12.2 REPLICATION OF CHROMOSOMES
The steps involved in DNA replication are universal, suggesting that they evolved in the common ancestor of all living organisms. In addition, they are the same whether they occur in a test tube or in a cell, or whether the segment of DNA being replicated is short or long. However, the replication of an entire linear chromosome poses particular challenges. Here, we consider two such challenges encountered by cells in replicating their chromosomes: how replication starts, and how it ends.
12.2.1 Replication of DNA in chromosomes starts at many places almost simultaneously.
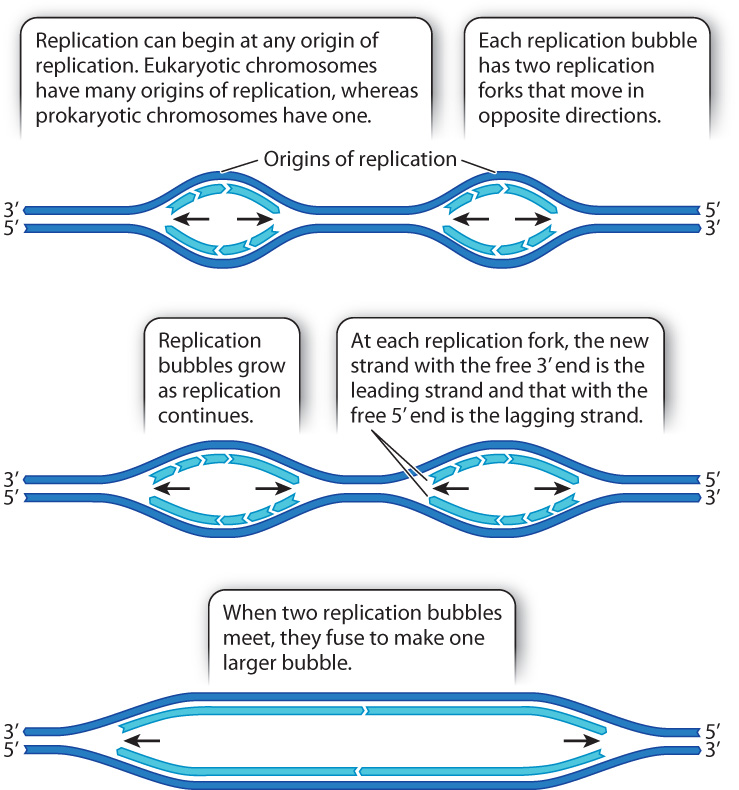
DNA replication is relatively slow. In eukaryotes, it occurs at a rate of about 50 nucleotides per second. At this rate, replication from end to end of the DNA molecule in the largest human chromosome would take almost two months. In fact, it takes only a few hours. This fast pace is possible because, in a long DNA molecule, replication begins almost simultaneously at many places. Each point at which DNA synthesis is initiated is called an origin of replication. The opening of the double helix at each origin of replication forms a replication bubble with a replication fork on each side, each with a leading strand and a lagging strand (Fig. 12.9). DNA synthesis takes place at each replication fork, and as the replication forks move in opposite directions the replication bubble increases in size. When two replication bubbles meet, they fuse to form one larger replication bubble.
Note in Fig. 12.9 that within a single replication bubble, the same daughter strand is the leading strand at one replication fork and the lagging strand at the other replication fork. This situation results from the fact that the replication forks in each replication bubble move away from each other. When two replication bubbles fuse and the leading strand from one meets the lagging strand from the other, the ends of the strands that meet are joined by DNA ligase, just as happens when the discontinuous fragments within the lagging strand meet (see Fig. 12.6).
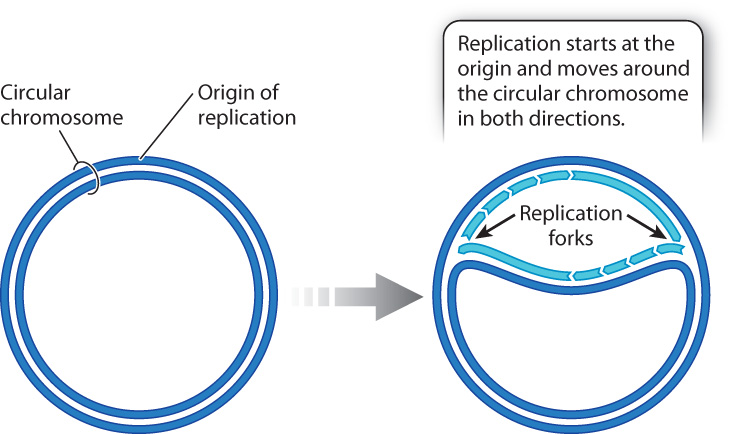
Some DNA molecules, including most of the DNA molecules in bacterial cells and the DNA in mitochondria and chloroplasts (Chapter 3) are small circles, not long linear molecules. Such circular DNA molecules typically have only one origin of replication (Fig. 12.10). Replication takes place at both replication forks, and the replication forks proceed in opposite directions around the circle until they meet and fuse on the opposite side, completing one round of replication.
12.2.2 Telomerase restores tips of linear chromosomes shortened during DNA replication.
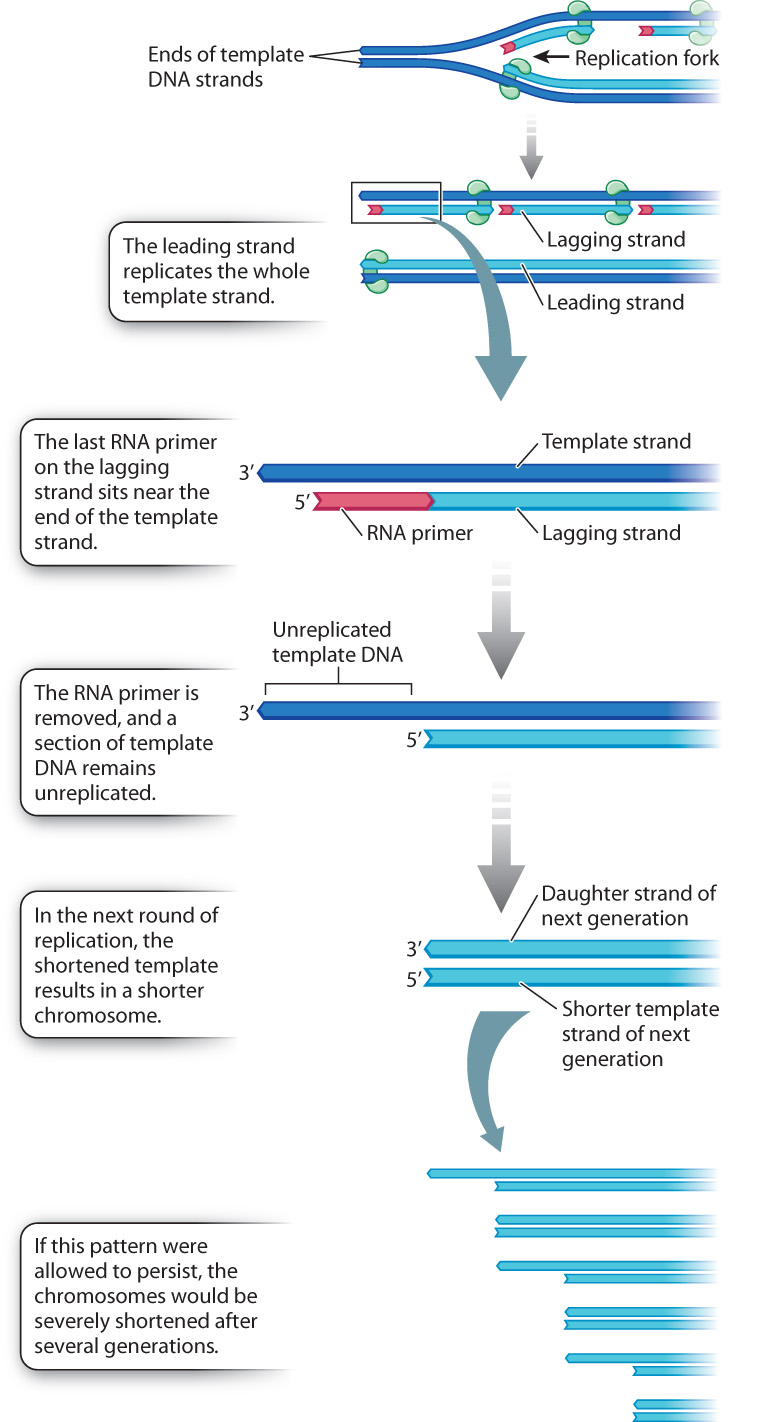
A circular DNA molecule can be replicated completely because it has no ends, and the replication forks can move completely around the circle (Fig. 12.10). Linear DNA molecules have ends, however, and at each round of DNA replication the ends become slightly shorter. The reason for the shortening is illustrated in Fig. 12.11. Recall that each fragment of newly synthesized DNA starts with an RNA primer. On the leading strand, the only primer required is at the origin of replication when synthesis begins. The leading strand is elongated in the same direction as the moving replication fork and is able to replicate the template strand all the way to the end. But on the lagging strand, which grows away from the replication fork, many primers are required, and the last RNA primer is synthesized on the end of the template strand about 100 nucleotides from the 3′ end of the template. When DNA replication is complete, this new daughter DNA strand (light blue in Fig. 12.11) is therefore missing about 100 base pairs from the tip. When the daughter strand is itself replicated, the newly synthesized strand must terminate at the shortened end of the template strand, and so the new duplex molecule is shortened by about 100 base pairs from the original parental molecule. The strand shortening in each round of DNA replication is a problem because without some mechanism to restore the tips, the DNA in the chromosome would eventually be nibbled away to nothing.
12-9
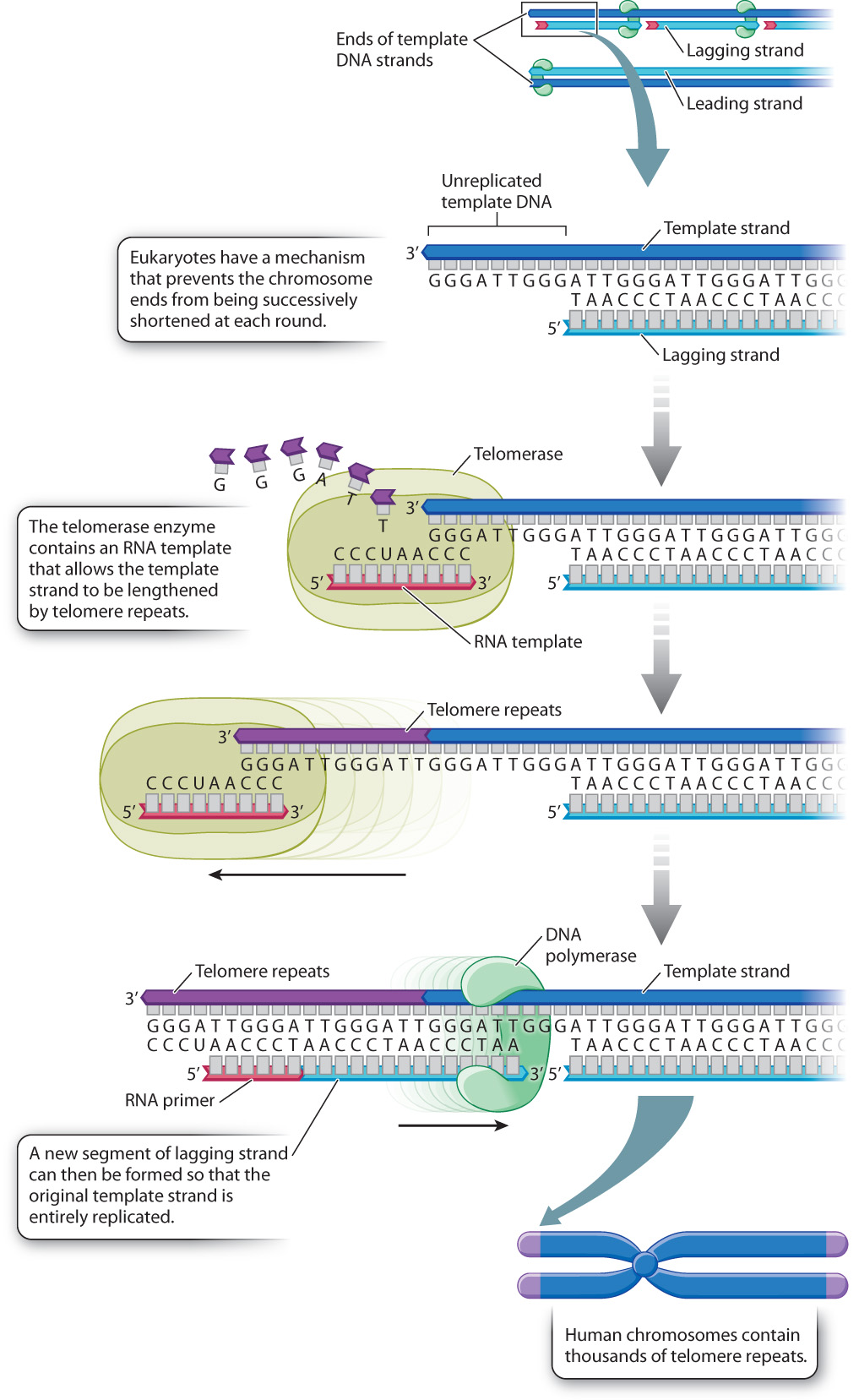
Fig. 12.12 illustrates the mechanism that eukaryotic organisms have evolved to solve the problem of shortened ends. Each end of a eukaryotic chromosome is capped by a repeating sequence called the telomere, shown in purple in Fig. 12.12. In human chromosomes, the telomere consists of the sequence 5′-TTAGGG-3′ repeated over and over again in about 1500–3000 copies. The telomere is slightly shortened in each round of DNA replication, as shown in Fig. 12.11, but before the next round of replication the shortened end is restored by an enzyme known as telomerase, which replaces the lost telomere repeats. Because there are no genes in the telomere, the slight shortening and subsequent restoration that take place have no harmful consequences.
Telomerase activity differs from one cell type to the next. It is fully active in germ cells, which produce sperm or eggs, and also in stem cells, which are undifferentiated cells that can undergo an unlimited number of mitotic divisions and can differentiate into any of a large number of specialized cell types. Stem cells are found in embryos, where they differentiate into all the various cell types. Stem cells are also found in some tissues of the body after embryonic development, where they replenish cells with a high rate of turnover, such as blood and intestinal cells, and play a role in tissue repair.
In contrast to the high activity of telomerase in germ cells and stem cells, telomerase is almost inactive in most cells in the adult body, the somatic cells. In these cells, the telomeres are actually shortened by about 100 base pairs in each mitotic division. Telomere shortening limits the number of mitotic divisions that the cells can undergo because human cells stop dividing when their chromosomes have telomeres with fewer than about 100 copies of the telomere repeat. Adult somatic cells can therefore undergo only about 50 mitotic divisions until the telomeres are so short that the cells stop dividing.
Many biologists believe that the limit on the number of cell divisions explains in part why our tissues become less youthful and wounds heal more slowly with age. The telomere hypothesis of aging is still controversial, but increasing evidence suggests that it is one of several factors that lead to aging. The flip side of the coin is observed in cancer cells, in which telomerase is reactivated and helps support the uncontrolled growth and division of abnormal cells.
12-10