Light absorption results in photochemical change
199
When a pigment molecule absorbs light, it enters an excited state. This is an unstable situation, and the molecule rapidly returns to its ground state, releasing most of the absorbed energy. So rapid is this process that it is measured in picoseconds (trillionths of a second). Within the antennae system of a photosystem (Figure 10.6A), the energy released by a pigment molecule (for example, chlorophyll b) is absorbed by other, adjacent pigment molecules. The energy (not as electrons but in the form of chemical energy called resonance) is passed from molecule to molecule until it reaches a chlorophyll a molecule at the reaction center of the photosystem (Figure 10.6B).
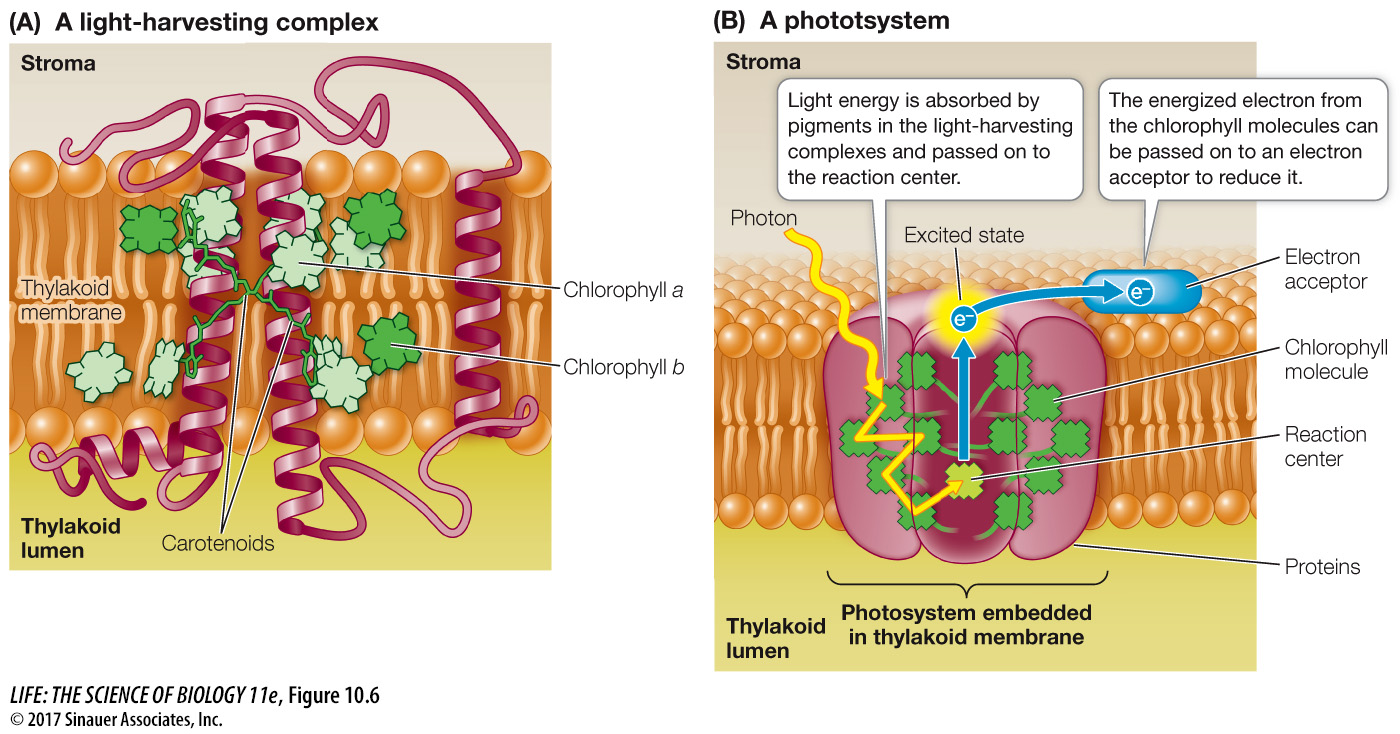
A ground-
Chl* + acceptor → Chl+ + acceptor– (10.6)
This, then, is the first consequence of light absorption by chlorophyll: the reaction center chlorophyll (Chl*) loses its excited electron in a redox reaction and becomes Chl+. As a result of this transfer of an electron, the chlorophyll gets oxidized, while the acceptor molecule is reduced.
Question
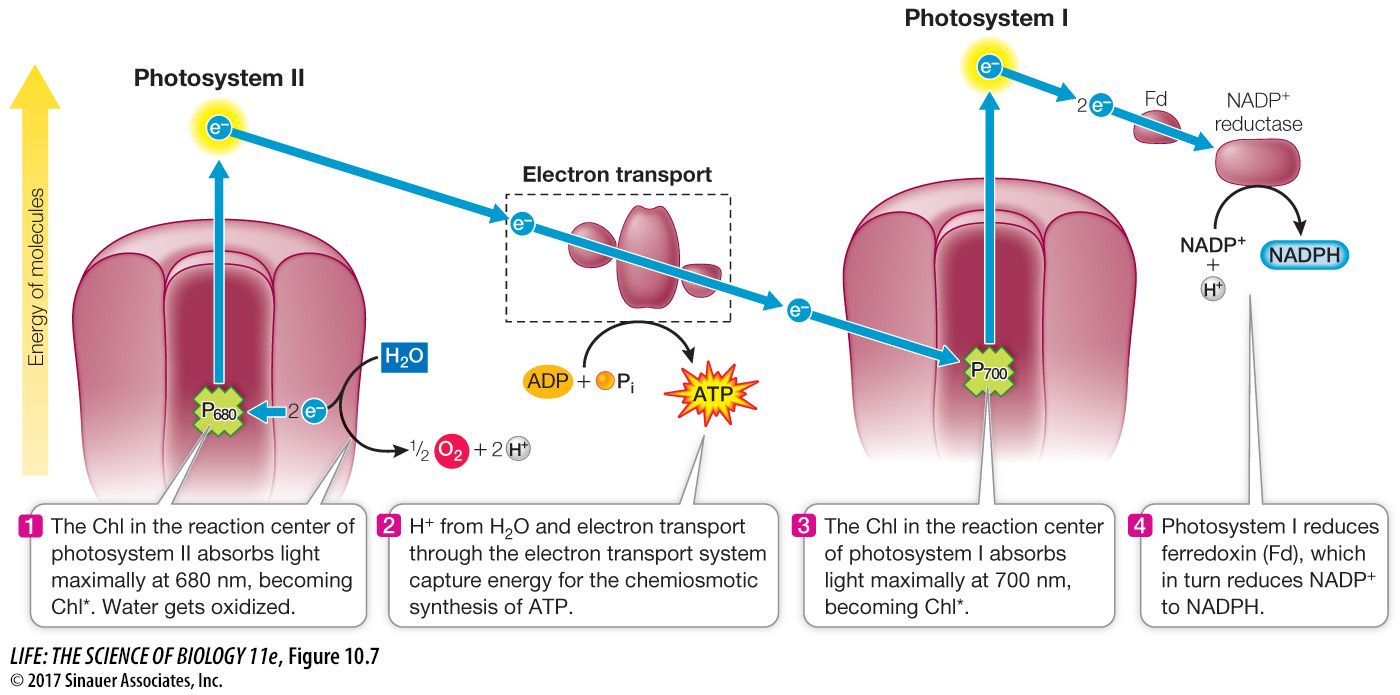
Q: An herbicide can act as an electron acceptor, becoming reduced by ferredoxin (Fd). What effect would this have on a plant?
The herbicide rather than NADP reductase would accept electrons from noncyclic photosystem I. NADPH would not be formed. This would severely reduce the transfer of solar energy to chemical energy in the light-