40.6 Protein Synthesis Is Regulated by a Number of Mechanisms
✓ 7 Explain how protein synthesis can be regulated.
Protein synthesis can be controlled at various stages in the translation process. We will first examine the control of protein synthesis by iron. An essential nutrient, iron is required for the synthesis of hemoglobin, cytochromes, and many other proteins. Recall that iron is a key component of the respiratory chain, the primary source of ATP in aerobic organisms (Chapter 20). However, excess iron can be quite harmful because, untamed by a suitable protein environment, iron can initiate a range of free-radical reactions that damage proteins, lipids, and nucleic acids. Animals have evolved sophisticated systems for the accumulation of iron in times of scarcity and for the safe storage of excess iron for later use. Key proteins include transferrin, a transport protein that carries iron in the blood serum, transferrin receptor, a membrane protein that binds iron-loaded transferrin and facilitates its entry into cells by the process of receptor mediated endocytosis, and ferritin, an iron-storage protein found primarily in the liver and kidneys.
Messenger RNA Use Is Subject to Regulation
Ferritin and transferrin-receptor expression levels are reciprocally related in their responses to changes in iron levels. When iron is scarce, the amount of transferrin receptor increases and little or no new ferritin is synthesized. Interestingly, the extent of mRNA synthesis for these proteins does not change correspondingly. Instead, regulation takes place at the level of translation.
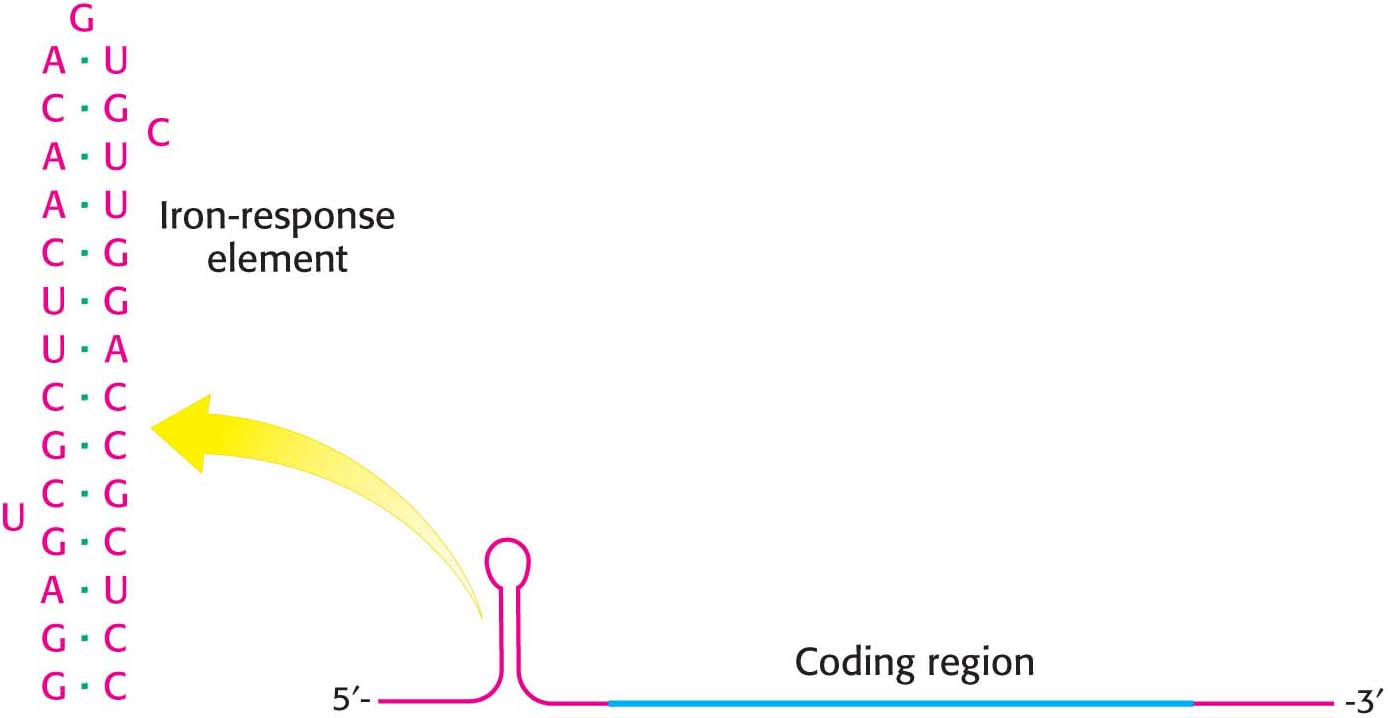
Let us consider ferritin first. Ferritin mRNA includes a stem-loop structure termed an iron-response element (IRE) in its 5′ untranslated region (Figure 40.20). This stem-loop structure binds a protein, called an IRE-binding protein (IRE-BP), which blocks the initiation of translation. When the iron level increases, the IRE-BP binds iron. The IRE-BP bound to iron cannot bind RNA, because the binding sites for iron and RNA substantially overlap. Thus, in the presence of iron, ferritin mRNA is released from the IRE-BP and translated to produce ferritin, which sequesters the excess iron.
Page 736
The Stability of Messenger RNA Also Can Be Regulated
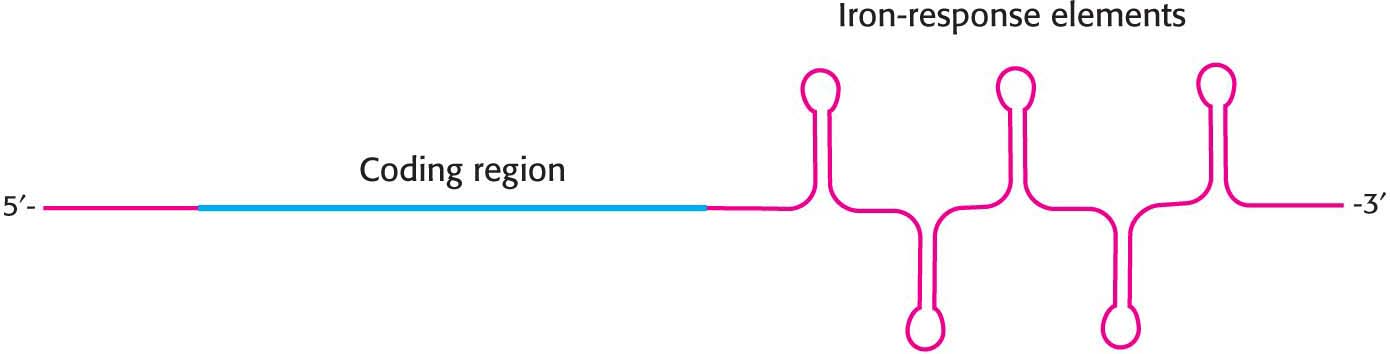
Transferrin-receptor mRNA also has several IRE-like regions. Unlike those in ferritin mRNA, these regions are located in the 3′ untranslated region rather than in the 5′ untranslated region (Figure 40.21). In the absence of iron, IRE-BP binds to these IREs. However, given the location of these binding sites, the transferrin-receptor mRNA can still be translated. What happens when the iron level increases? IRE-BP bound to iron no longer binds transferrin-receptor mRNA. Freed from the IRE-BP, transferrin-receptor mRNA is rapidly degraded. Thus, an increase in the cellular iron level leads to the destruction of transferrin-receptor mRNA and, hence, a reduction in the production of transferrin-receptor protein. The IRE-BP serves as an iron sensor. If there is enough iron present to bind the IRE-BP, then ferritin will be synthesized to store the iron. Likewise, if enough iron is present to be stored, there is no need to produce the receptor for its uptake. An environmental signal—the concentration of iron—controls the translation of proteins required for the metabolism of this metal.
Small RNAs Can Regulate mRNA Stability and Use
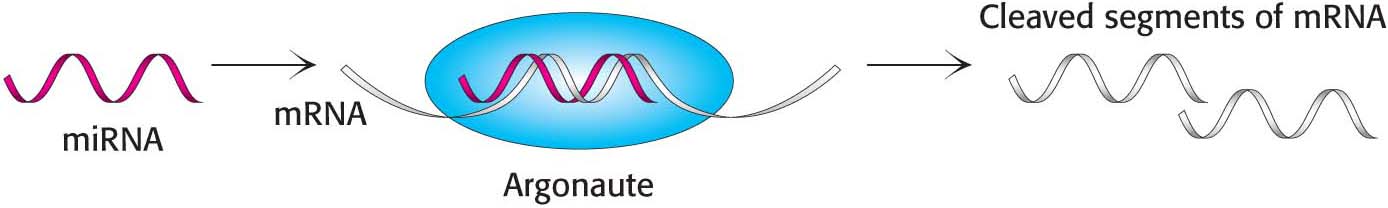
In recent years, an entirely new means of regulating protein synthesis was discovered. RNA interference (RNAi) was originally identified as a process that leads to mRNA degradation, induced by the presence of double-stranded RNA (dsRNA). RNAi, observed in all eukaryotes, may have evolved as a protective mechanism against viruses that employ dsRNA at some point in the viral life cycle. In RNAi, Dicer, an RNase, cleaves double-stranded RNAs into 21-nucleotide fragments. Single-stranded components of the cleavage products, called small interfering RNA (siRNA), are bound by members of a class of proteins called the Argonaute family to form an RNA induced silencing complex (RISC). This complex uses the single-stranded siRNA to locate and degrade complementary mRNAs (Figure 40.22).
The investigation of RNAi led to the discovery of a new class of RNAs that regulate gene expression posttranscriptionally. Whereas siRNAs are derived from exogenous RNA (either from a virus or introduced experimentally), microRNAs (miRNAs) are a class of noncoding RNAs generated from larger transcripts produced by RNA polymerase II and, in some cases, RNA polymerase III. These transcripts are cleaved by specific nucleases to yield fragments of double-stranded RNA (dsRNA) ∼21 base pairs in length. Further processing leads to the degradation of one of the strands of this ∼21-base-pair dsRNA; the other strand associates with Argonaute to form RISC, also called a microRNP (miRNP). The complex is believed to function in one of two ways. If the small RNA component of the complex binds to an mRNA by precise Watson–Crick base-pairing, the mRNA is destroyed. If the complementarity is not precise, the mRNA is simply not translated. In either mode of action, protein synthesis is inhibited.
Page 737
Gene regulation by miRNAs was originally thought to be limited to a small number of species. However, subsequent studies have revealed that this mode of gene regulation is nearly ubiquitous in eukaryotes. Indeed, more than 700 miRNAs encoded by the human genome have been identified. Each miRNA can regulate many different genes because many different target sequences are present in each mRNA. An estimated 60% of all human genes are regulated by one or more miRNAs. MicroRNA has been shown to regulate a wide variety of biochemical processes, including development, cell differentiation, and oncogenesis—the development of the cancer.
The discovery of small RNAs that regulate gene expression is among the most exciting biochemical discoveries in recent years. Elucidating the precise mechanism of action of these RNAs is an active area of biochemical research.