Chapter 2. Analyzing Enzymes and Enzyme Kinetics I
Learning Objectives
General Purpose
Conceptual
- Be able to apply the general model of enzyme catalysis to real reactions.
- Be able to correlate changes in light absorption to changes in the concentration of a substance.
- Be able to use a standard curve to determine the concentration of an unknown.
Procedural
- Be able to accurately use a pipette.
- Be able to accurately use a spectrophotometer
General Purpose
This lab will introduce the concept of enzyme-catalyzed reactions and their role in biology. It will also serve to introduce you to a number of laboratory techniques as well as some important aspects of data collection and analysis.
General Background Information
Chemical reactions are the basis of most biological processes. Many of these reactions require a catalyst to occur at rates that are necessary for living systems. A catalyst increases the rate of a chemical reaction without being consumed or changed during the process. This change in the reaction rate is accomplished by reducing the activation energy (Ea) between reactants and products (Figure 2-1).
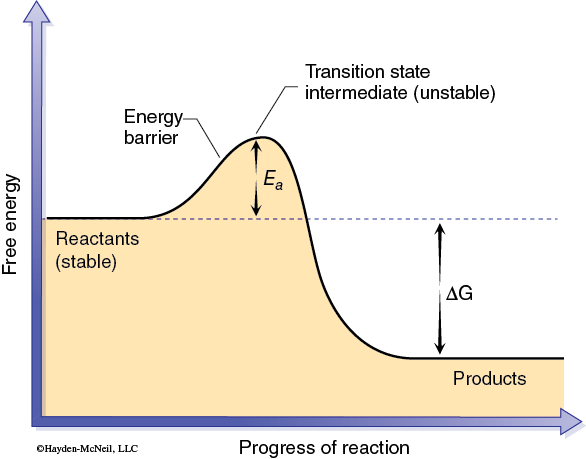
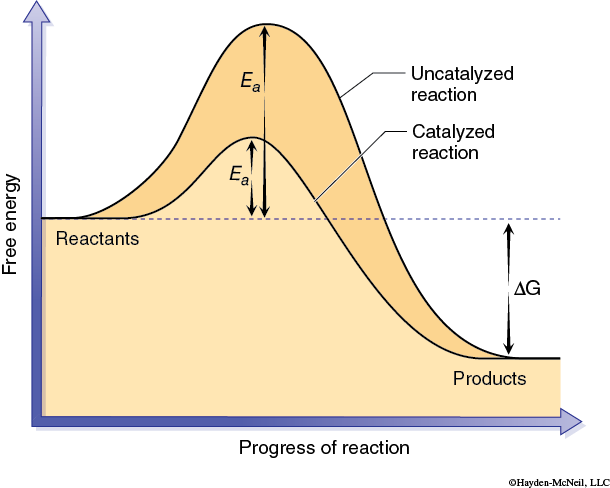
The activation energy can be thought of as the energy required to produce an activated complex needed for a reaction to occur. This activated complex may involve bringing two or more reactant molecules together in a proper alignment, or altering the shape of a particular reactant, or both. In effect a catalyst allows an activated complex, or transition state, to form with less energy (Figure 2-2).
The chemical reactions that are part of life are made possible by organic catalysts known as enzymes. Unlike other types of catalysts, enzymes are highly specific. Typically, a single enzyme molecule will catalyze only one type of chemical reaction. Biologically this means that almost every one of the innumerable reactions that occur in an organism is catalyzed by a different enzyme. Most enzymes are proteins and therefore have complex three-dimensional shapes or conformations. The conformation of the enzyme is related to how the enzyme functions. A general model of enzyme catalysis begins with the enzyme and substrate (reactant) molecules separate. The enzyme binds the substrate, and they form the enzyme–substrate complex (Figure 2-3).
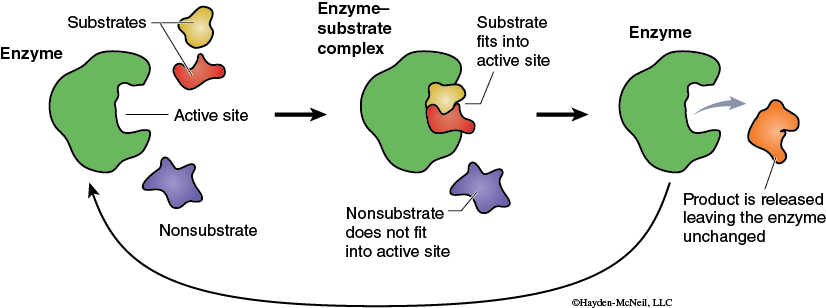
This activated enzyme–substrate complex is now in the proper alignment and the reaction proceeds. The result of the reaction is the conversion of the substrate into the product. The product, being chemically different from the substrate, no longer is attracted to the enzyme (Figure 2-3). The product and the enzyme separate, and the enzyme molecule is now available to combine with new substrate and go through this process again. A more detailed model of enzyme function is the Induced-Fit Model (Figure 2-4).
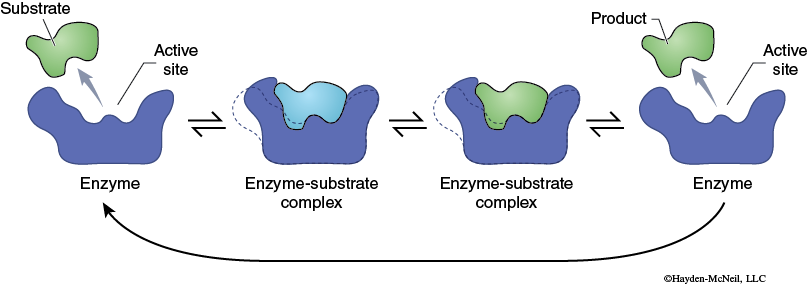
This model, the Induced-Fit Model, again begins with the enzyme and substrate molecules being separate from one another. The substrate is attracted to the active site on the enzyme and interacts chemically with the amino acids in the active site. This interaction causes the conformation of the enzyme to change which results in the formation of the activated complex, and the reaction proceeds. The substrate is converted into the product. The product interacts differently with the active site, causing a different enzyme conformation. This results in the product leaving the active site. The enzyme returns to its original conformation and can now bind new substrate and catalyze the reaction once again.
These models point out two important properties of this process. First, the enzyme and the substrate molecules come into physical contact and interact chemically. Second, the specificity of an enzyme is due to the conformation of the enzyme relative to the substrate molecule(s). Different enzymes have different conformations, especially at the active sites, which results in very specific catalysis (Figure 2-5).
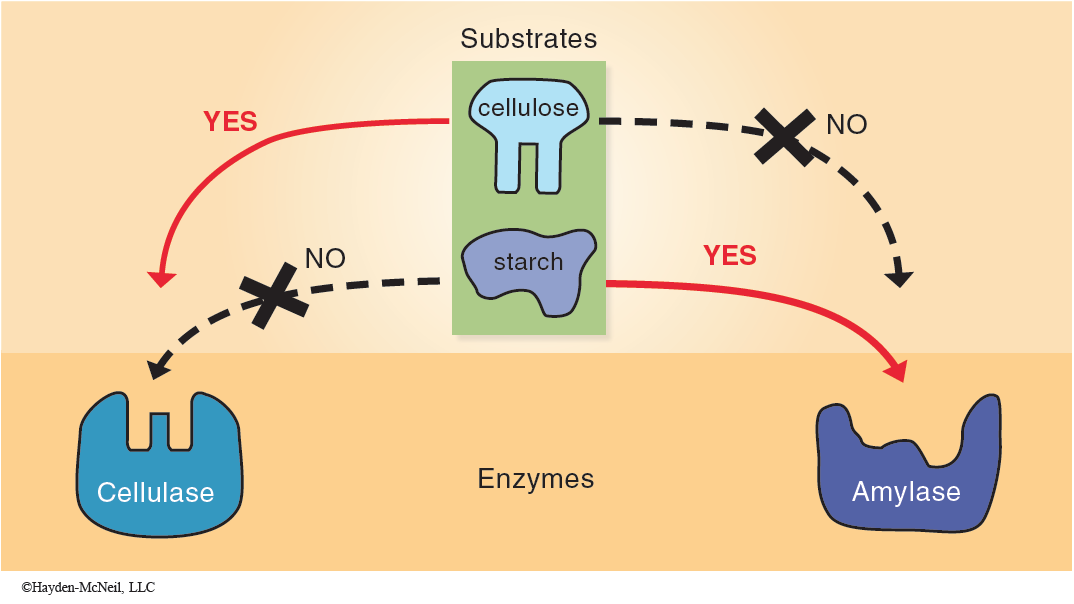
Since the correct conformation of the enzyme and the active site are critical to catalytic activity, any factors which affect the shape of an enzyme also affect the rate of the reaction that the enzyme catalyzes.
Specific Background Information
The reaction we will follow in this series of enzyme laboratories is the conversion of catechol to benzoquinone (Figure 2-6). Catechol is a naturally occurring compound found in many types of plants; it is especially common in many fruits and plant storage organs.
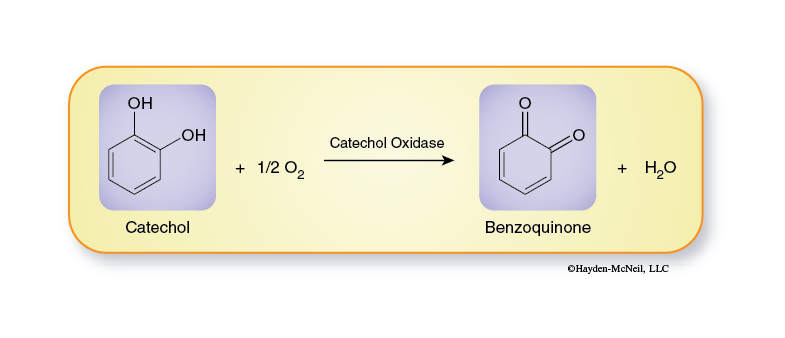
Enzymes typically are given the suffix –ase and are usually named for the reaction they catalyze. Since this reaction is an oxidation reaction, the enzyme is an oxidase and because catechol is the substance being oxidized, a more exact name for the enzyme is catechol oxidase (also commonly called catecholase).
To determine the rate of a chemical reaction it is essential to be able to observe the disappearance of reactant or the appearance of product. In the reaction we will monitor the conversion of catechol, which is colorless when in solution, to benzoquinone, a yellowish-brown substance. This color change makes it easy to follow the progress of this reaction using a spectrophotometer. You may want to review Appendix A for this laboratory exercise. The benzoquinone (the product of the reaction) absorbs light at specific wavelengths.
This absorption of light follows Beer’s law so that when monochromatic light of a specific wavelength passes through the solution containing the absorbing substance, the amount of light absorbed is directly proportional to the concentration of the absorbing substance. In other words, as the concentration of the product in the solution increases, the amount of light absorbed by the solution increases.
In this first enzyme lab you will determine the rate of the reaction by observing the increase in the product over time. This will provide a basic understanding of enzyme kinetics so that you may then design your own experimental protocol to investigate the effect of other factors on the rate of enzyme-catalyzed reactions in the second enzyme lab.
View the following video for a discussion of basic aspects of enzyme reactions.
Pre-Lab Quiz
Proceed to the Pre-Lab Quiz