
7.4 Enzymes Can Be Studied One Molecule at a Time
Most experiments that are performed to determine an enzyme characteristic require an enzyme preparation in a buffered solution. Even a few microliters of such a solution will contain millions of enzyme molecules. Much that we have learned about enzymes thus far has come from such experiments, called ensemble studies. A basic assumption of ensemble studies is that all of the enzyme molecules are the same or very similar. When we determine an enzyme property such as the value of KM in ensemble studies, that value is of necessity an average value of all of the enzyme molecules present. However, we know that molecular heterogeneity—
Consider this hypothetical situation. A Martian visits Earth to learn about higher education. The spacecraft hovers high above a university, and the Martian meticulously records how the student population moves about campus. Much information can be gathered from such studies: where students are likely to be at certain times on certain days; which buildings are used when and by how many. Now, suppose our visitor developed a high-
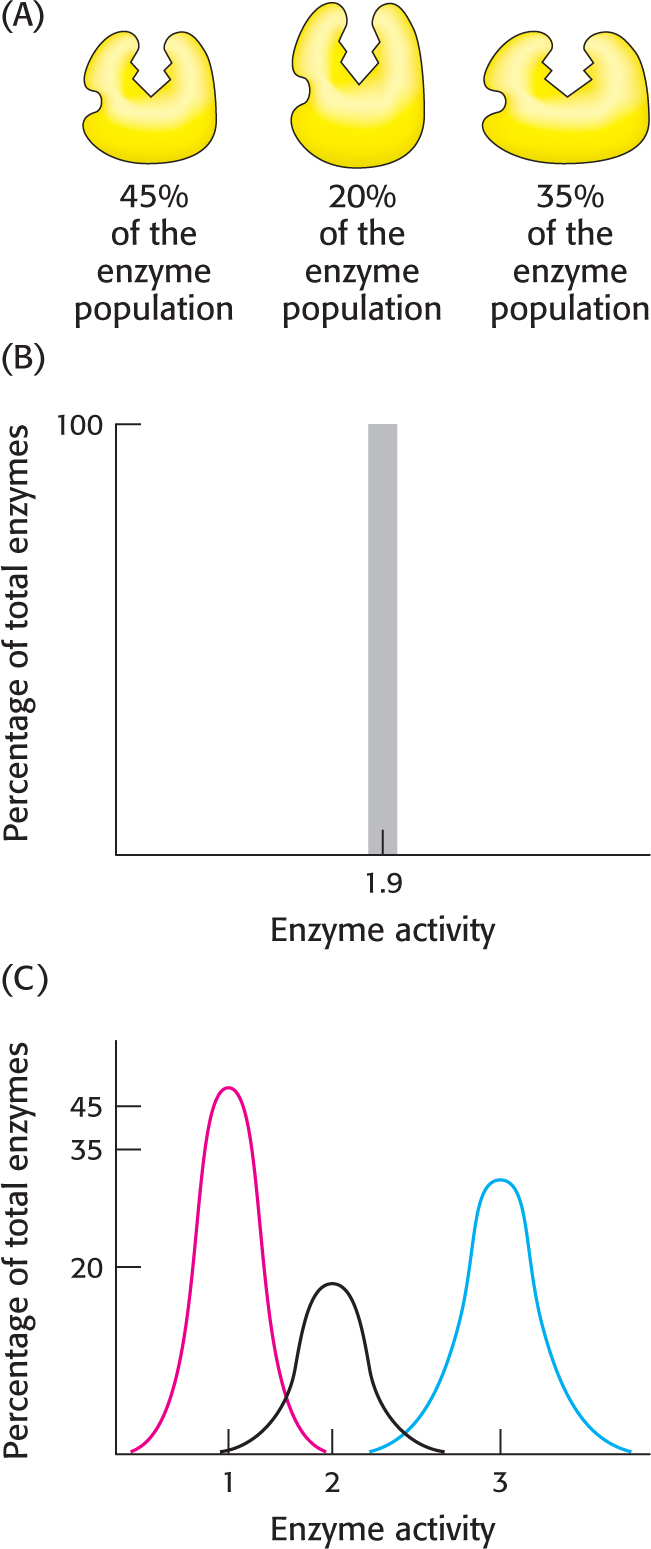
Let us leave the Martian to his observations and consider a more biochemical situation. Figure 7.15A shows an enzyme that displays molecular heterogeneity, with three active forms that catalyze the same reaction but at different rates. These forms have slightly different stabilities, but thermal noise is sufficient to power conversion among the forms. Each form is present as a fraction of the total enzyme population as indicated. If we were to perform an experiment to determine enzyme activity under a particular set of conditions with the use of ensemble methods, we would get a single value, which would represent the average of the heterogeneous assembly (Figure 7.15B). However, were we to perform a sufficient number of single-
The development of powerful techniques has enabled biochemists to look into the workings of individual molecules. We are now able to observe events at a molecular level that reveal rare or transient structures and fleeting events in a reaction sequence, as well as to measure mechanical forces affecting or generated by an enzyme. Single-