Actin–myosin interactions are controlled by calcium ions
Like neurons, muscle cells are excitable—that is, their cell membranes generate and conduct action potentials. Action potentials are initiated in skeletal muscle fibers by motor neurons arriving at neuromuscular junctions (see Key Concept 44.3). The axon terminals of motor neurons are generally highly branched and form synapses with hundreds of muscle fibers (Figure 47.4). A motor neuron and all of the fibers with which it synapses constitute a motor unit. The fibers contract simultaneously when the unit’s motor neuron fires. One muscle can consist of many motor units. Thus there are two ways to increase a muscle’s strength of contraction—increase the firing rate of an individual motor neuron, or recruit more motor neurons.

Figure 47.4 The Neuromuscular Junction Axons branching from a single motor neuron end in terminals that innervate multiple skeletal muscle fibers.
Question
Q: Is there a relationship between the number of fibers that a motor neuron innervates and fine motor control, or between the number of fibers innervated and the exertion of force?
If a motor neuron innervates only one or a few fibers, it is likely that those fibers are used in fine movements. If a single motor neuron innervates many muscle fibers of a muscle, it will be able to command strong, forceful movements.
Activity 47.2 The Neuromuscular Junction
When an action potential arrives at a neuromuscular junction, the neurotransmitter acetylcholine is released from the motor neuron terminals, diffuses across the synaptic cleft, binds to receptors in the postsynaptic membrane, and causes ion channels in the motor end plate to open (see Figure 44.11). Most of the ions that flow through these channels are Na+, and therefore the motor end plate is depolarized. The depolarization spreads to the surrounding cell membrane of the muscle fiber, which contains voltage-gated sodium channels. When threshold is reached, the cell membrane fires an action potential that is conducted rapidly to all points on the surface of the muscle fiber.
An action potential in a muscle fiber also travels deep within the cell. The cell membrane is continuous with a system of T tubules that descend into the muscle fiber cytoplasm (also called the sarcoplasm). The “T” stands for “transverse.” The contents of the T tubules are ouside the cell. The T tubules are like the indentations created in a water-filled balloon by poking your finger into it. The action potential that spreads over the cell membrane also spreads through this system of T tubules (Figure 47.5).
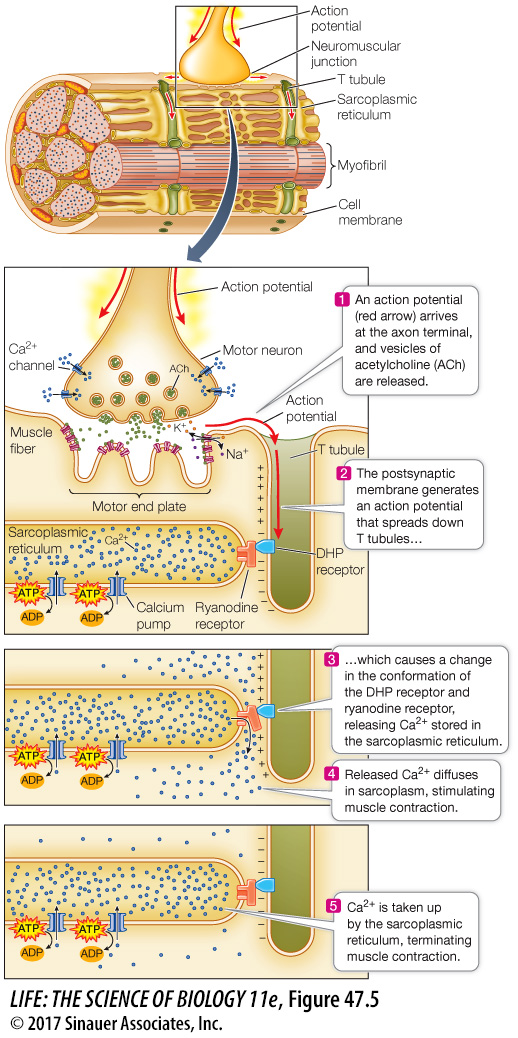
Figure 47.5 T Tubules Spread Action Potentials into the Fiber An action potential at the neuromuscular junction spreads throughout the muscle fiber via a network of T tubules, triggering the release of Ca2+ from the sarcoplasmic reticulum.
The T tubules come very close to the endoplasmic reticulum (ER) of the muscle cell. In muscle cells the ER is called the sarcoplasmic reticulum, and it is a closed compartment surrounding every myofibril. Calcium pumps in the sarcoplasmic reticulum take up Ca2+ from the sarcoplasm. Therefore when the muscle fiber is at rest, there is a high concentration of Ca2+ in the sarcoplasmic reticulum and a very low concentration of Ca2+ in the sarcoplasm.
Spanning the space between the membranes of the T tubules and the membranes of the sarcoplasmic reticulum are two proteins. One protein, the dihydropyridine (DHP) receptor, is located in the T tubule membrane; it is voltage-sensitive and changes its conformation in response to an action potential spreading through the T tubule system. The other protein, the ryanodine receptor, is located in the sarcoplasmic reticulum membrane and is a Ca2+ channel. These two proteins are physically connected. When the DHP receptor changes its conformation due to an action potential, it causes Ca2+ to flow through the ryanodine receptor from the sarcoplasmic reticulum to the sarcoplasm. Ca2+ ions diffusing through the sarcoplasm surrounding the actin and myosin filaments trigger the interaction of actin and myosin and the sliding of the filaments. How do the Ca2+ ions do this?
An actin filament, as you have seen, is a helical arrangement of actin monomers. Twisted around the actin filament are two strands of the protein tropomyosin (Focus: Key Figure 47.6; see also Figure 47.3). At regular intervals, the filament also includes a globular protein, troponin. The troponin molecule has three subunits: one binds actin, one binds tropomyosin, and one binds Ca2+.
focus: key figure
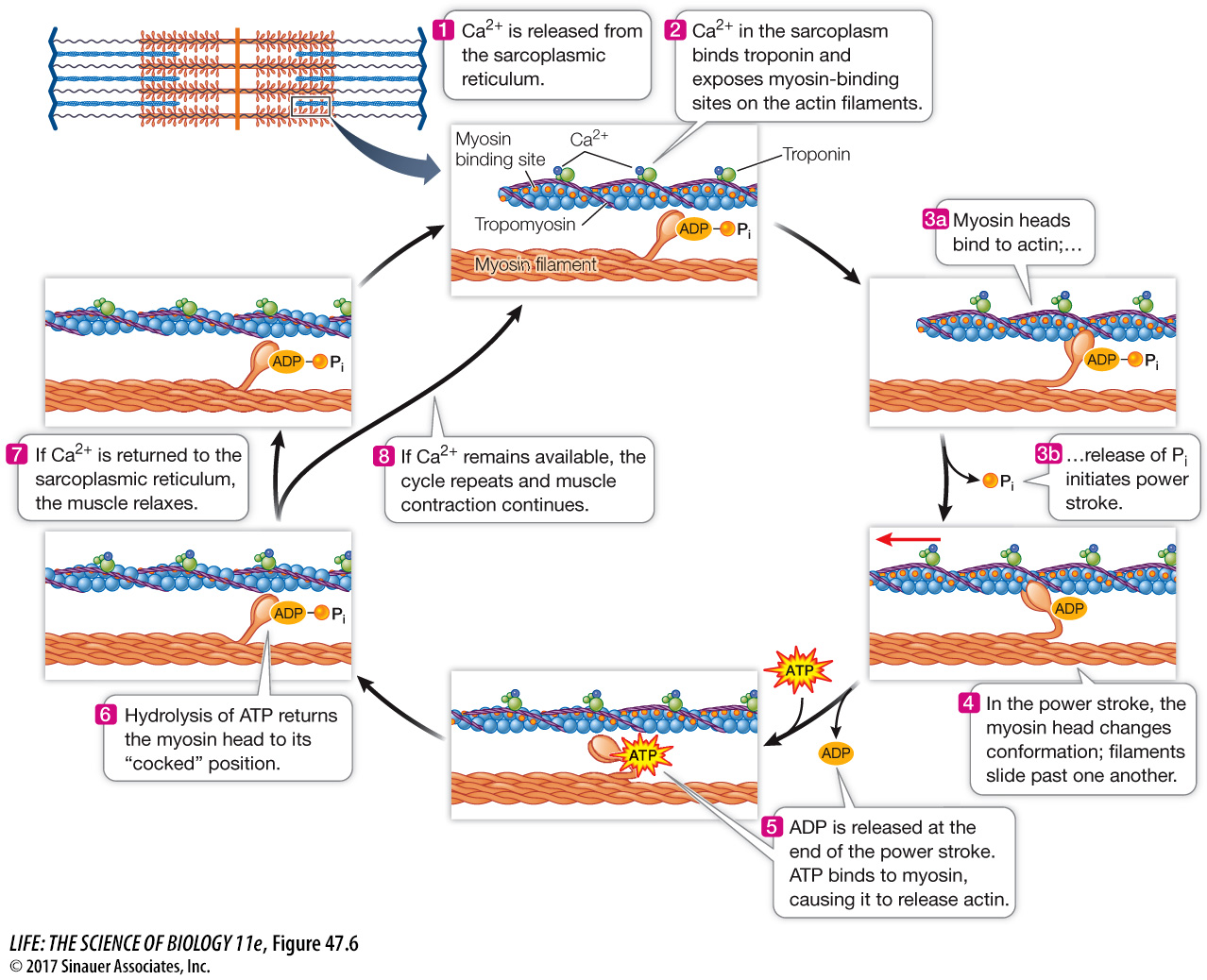
Figure 47.6 Release of Ca2+ from the Sarcoplasmic Reticulum Triggers Muscle Contraction When Ca2+ binds to troponin, it changes the conformation of troponin, exposing myosin-binding sites on the actin. As long as binding sites and ATP are available, the cycle of actin and myosin interactions continues and the filaments slide past each other. Note that in the relaxed condition, the myosin heads are energized.
Question
Q: What are three roles played by ATP in skeletal muscle contraction?
ATP is necessary for skeletal muscle contraction to: (1) establish the ion gradient across the cell membranes, (2) break the actin–myosin bonds and “re-cock” the myosin heads, and (3) pump Ca2+ from the sarcoplasm into the sarcoplasmic reticulum.
When the muscle is at rest, the tropomyosin strands are positioned so that they block the myosin-binding sites on the actin filaments. When Ca2+ is released into the sarcoplasm, it binds to troponin, changing its conformation. Because the troponin is bound to the tropomyosin, this conformational change twists the tropomyosin enough to expose the actin–myosin binding sites, and the cycle of making and breaking actin–myosin bonds is initiated. The filaments pull past each other, and the muscle fiber contracts. When the calcium pumps in the sarcoplasmic reticulum membranes remove the Ca2+ ions from the sarcoplasm, the conformation of the tropomyosin reverts and again blocks the binding of myosin heads to actin, and the muscle fiber returns to its resting condition. Figure 47.6 summarizes this cycle.


