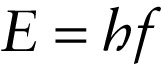Energy of a photon (22-26)
Question 1 of 3
Question
Energy of a photon
{"title":"Energy of a photon","description":"Correct!","type":"correct","color":"#99CCFF","code":"[{\"shape\":\"poly\",\"coords\":\"82,133\"},{\"shape\":\"rect\",\"coords\":\"10,16,12,16\"},{\"shape\":\"poly\",\"coords\":\"144,22\"},{\"shape\":\"rect\",\"coords\":\"1,8,41,55\"}]"} {"title":"Wave frequency","description":"Incorrect","type":"incorrect","color":"#ffff00","code":"[{\"shape\":\"rect\",\"coords\":\"135,4,161,63\"}]"} {"title":"Planck’s constant = 6.62606957 × 10–34 J • s","description":"Incorrect","type":"incorrect","color":"#00ff00","code":"[{\"shape\":\"rect\",\"coords\":\"102,4,133,59\"}]"}Review
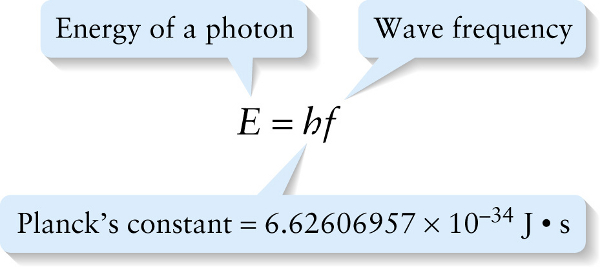
In 1905, the same year that he published his special theory of relativity, Albert Einstein proposed a simple but radical explanation for the strange behavior of the photoelectric effect. He suggested that light of frequency \(f\) comes in small packets, each with an energy \(E\) that is directly proportional to the frequency. Today these packets are called photons. We first encountered this idea in Section 22-4: