Chapter 2. Movement of Materials Across the Cell Membrane I
General Purpose
This pre-lab will present some of the concepts related to the process of osmosis.
Learning Objectives
General Purpose
Conceptual
- Define and use the following terms: solvent, solute, diffusion, selectively permeable, osmosis, hypotonic, hypertonic, and isotonic.
- Gain an understanding of some of the factors that influence the movement of materials across cell membranes including the principle of osmosis.
- Develop an appreciation of the osmotic responses and water balance of plant and animal cells.
Background Information
Membranes allow cells and organisms to control their internal chemical environment. Regulating the movement of substances can also result in the control of various metabolic pathways by altering the availability of materials. Biological membranes are typically comprised of phospholipid bilayers (Figure 5-1).
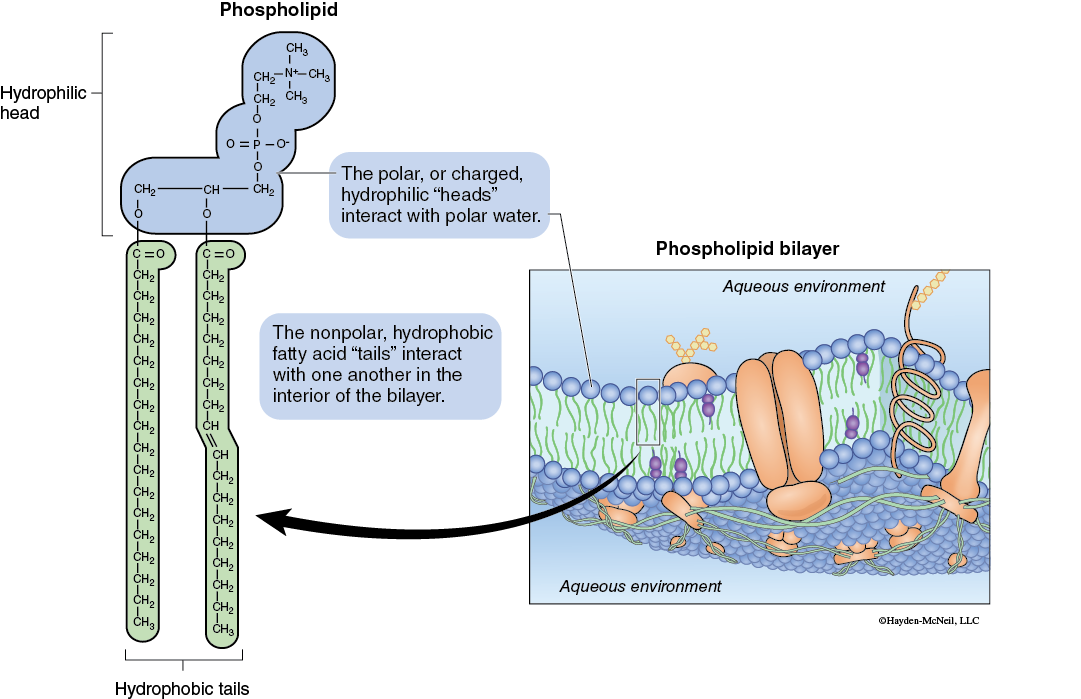
The phospholipid bilayers of biological membranes have protein molecules (both integral and peripheral) associated with their structure. The nature and arrangement of the proteins and lipids produces a membrane that is selectively permeable. This selective permeability results in some materials crossing the membrane while other substances either cannot move across or do so only rarely.
Many kinds of molecules surround or are contained within cells, but water is perhaps the single most important molecule in any living system. Water, although polar, is able to cross through the phospholipid bilayer because of its small size. Water molecules are moving across cell membranes at all times. Osmosis is a situation where more water molecules are moving across the membrane in one direction than the other. This net movement of water across a selectively permeable membrane is caused by differences in the concentration of water across the membrane.
The force causing this net movement of water is a change in the free energy of the water molecules. The higher the water concentration, the more likely that water molecules will interact and hydrogen bond with other water molecules to form highly organized configurations. This increased organization, or decreased disorder, has a higher free energy. When other material is put in solution this material disrupts the pattern of the water molecules thereby increasing the disorder of the water molecules and consequently reducing their free energy. If water molecules can move to sites of lower concentration (e.g., across a membrane), then there will be a decrease in organization or free energy. The change in the free energy of the water molecules is based on the following equation:
where:
ΔG = the change in free energy
ΔH = the change in enthalpy (heat energy)
T = temperature in degrees Kelvin (K)
ΔS = the change in entropy (disorder)
In most osmotic situations the enthalpy of the water molecules does not change and the temperature is relatively constant across the membrane so this equation effectively reduces to:
The presence of the negative sign on the right-hand side of this equation indicates that the change in free energy of the water molecules is inversely proportional to the change in entropy of the water molecules. As water molecules increase in disorder, their free energy decreases. As water molecules decrease in disorder, their free energy increases.
Like all spontaneous processes osmosis involves water molecules moving down their free energy gradient. During osmosis the net movement of water molecules will be from areas of high free energy to areas of low free energy, downhill energetically.
Several other conceptual schemes have been developed to describe and help us understand osmosis. These various schemes are all slightly different, but are basically equivalent to one another. The varying schemes are used in different disciplines. You will most likely encounter more than one of these schemes as you take courses in different disciplines. Keep in mind that they are just different ways of explaining the same phenomenon. Table 5-1 compares several of the most common terms used to describe osmosis.
Table 5-1. Comparison of terms used to describe osmosis.
Osmosis
Osmosis results in a net movement of water from a solution that has a lower osmotic concentration to a solution that has a higher osmotic concentration. The presence of a greater concentration of solutes on one side of a membrane compared with the other establishes a gradient along which water moves. Remember that the movement of water is not affected by what is dissolved in the water but how much is dissolved.
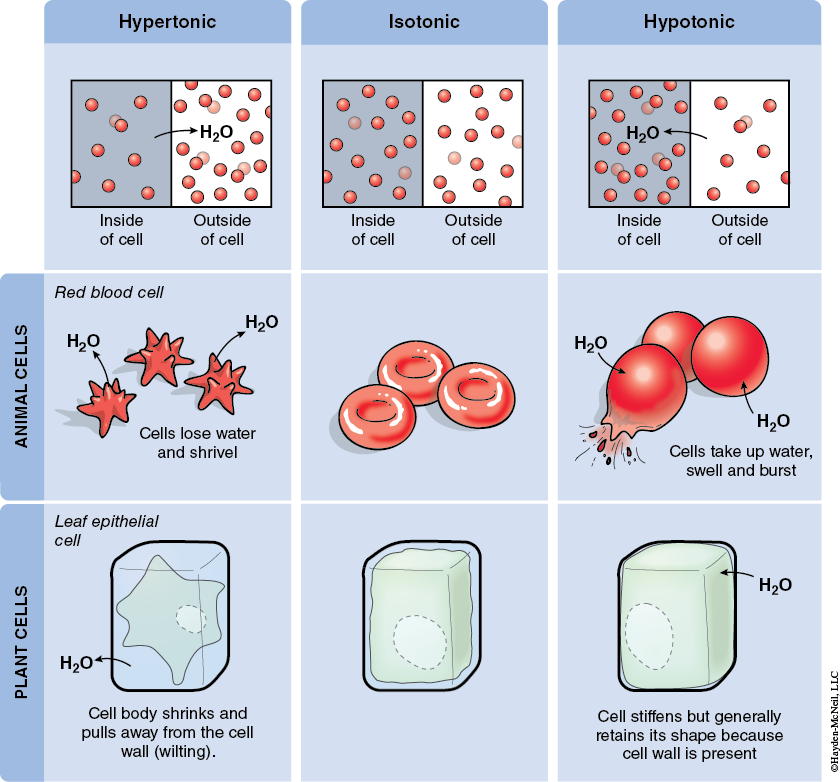
When comparing solutions of different concentrations, the one that has more solute is known as hypertonic, and the one with less solute is known as hypotonic. Solutions that have equal amounts of solute are known as isotonic. Depending on the nature of the cell or organism, solutions of different concentrations will have different cellular effects (Figure 5-2).
Pre-Lab Quiz
Proceed to the Pre-Lab Quiz