LESSON 8: What Goes Around Comes Around: Conservation of Matter
31
THINK ABOUT IT
The element copper can be mixed with other substances to make a colorful assortment of compounds. What are these compounds? How can you demonstrate that they all contain copper?
What happens to elements in a chemical change?
To answer this question, you will explore
Translating the Copper Cycle
Tracking an Element
Conservation of Matter
Translating the Copper Cycle
EXPLORING THE TOPIC
Translating the Copper Cycle

Chemical names and chemical formulas are powerful tools you can use to keep track of matter. In fact, you can use them to figure out what was made at various steps in the copper cycle.

There are several approaches you can take to figure out what was made at each step of the cycle. First, you can compare the appearance of the compounds that were obtained in each step with compounds that you have seen before. For example, in an earlier class you examined samples of copper compounds that looked like those shown in the two photos here.

Visual observation of compounds can give you some clues, but it is not enough to make a definite identification. For example, it is hard to tell the difference between copper sulfate and copper nitrate through observation because they are both blue. In fact, several copper compounds are blue.
Another approach is to examine the chemical names and formulas of the substances that were mixed together. The new substances are formed from parts of the starting materials. For example, when sodium hydroxide, NaOH, is added to the beaker in the second step of the cycle, a compound called copper hydroxide, Cu(OH)2, is produced. Because a blue solid is formed and hydroxide, OH–, is one of the starting ingredients, you can deduce that copper hydroxide is the product.
ADDING NITRIC ACID TO COPPER
Start at the beginning of the copper cycle. See if you can figure out what was created after the first step by translating each step into chemical symbols and formulas.
32
Step 1: Nitric acid is added to copper powder. A clear blue solution and a brown gas are formed.
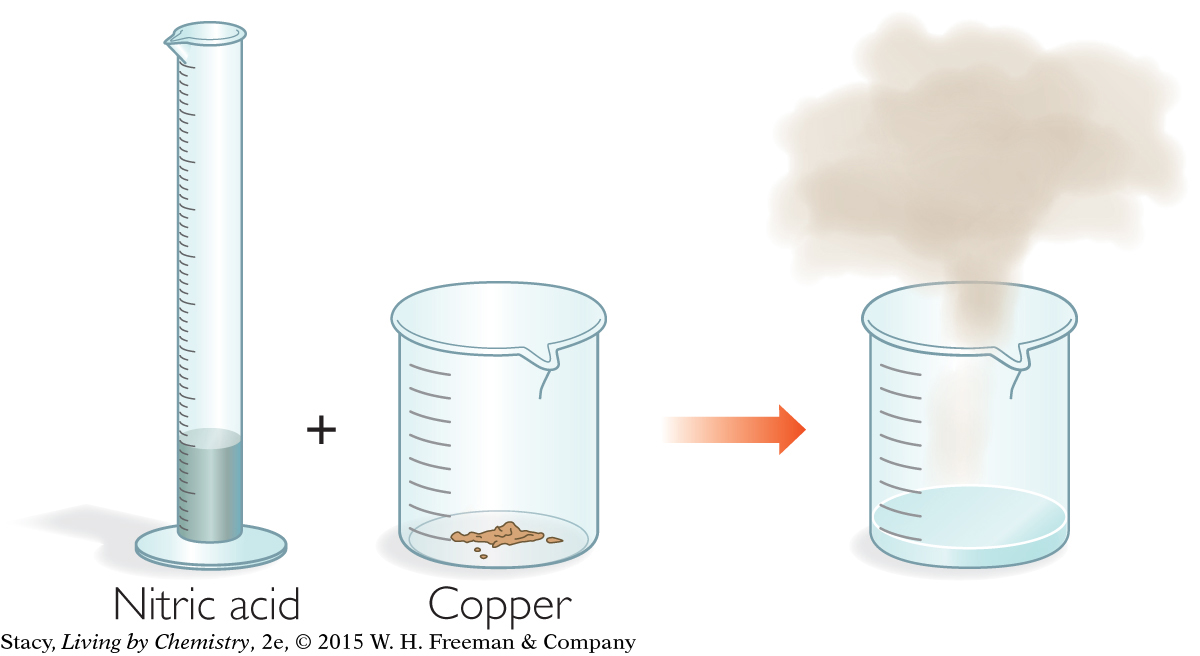
We know the chemical names that go along with the first half of this step. Nitric acid is added to copper powder resulting in a blue solution and a brown gas. Using chemical formulas, you can write this as:

So far in class, you’ve seen two blue liquids. One was copper sulfate and the other was copper nitrate.
Nitric acid, HNO3(aq), was added to the copper powder, so you can deduce that the blue solution that formed was copper nitrate, Cu(NO3)2(aq), and not copper sulfate, CuSO4(aq).
HNO3(aq) is added to Cu(s) resulting in Cu(NO3)2(aq) and ? .
How about the brown gas? The brown gas must contain some combination of H, N, O, or Cu because these are the only starting ingredients. Copper, Cu, does not form gaseous compounds, but the other three elements do combine to form several different gases. The chemical formulas and colors of these gases can be found in reference books and are listed in the table below.

|

Aqueous copper nitrate, Cu(NO3)2(aq)
|
33
ENVIRONMENTAL CONNECTION
ENVIRONMENTAL
CONNECTION
Nitrogen dioxide, NO2(g), is a part of smog. It causes the red-brown color in the skies above cities with large amounts of air pollution. Nitrogen dioxide irritates the eyes, nose, throat, and respiratory tract. Continued exposure can cause bronchitis.

The only brown gas in this table is nitrogen dioxide, NO2(g). The completed chemical sentence for Step 1 is:
HNO3(aq) is added to Cu(s) reulting in Cu(NO3)2(aq) and NO2(g).
This reaction also produces one more compound: water, H2O. You would not have noticed this because it is clear and colorless. So the final chemical sentence for Step 1 is:
HNO3(aq) is added to Cu(s) reulting in Cu(NO3)2(aq) and NO2(g) and H2O(I).
Notice that all the elements in the starting ingredients also appear in the products. No elements are created or destroyed.
You can deduce the products for the other steps in the copper cycle in a similar way.
Tracking an Element
Tracking an Element
Once you have figured out the products of Steps 2, 3, and 4 of the copper cycle, you can track the journey of copper through the cycle. The illustration below shows the copper compounds that form at each step of the cycle.
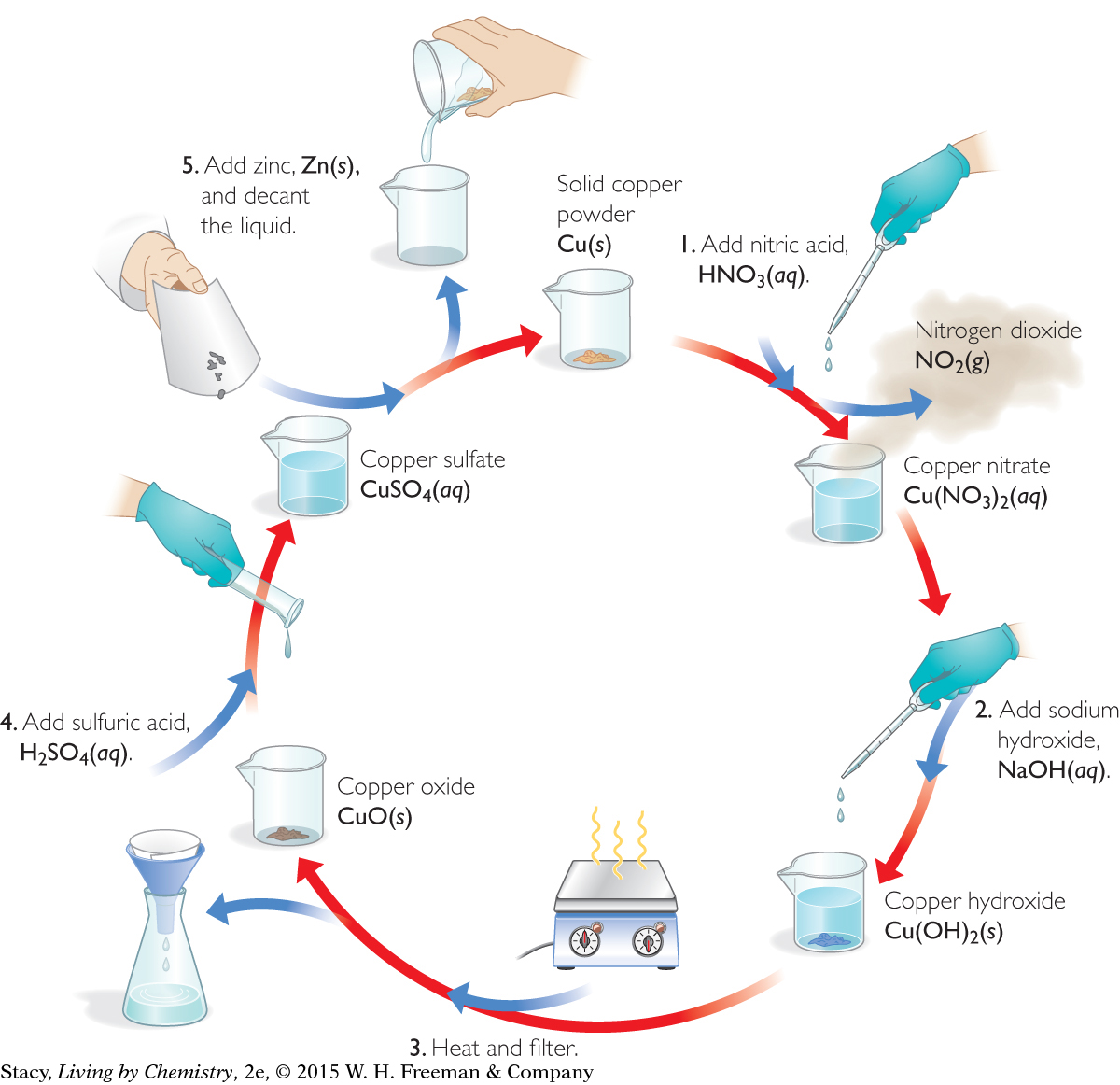
34
The symbol Cu is found at each stage of the cycle. Thus, the element copper is somehow combined in each of these compounds. And of course, the solid that forms at the end of the experiment is elemental copper, Cu(s).
What you have observed with copper is true of other elements as well. For example, nickel can be taken through a similar cycle, where various substances are added to nickel powder, Ni(s). Just as with the copper cycle, you can recover elemental nickel, Ni(s), from compounds that contain nickel.
Conservation of Matter
Conservation of Matter
HISTORY CONNECTION
HISTORY
CONNECTION
Pennies were last made of solid copper in 1836. Pennies made from 1962 to 1982 are 95% copper and 5% zinc. These pennies have a density of 8.6 g/cm3, which is just slightly less than the density of copper, 9.0 g/cm3. Since 1982, pennies have been made mostly of zinc with a copper coating. These pennies have a density of 7.2 g/cm3, which is very close to the density of pure zinc, 7.1 g/cm3.

The copper-cycle experiment brought you full circle, back to where you started. You took a sample of the element copper and added substances to it. After several steps, you ended up with copper powder once again. No matter what was done to the copper, the copper was always there in some form. In other words, it was not created or destroyed by the chemical transformations. Over many centuries, scientists have gathered evidence that matter can never be destroyed or created through chemical transformation. There is so much evidence that this is considered a scientific law.
The law of conservation of mass states that mass cannot be gained or lost in a chemical reaction. In other words, matter cannot be created or destroyed.
It is possible to prove that no copper was gained or lost during the copper-cycle experiment by measuring the mass of the copper powder at the beginning and again at the end. If you did the experiment perfectly, you would end up with exactly the same amount of copper powder that you started with. However, in real life, the mass of the copper at the end of this experiment is a bit less than the mass of the copper at the beginning due to several factors. Little amounts of copper are lost along the way, because of spills, measurement errors, and sticking to the filter paper or beaker. These small errors are hard to avoid. In addition, some copper compounds remain in the discarded solutions. Nevertheless, mass is still conserved; even if the copper isn’t visible in the beaker at the end, it still exists somewhere.
Law of Conservation of Mass
Matter cannot be created or destroyed.
LESSON SUMMARY
LESSON SUMMARY
What happens to elements in a chemical change?
KEY TERM
law of conservation of mass
Chemical names and formulas are used to keep track of elements and compounds as they undergo chemical or physical changes. When elemental copper is tracked through a series of reactions, the symbol Cu shows up in the formulas of the new compounds that are made. Elemental copper can be combined with other substances to form new compounds, but it is not destroyed by the chemical transformations. This concept is known as the law of conservation of mass, which states that matter cannot be created or destroyed.
35
Exercises
Reading Questions
How can chemical names and symbols help you figure out what copper compound was made in each step of the copper cycle? Give an example.
Explain the law conservation of mass in your own words.
Reason and Apply
Lab Report Write a lab report for the Lab: The Copper Cycle. In your report, include the title of the experiment, purpose, procedure, observations, and conclusions.

Explain how the copper-cycle experiment supports the claim that copper is an element—a basic building block of matter.
Nickel sulfate, NiSO4(aq), is a green solution. Nickel chloride, NiCl2(aq), is a yellow solution. And hydrochloric acid, HCl(aq), is a clear, colorless solution. If you add nickel, Ni(s), to hydrochloric acid, HCl(aq), what color solution do you expect to form? Explain your reasoning.
In the final step of the copper cycle, zinc, Zn(s), is added to copper sulfate, CuSO4(aq). Elemental copper appears as a solid. Explain what you think happens to the elemental zinc.
Matter cannot be created or destroyed. List at least two long-term impacts that this concept has for us on this planet.