47.6 BIOMES AND DIVERSITY GRADIENTS
Populations are dynamic, continually increasing and decreasing in response to local physical and biological influences. Communities and ecosystems are also dynamic, with species coming and going as a result of disturbance and succession. Nevertheless, the assemblages of species found over broad regions of Earth are both distinctive and stable. Such broad, ecologically uniform areas are called biomes. On land, different biomes are recognized by their characteristic vegetation, which reflects the evolutionary adaptation of form and physiology to climate. Terrestrial biomes range from the low-growing tundra found at high latitudes to tropical rain forests, and from grass-dominated steppes and deserts to coastal estuaries (Fig. 47.17). Biomes occur in the oceans as well as on land, and the largest and least-understood biome of all is the vast expanse of cold dark water in the deep sea.
Fig. 47.17: Some common land biomes










47.6.1 Biomes reflect the interaction of Earth and life.
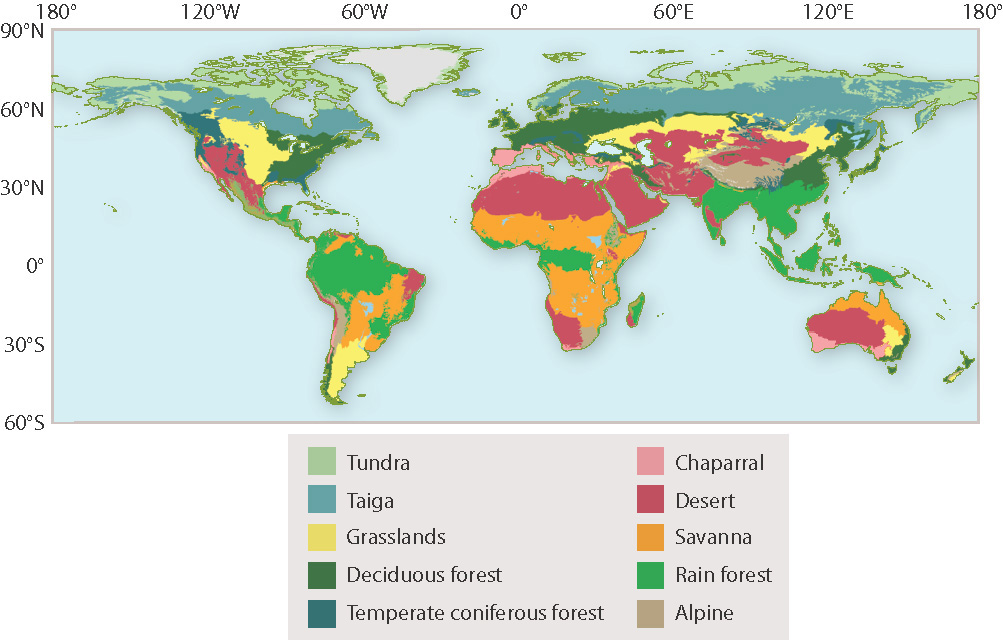
Globally, the distribution of biomes corresponds closely to climate (Fig. 47.18). As noted earlier, this correspondence reflects the need for plants to balance CO2 intake against water loss, a balance commonly struck by means of adaptations in the shape and physiology of leaves, stems, and roots (Chapter 29). To explore this further, we need to discuss a new concept: evapotranspiration ratio.
The intensity of solar radiation varies with latitude and altitude, as does the amount of annual precipitation and wind. These factors determine the amount of evaporation from Earth’s surface to the atmosphere, including the amount transpired by plants. The evapotranspiration ratio is the ratio of the amount of water evaporated from the Earth’s surface directly from ponds, rivers, and soils, to the amount of water vapor transpired by plants (Fig. 47.19).
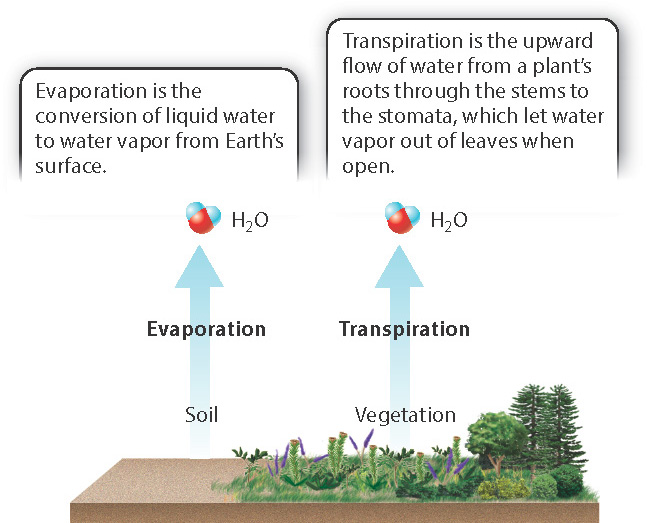
The ratio of evaporation to transpiration effectively tells us what type of vegetation can be supported in a given region. Deserts, with low annual precipitation, have a high evapotranspiration ratio: Soil moisture evaporates rapidly, and plants must keep transpiration (and therefore photosynthetic rate) to a minimum. At the other extreme, rain forests have low evapotranspiration ratios: High precipitation permits nearly unlimited transpiration (and therefore high photosynthetic rates, leading to luxuriant growth), enabling the development of wet forests with high biomass. Intermediate evapotranspiration ratios occur in the temperate zones that encompass much of the United States, Europe, and parts of eastern Asia, as well as at mid-altitudes, below the highest mountain peaks but well above the coastline. Both latitude and altitude influence the evapotranspiration ratio through their effects on temperature and precipitation, and the combined effects of these factors are reflected in the distribution of biomes around the world.
In similar biomes on different continents, dominant plants generally look similar but are not closely related. That is, plants have independently evolved structures by convergent evolution as adaptations to particular climatic regimes. For example, the cacti that grow in deserts in western North America look very similar to plants in drylands of Africa, but in Africa, these plants belong to a different family entirely (Fig. 47.20). Convergent evolution is also seen in rain forest trees that commonly have leaves with long tips that allow excess water to drip away, a reddish color in new leaves that protects them from intense sunlight, and roots with high buttresses that support tall trees atop thin soil.

47.6.2 Tropical biomes usually have more species than temperate biomes.
On a half-hour walk through a temperate forest—for example, in the United States or southern Canada—we can expect to see maybe a dozen different kinds of tree: two or three kinds of oak perhaps, a couple of maple species, birch, beech, hornbeam, ironwood, one or two pine species, and possibly a sycamore, poplar, or spruce, depending on just where we are. Standing in front of a tree and turning around, we can almost always see several more trees of the same kind. In contrast, on a similar walk through a forest in the Amazon, or in Borneo, we would pass 300 or 400 different tree species, with almost no near neighbors of the same kind. The great Victorian naturalist Alfred Russel Wallace took note of the low population density of trees in tropical forests and observed, “If the traveller notices a particular species and wishes to find more like it, he may often turn his eyes in vain in every direction.”
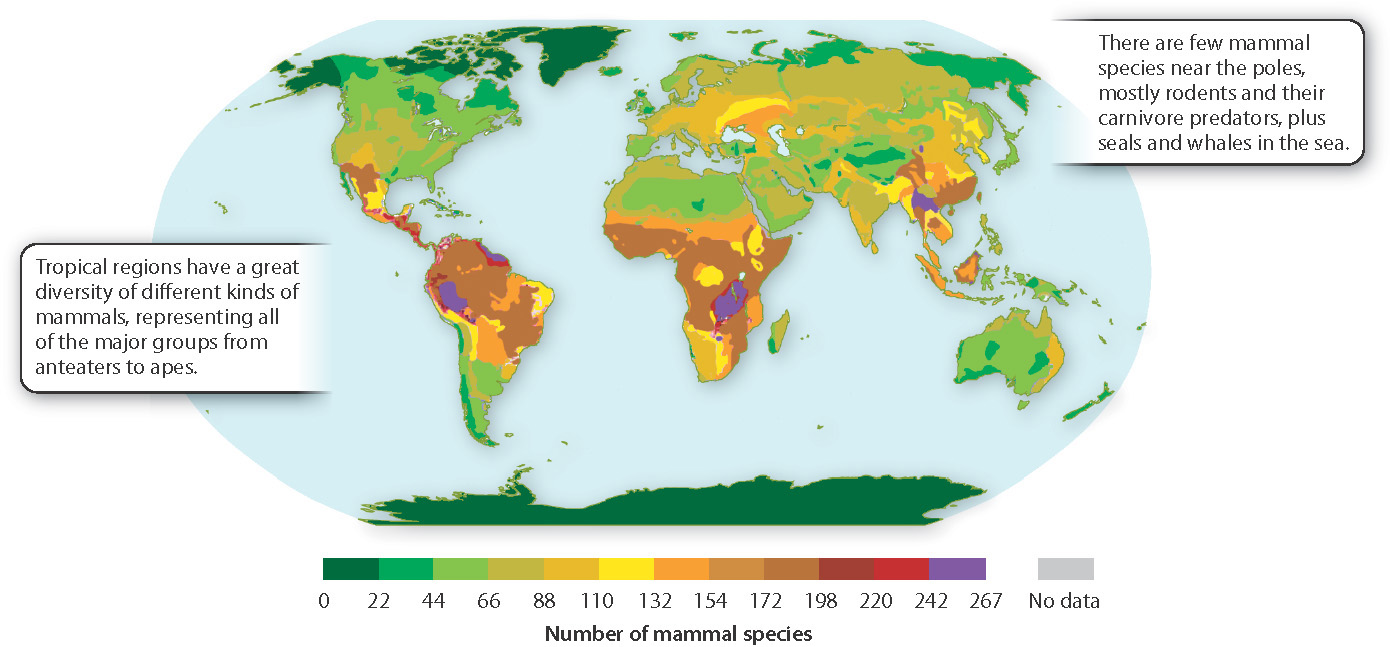
It isn’t just trees. Mammals, birds, insects, and many other kinds of animals show similar patterns of species diversity (Fig. 47.21). One survey found more than 700 species of leaf beetles in one small set of trees in the Amazon, a number equal to the total diversity of leaf beetles in all of eastern North America. A single tree in this same forest harbored 43 species of ants, exceeding the total for all of Great Britain.
When we compare biomes across the continents, the pattern just described stands out clearly: Species diversity generally peaks near the equator and declines toward the poles. What are causes of this latitudinal diversity gradient, and what are its consequences? Does this global pattern reflect the ecological processes that characterize populations?
47.6.3 Why are tropical species so diverse?
Case 8 Biodiversity Hotspots:Rain Forests and Coral Reefs
Theories abound on why the tropics contain so many species. One hypothesis, for which there is broad support, looks to the relative ages of tropical and higher-latitude biomes. Tropical biomes tend to be older, having evolved over tens of millions of years, whereas biomes at higher latitudes have been shaped by climatic change, and ultimately an ice age, within the past few million years. Because tropical habitats have existed much longer than temperate ones, biologists hypothesize that there has been more time for species to evolve and diversify at low latitudes. Several studies of diversification rate, in frogs for example, show comparable rates of diversification in temperate and tropical biomes, supporting the hypothesis that the higher tropical diversity reflects the longer interval through which diversification has taken place.
Time, however, is just one of several factors that influence diversity. Another hypothesis is that temperate communities have fewer species because it is relatively difficult to adapt successfully to cold, dry winters and the range of weather typically experienced at higher latitudes. A tree growing in a temperate area like New England experiences a wide range of temperatures over a year (and sometimes even over a single day) as winds blow and snow falls, or heat waves move through the area. Because species living at high latitudes are adapted to a greater range of conditions on average than those nearer the equator, they tend to have larger geographic ranges than tropical species, which are adapted to the narrow range of environmental variation experienced in any particular place.
It has also been suggested that the many insects and fungi specialized to attack particular kinds of trees are generally more abundant in wet tropical forests, and so trees of a given species tend to be as far apart as they can be, thus reducing the chance of being found by their enemies. This means that there would be more room for different species in any single hectare of land. It has also been noted that there is more land surface area at low latitudes, so the species-area curve (Chapter 46) plays a role in the latitudinal diversity gradient.
Very likely, the latitudinal diversity gradient reflects all these influences and more not yet appreciated. What is clear, however, is that where there are more species of trees, there are also more species of animals and other consumers. Tropical temperatures and high humidity have permitted a wide range of plant species to evolve over the past 100 million years, in turn creating many niches for the animals that depend on them.
The high diversity—and therefore the low population density—of many tropical forests prompts another question. What determines how far apart individual trees can be and still maintain a viable population? One important consideration goes back to mutualisms: Animal pollination ensures that pollen from one tree can reach another individual of the same species if it is within the foraging range of a pollinator carrying its pollen. Of course, if trees are farther apart than any single pollinator can visit within a foraging excursion, they will not be pollinated. Animal pollination, therefore, contributes to the diversity differences between tropical trees and higher-latitude species, which are more likely to be pollinated by wind.
Question Quick Check 5
R/dVg+p5v0CQkoMLMGsrY3xB09WH3O05qnrwL50arH8u5druVrWusfuSc71l6LlyDErmlI5aUOD79ds98am7ma9Y2GNM7B4Quvl2pTT0WDF4dTHzLAX+K//PAv+QOw/OcQuiclM3zz+NE+6KXLeE4rhdFNQS34Z1kgqKC272eBI=47.6.4 Evolutionary and ecological history underpins diversity.
Earth’s biomes are the historical outcome of natural selection and environmental change through time. For this reason, the fossil record can shed light on how current patterns of diversity came to be. In general, fossils only tell us a little about the origins of tropical forests, but on the island of Hispaniola, there is a remarkable exception: nuggets of amber that provide a time capsule of ancient rain forest life. Dominican amber provides an extraordinary window on the diversity of life in the rain forest of Hispaniola 30 to 23 million years ago.
Amber is fossilized tree resin, and in Hispaniola, it comes from the tree Hymenea, a member of the pea family. Hymenea trees grew in the Hispaniola rain forest 30 to 23 million years ago, much as they do today. Lizards, tree frogs, insects, plants, and fungi were all captured in their sticky sap, which hardened into amber, preserving a record of forest diversity and ecology (Fig. 47.22). Studies of these fossils reveal ecological relationships similar to some observed on Hispaniola today. Not surprisingly, some of the most abundant animals in the amber are insects whose modern counterparts have ecological associations with Hymenea. Today, ants commonly patrol the bark of Hymenea and other tropical trees in search of food, and beetles often feed on trees that secrete resin as a defense mechanism, while stingless bees gather resins for their nests. These insects are preserved intact in the amber as if they were just caught. Indeed, many of the insects seen in this amber, perhaps even the majority, are practically indistinguishable from species that continue to exist today on Hispaniola.
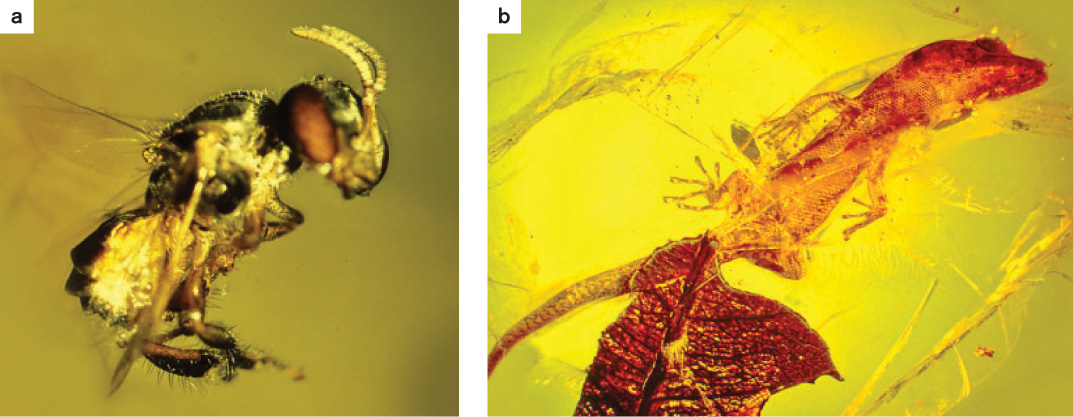
Dominican amber also reveals species that are no longer found on this island. For example, many of the more specialized kinds of ants found in the amber no longer occur on Hispaniola. A kind of beetle that feeds on palm trees in South America is found in amber but not on the island today, even though there are plenty of palms. There are no longer stingless bees on Hispaniola, although the flowers they visit persist, and the bees occur on nearby Jamaica.
Vertebrate fossils are rare in the Dominican amber, but they include Anolis lizards closely related to anoles specialized for life in the canopies and high on the trunks of forest trees—resource partitioning began long ago in the Hispaniola rain forest. More vertebrate fossils occur in other settings, and these show that most of the birds and mammals found 25 million years ago are quite different from those in modern forests.
Such fossils allow us to add a deeper dimension of time to our understanding of how ecosystems have come to be. Whether forest, reef, or grassland, every ecosystem contains the present-day survivors of groups representing a broad range of origins in evolutionary time and geographic space. The amber fossils remind us that what G. Evelyn Hutchinson called “the ecological theater and the evolutionary play” have been intertwined throughout our planet’s history, producing the diversity of species we see today.