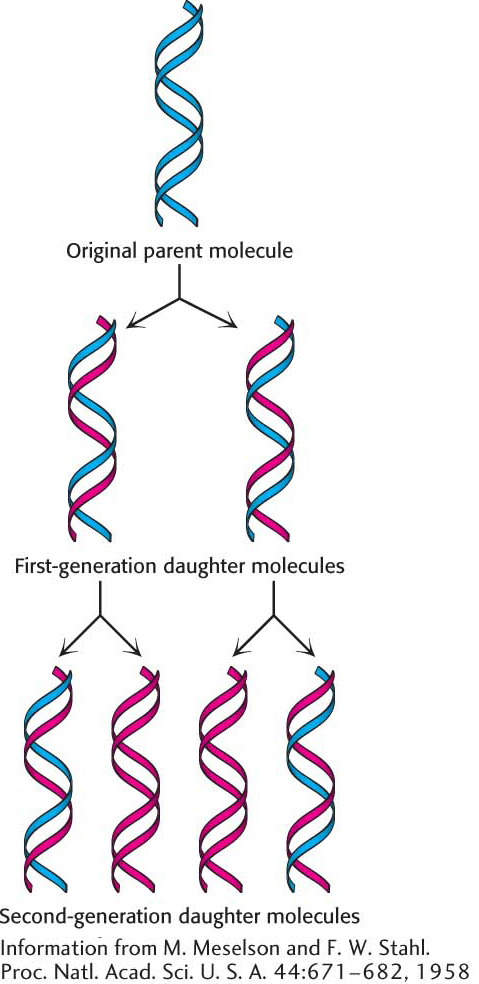
Meselson and Stahl Demonstrated That Replication Is Semiconservative
How do we know that DNA replication is semiconservative? Matthew Meselson and Franklin Stahl carried out an ingenious experiment to establish this fundamental point. They labeled the parent DNA with 15N, a heavy isotope of nitrogen, to make it denser than ordinary DNA. The labeled DNA was generated by growing E. coli for many generations in a medium that contained 15NH4Cl as the sole nitrogen source. After the incorporation of heavy nitrogen was complete, the bacteria were abruptly transferred to a medium that contained 14N, the ordinary, lighter isotope of nitrogen. The question asked was: What is the distribution of 14N and 15N in the DNA molecules after successive rounds of replication?
The distribution of 14N and 15N was revealed by the technique of density-
DNA was then extracted from the bacteria at various times after they had been transferred from a 15N to a 14N medium and centrifuged. Analysis of these samples showed that there was a single band of DNA after one generation. The density of this band was precisely halfway between the densities of the 14N DNA and 15N DNA bands (Figure 33.16). The absence of 15N DNA indicated that parental DNA was not preserved as an intact unit after replication. The absence of 14N DNA indicated that all of the daughter DNA derived some of their atoms from the parent DNA. This proportion had to be half, because the density of the hybrid DNA band was halfway between the densities of the 14N DNA and 15N DNA bands.
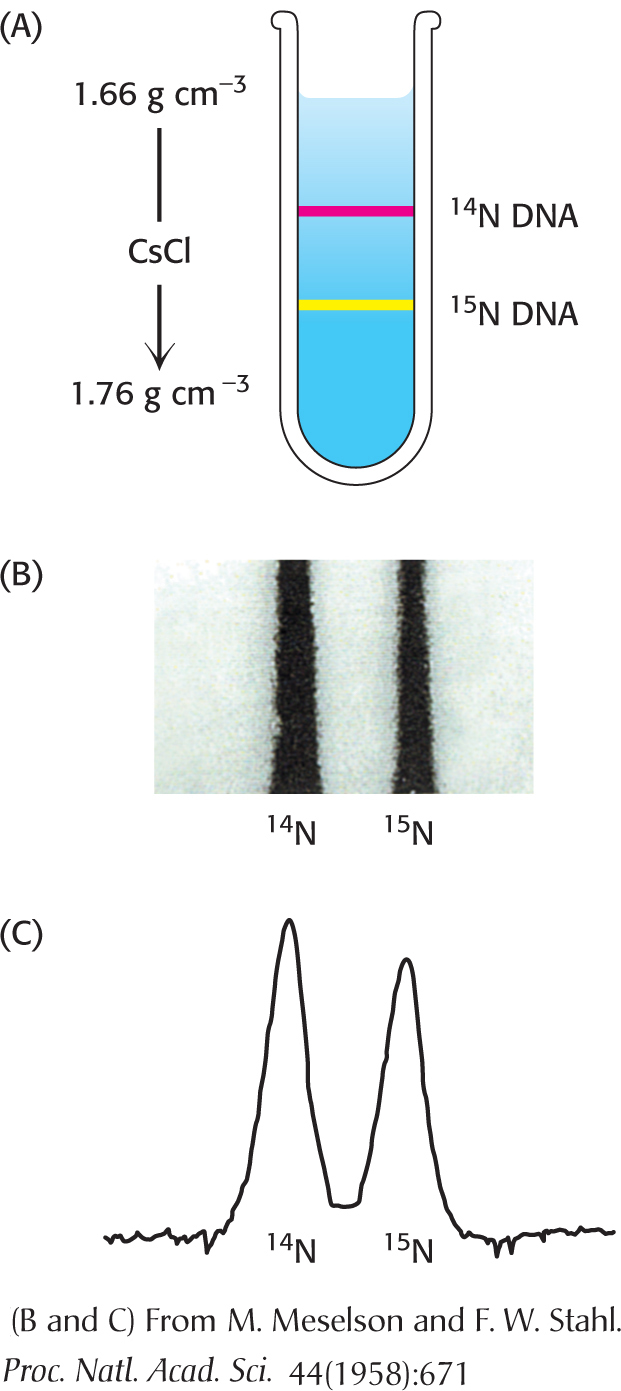
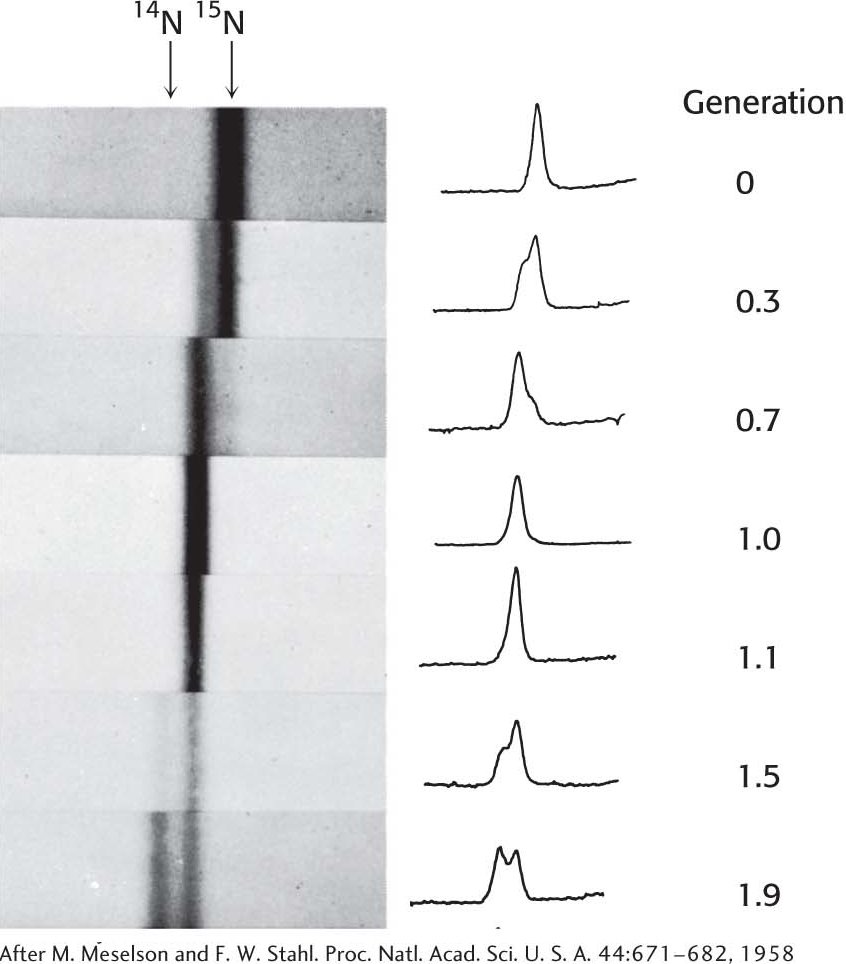
After two generations, there were equal amounts of two bands of DNA. One was hybrid DNA, and the other was 14N DNA. Meselson and Stahl concluded from these incisive experiments “that the nitrogen in a DNA molecule is divided equally between two physically continuous subunits; that following duplication, each daughter molecule receives one of these; and that the subunits are conserved through many duplications.” Their results agreed perfectly with the Watson–
The Strands of the Double Helix Can Be Reversibly Separated
During DNA replication and transcription, the two strands of the double helix must be separated from one another, at least in a local region. In other words, sometimes the archival information must be accessible to be useful. In the laboratory, the two strands of the double helix can be separated by heating a solution of DNA. The thermal energy causes the DNA molecules to move to such a degree that the weak forces holding the helix together, such as the hydrogen bonds between the bases, break apart. The dissociation of the double helix is called melting or denaturation, and it occurs relatively abruptly at a certain temperature. The melting temperature Tm is defined as the temperature at which half the helical structure is lost. Strands can also be separated by adding acid or alkali to ionize the nucleotide bases and disrupt base-
QUICK QUIZ 1
What are the chemical forces that stabilize the double helix?
Hydrogen bonds between base pairs and van der Waals interactions among the bases. The van der Waals interactions come into play because of the hydrophobic effect, which forces the bases to the interior of the helix.
Separated complementary strands of nucleic acids spontaneously reassociate to form a double helix when the temperature is lowered below Tm. This renaturation process is sometimes called annealing. The facility with which double helices can be melted and then reassociated is crucial for the biological functions of nucleic acids.