40.3 Bacteria and Eukaryotes Differ in the Initiation of Protein Synthesis
✓ 6 Compare and contrast bacterial and eukaryotic protein synthesis.
The basic plan of protein synthesis in eukaryotes and archaea is similar to that in bacteria. The major structural and mechanistic themes recur in all domains of life. However, eukaryotic protein synthesis requires more protein components than does bacterial protein synthesis, and some steps are more complicated. Some noteworthy similarities and differences are as follows:
Ribosomes. Eukaryotic ribosomes are larger. They consist of a 60S large subunit and a 40S small subunit, which come together to form an 80S complex having a mass of 4.2 MDa, compared with 2.7 MDa for the bacterial 70S ribosome. The 40S subunit contains an 18S RNA that is homologous to the bacterial 16S RNA. The 60S subunit contains three RNAs: the 5S RNA, which is homologous to the bacterial 5S rRNA; the 28S RNA, which is homologous to the bacterial 23S molecule; and the 5.8S RNA, which is homologous to the 5′ end of the bacterial 23S RNA.
Initiator tRNA. In eukaryotes, the initiating amino acid is methionine rather than N-formylmethionine. However, as in bacteria, a special tRNA participates in initiation. This aminoacyl-
tRNA is called Met- tRNAi or Met- tRNAf (the subscript “i” stands for initiation, and “f” indicates that it can be formylated in vitro). Page 729Initiation. The initiating codon in eukaryotes is always AUG. Unlike bacteria, eukaryotes do not have a specific purine-
rich sequence on the 5′ side to distinguish initiator AUGs from internal ones. Instead, the AUG nearest the 5′ end of mRNA is usually selected as the start site. Eukaryotes utilize many more initiation factors than do bacteria, and their interplay is much more complex. The prefix eIF denotes a eukaryotic initiation factor. 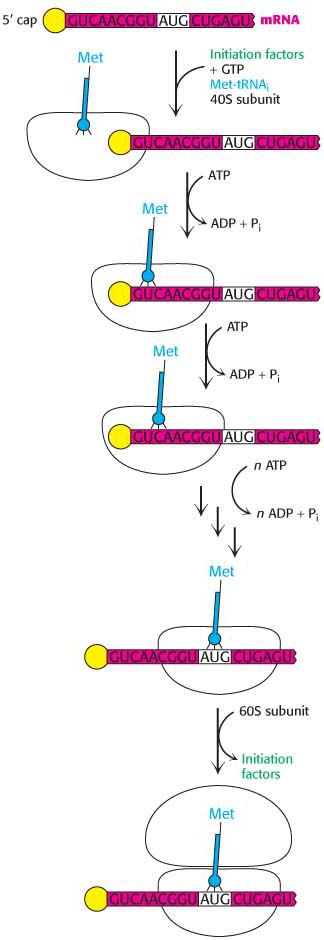 Figure 40.12 Eukaryotic translation initiation. In eukaryotes, translation initiation starts with the assembly of a complex on the 5′ cap that includes the 40S subunit and Met-
Figure 40.12 Eukaryotic translation initiation. In eukaryotes, translation initiation starts with the assembly of a complex on the 5′ cap that includes the 40S subunit and Met-tRNAi. Driven by ATP hydrolysis, this complex scans the mRNA until the first AUG is reached. The 60S subunit is then added to form the 80S initiation complex. Initiation begins with the formation of a ternary complex consisting of the 40S ribosome and Met-
tRNAi in association with eIF- 2. The complex is called the 43S preinitiation complex (PIC). The PIC binds to the 5′ end of mRNA and begins searching for an AUG codon by moving step- by- step in the 3′ direction. Initiation factor eIF- 4E binds to the 5′ cap of the mRNA and facilitates binding of PIC to the mRNA (Figure 40.12). This scanning process is catalyzed by helicases that move along the mRNA powered by ATP hydrolysis. Pairing of the anticodon of Met- tRNAi with the AUG codon of mRNA signals that the target has been found. In almost all cases, eukaryotic mRNA has only one start site and, hence, is the template for a single protein. In contrast, a bacterial mRNA can have multiple Shine– Dalgarno sequences and, hence, multiple start sites, and it can serve as a template for the synthesis of several proteins. The difference in initiation mechanisms between bacteria and eukaryotes is, in part, a consequence of the difference in RNA processing. The 5′ end of mRNA is readily available to ribosomes immediately after transcription in bacteria. In contrast, in eukaryotes pre-
mRNA must be processed and transported to the cytoplasm before translation is initiated. The 5′ cap provides an easily recognizable starting point. The Structure of mRNA. Eukaryotic mRNA is circular (Figure 40.13). Soon after the PIC binds the mRNA, eIF-
4G links eIF- 4E to a protein associated with the poly (A) tail, the poly (A)-binding protein I (PABPI). Cap and tail are thus brought together to form a circle of mRNA. The benefits of circularization of mRNA remain to be determined, but a requirement for circularization may prevent translation of mRNA molecules that have lost their poly A tails. 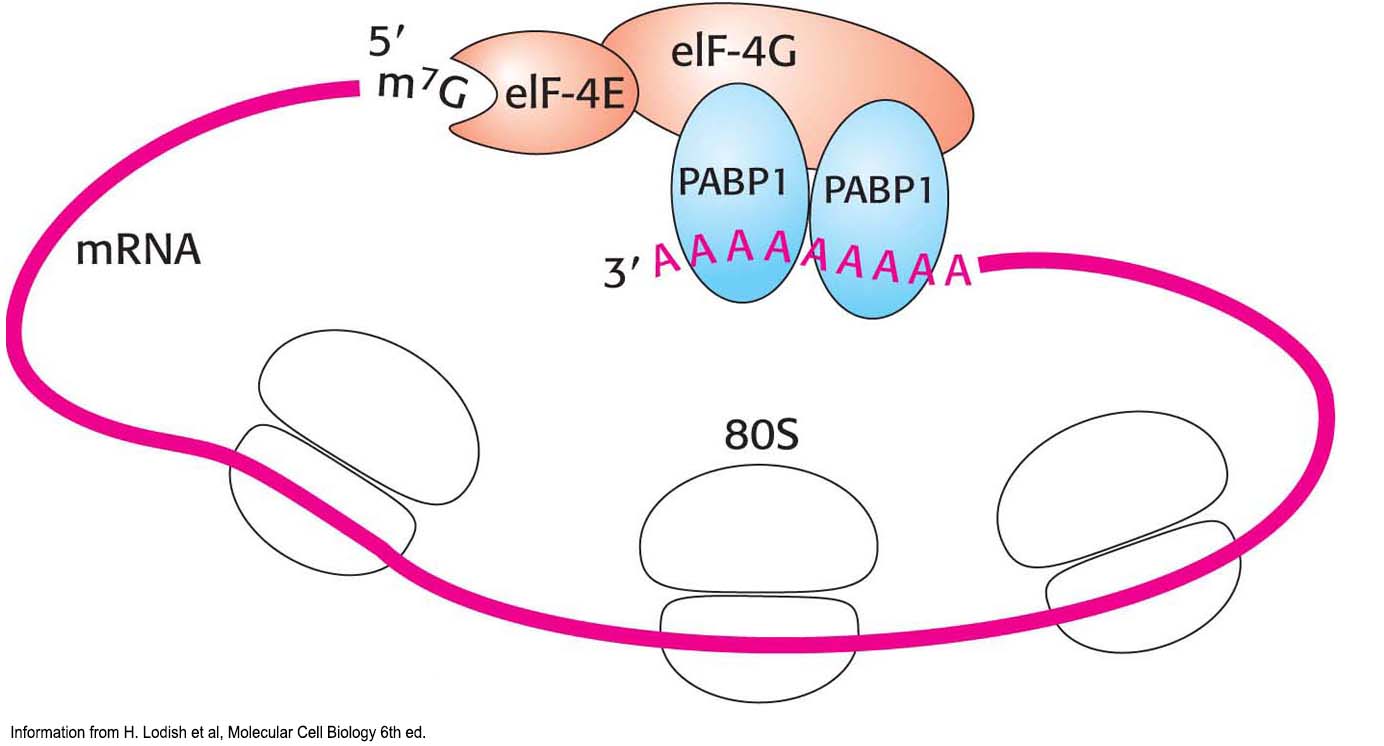 Figure 40.13 Protein interactions circularize eukaryotic mRNA.
Figure 40.13 Protein interactions circularize eukaryotic mRNA.Elongation and Termination. Eukaryotic elongation factors EF1α and EF1βγ are the counterparts of bacterial EF-
Tu and EF- Ts, whereas eukaryotic EF2 corresponds to the EF- G (translocase) in bacteria. Termination in eukaryotes is carried out by a single release factor, eRF1, compared with two in bacteria. Initiation factor eIF- 3 accelerates the activity of eRF- 1. Organization. The components of the translation machinery in higher eukaryotes are organized into large complexes associated with the cytoskeleton. This association is believed to facilitate the efficiency of protein synthesis. Recall that the organization of elaborate biochemical processes into physical complexes is a recurring theme in biochemistry.
 CLINICAL INSIGHT
CLINICAL INSIGHTMutations in Initiation Factor 2 Cause a Curious Pathological Condition
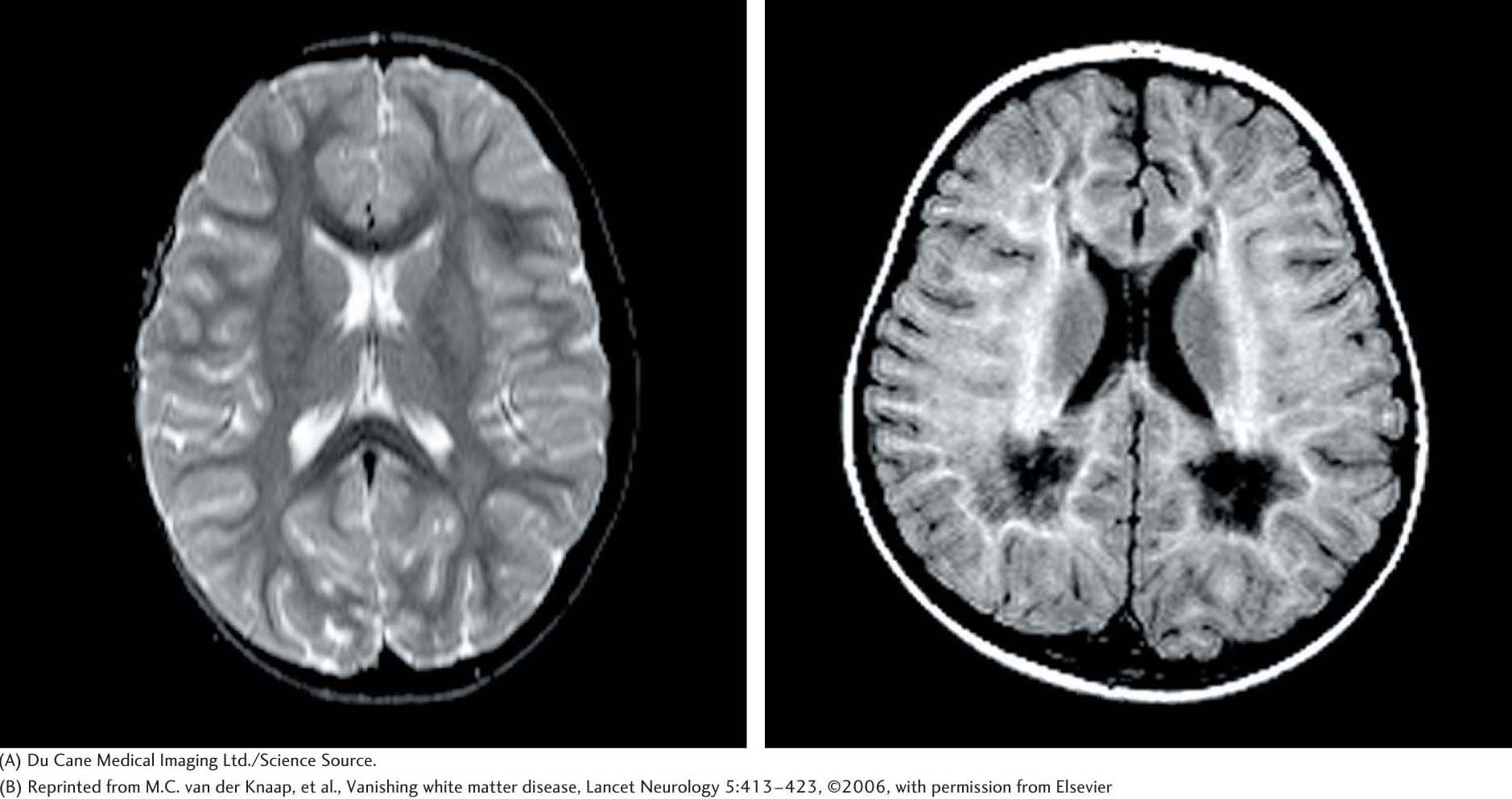
Mutations in eukaryotic initiation factor 2 result in a mysterious disease, called vanishing white matter (VWM) disease, in which nerve cells in the brain disappear and are replaced by cerebrospinal fluid (Figure 40.14). The white matter of the brain consists predominately of nerve axons that connect the gray matter of the brain to the rest of the body. Death, resulting from fever or extended coma, is anywhere from a few years to decades after the onset of the disease. An especially puzzling aspect of the disease is its tissue specificity. A mutation in a biochemical process as fundamental to life as protein-