Aquaporins Increase the Water Permeability of Cellular Membranes
The natural tendency of water to flow across cellular membranes as a result of osmotic pressure raises an obvious question: why don’t the cells of freshwater animals burst in water? Frogs, for example, lay their eggs in pond water (a hypotonic solution), but frog oocytes and eggs do not swell with water even though their internal salt (mainly KCl) concentration is comparable to that of other cells (~150 mM KCl). These observations were what first led investigators to suspect that the plasma membranes of most cell types, but not of frog oocytes, contain water-channel proteins that accelerate the osmotic flow of water. The experimental results shown in Figure 11-7 demonstrate that an aquaporin from the erythrocyte plasma membrane functions as a water channel.

EXPERIMENTAL FIGURE 11-7 Expression of aquaporin by frog oocytes increases their permeability to water. Frog oocytes, which normally are impermeable to water and do not express an aquaporin protein, were microinjected with mRNA encoding aquaporin. These photographs show control oocytes (bottom cell in each panel) and microinjected oocytes (top cell in each panel) at the indicated times after transfer from an isotonic salt solution (0.1 M) to a hypotonic salt solution (0.035 M). The volume of the control oocytes remained unchanged because they are not very permeable to water. In contrast, the microinjected oocytes expressing aquaporin swelled and then burst because of an osmotic influx of water, indicating that aquaporin is a water-channel protein. See L. S. King, D. Kozono, and P. Agre, 2004, Nat. Rev. Mol. Cell Biol. 5:687–698.
[Courtesy of Gregory M. Preston and Peter Agre.]
In its functional form, an aquaporin is a tetramer of identical 28-kDa subunits (Figure 11-8a). Each subunit contains six membrane-spanning α helices that form a central pore through which water can move in either direction, depending on the osmotic gradient (Figure 11-8b, c). The ~2-nm-long water-selective channel, or pore, at the center of each monomer is only 0.28 nm in diameter—only slightly larger than the diameter of a water molecule. The molecular sieving properties of the channel are determined by several conserved hydrophilic amino acid residues whose side-chain and carbonyl groups extend into the middle of the channel and by a relatively hydrophobic wall that lines one side of the channel. Several water molecules can move simultaneously through the channel, each molecule sequentially forming specific hydrogen bonds with the channel-lining amino acids and displacing another water molecule downstream. Aquaporins do not undergo conformational changes during water transport, so they transport water orders of magnitude faster than GLUT1 transports glucose. The formation of hydrogen bonds between the oxygen atom of water and the amino groups of two amino acid side chains ensures that only uncharged water (i.e., H2O, but not H3O+) passes through the channel; the orientations of the water molecules in the channel prevent protons from jumping from one to the next and thus prevent the net movement of protons through the channel. As a consequence, ionic gradients are maintained across membranes even when water is flowing across them through aquaporins.
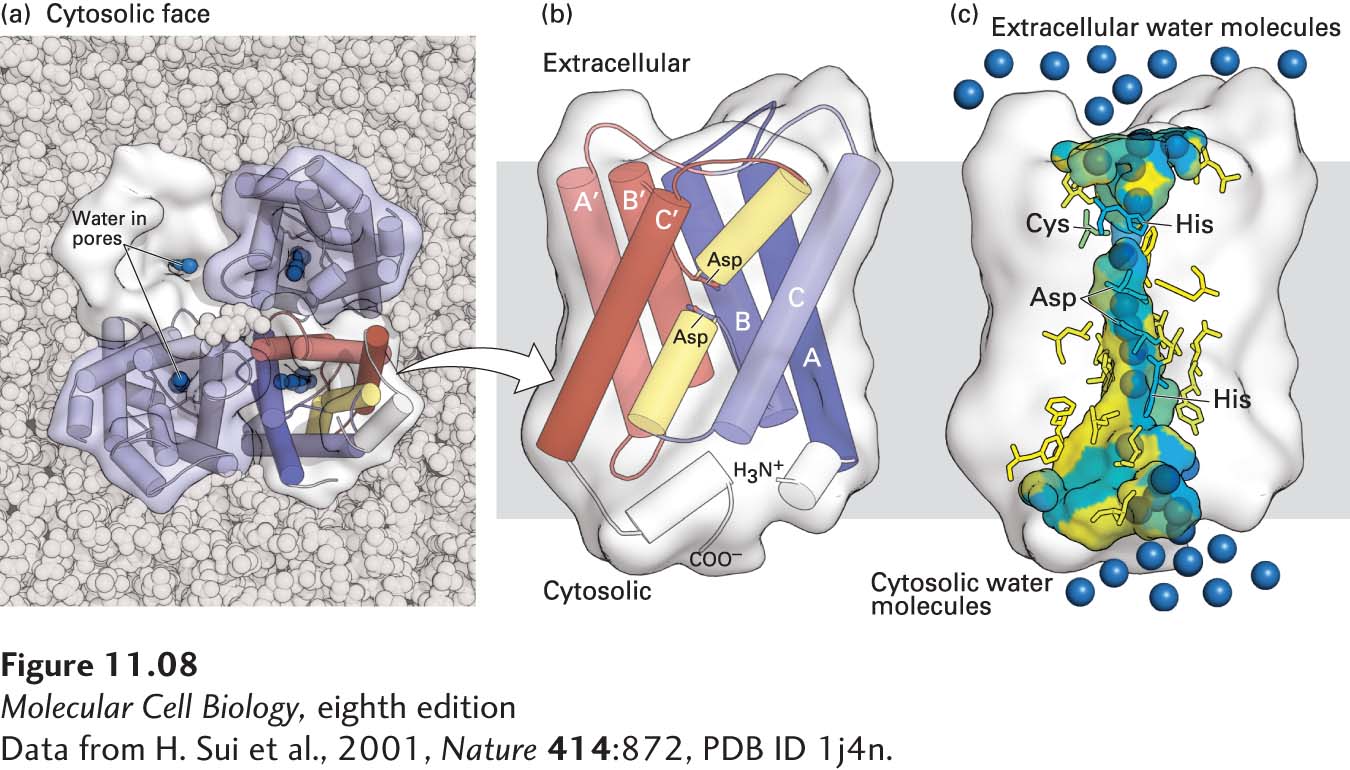
FIGURE 11-8 Structure of an aquaporin. (a) Structural model of the tetrameric protein comprising four identical subunits. Each subunit forms a water channel, as seen in this view looking down on the protein from the exoplasmic side. One of the monomers is shown as a water-accessible surface model, in which the pore entrance can be seen. (b) Schematic diagram of the topology of a single aquaporin subunit in relation to the membrane. Three pairs of homologous transmembrane α helices (A and A′, B and B′, and C and C′) are oriented in the opposite direction with respect to the membrane and are connected by two hydrophilic loops containing short non-membrane-spanning helices and conserved asparagine (N) residues. The loops bend into the cavity formed by the six transmembrane helices, meeting in the middle to form part of the water-selective gate. (c) Side view of the pore in a single aquaporin subunit, in which several water molecules (blue spheres) are seen within the 2-nm-long water-selective gate that separates the water-filled cytosolic and extracellular vestibules. The gate contains highly conserved hydrophilic amino acid residues whose side chains form hydrogen bonds with transported water molecules. The amino acids lining the pore are colored from hydrophilic (blue) to hydrophobic (yellow). The arrangement of these hydrogen bonds and the narrow pore diameter of 0.28 nm prevent passage of protons (i.e., H3O+) or other ions. See T. Zeuthen, 2001, Trends Biochem. Sci. 26:77, and K. Murata et al., 2000, Nature 407:599.
[Data from H. Sui et al., 2001, Nature 414:872, PDB ID 1j4n.]
Mammals express a family of aquaporins; 11 such genes are known in humans. Aquaporin 1 is expressed in abundance in erythrocytes, and the homologous aquaporin 2 is found in the kidney epithelial cells that resorb water from the urine, thus controlling the amount of water in the body. The activity of aquaporin 2 is regulated by vasopressin, also called antidiuretic hormone, in a manner that resembles the regulation of GLUT4 activity in fat and muscle. When the cells are in their resting state and water is being excreted to form urine, aquaporin 2 is sequestered in intracellular vesicle membranes and so is unable to mediate water import into the cell. When the polypeptide hormone vasopressin binds to the cell-surface vasopressin receptor, it activates a signaling pathway using cAMP as the intracellular signal (detailed in Chapter 15) that causes these aquaporin 2–containing vesicles to fuse with the plasma membrane, increasing the rate of water uptake and return to the circulation. Inactivating mutations in either the vasopressin receptor or the aquaporin 2 gene cause diabetes insipidus, a disease marked by excretion of large volumes of dilute urine. This finding demonstrates that the level of aquaporin 2 is rate limiting for water resorption from urine being formed by the kidney.
Other members of the aquaporin family transport hydroxyl-containing molecules such as glycerol rather than water. Human aquaporin 3, for instance, transports glycerol and is similar in amino acid sequence and structure to the Escherichia coli glycerol transport protein GlpF.

