Degradation of an S Phase CDK Inhibitor Triggers DNA Replication
Entry into S phase is defined by the unwinding of origins of DNA replication. The molecular events leading to this unwinding are best understood in S. cerevisiae. G1/S phase CDKs play an essential role in this process. They turn off the machinery that degrades S phase cyclins during exit from mitosis and G1, and they induce the degradation of a CKI that inhibits S phase CDKs.
One of the important substrates of the G1/S phase cyclin-CDK complexes is Cdh1. During late anaphase, this substrate targeting factor directs APC/C to ubiquitinylate substrate proteins, including S phase and mitotic cyclins, marking them for proteolysis by proteasomes. APC/CCdh1 remains active throughout G1, preventing the premature accumulation of S phase and mitotic cyclins. Phosphorylation of Cdh1 by G1/S cyclin-CDKs causes it to dissociate from the APC/C complex, inhibiting further ubiquitinylation of S phase and mitotic cyclins during late G1 (Figure 19-13). This inhibition, combined with the induced transcription of S phase cyclins during late G1, allows S phase cyclins to accumulate as G1/S cyclin-CDK levels rise. Later in the cell cycle, S phase and mitotic CDKs take over to maintain Cdh1 in the phosphorylated, and hence inactive, state. Only as mitotic CDKs decline and a protein phosphatase known as Cdc14 is activated are these inhibitory phosphates removed from Cdh1, leading to its reactivation. In mammalian cells, similar mechanisms are responsible for stabilizing S phase and mitotic cyclins, but the phosphatase(s) involved in dephosphorylation of Cdh1 have not been identified.
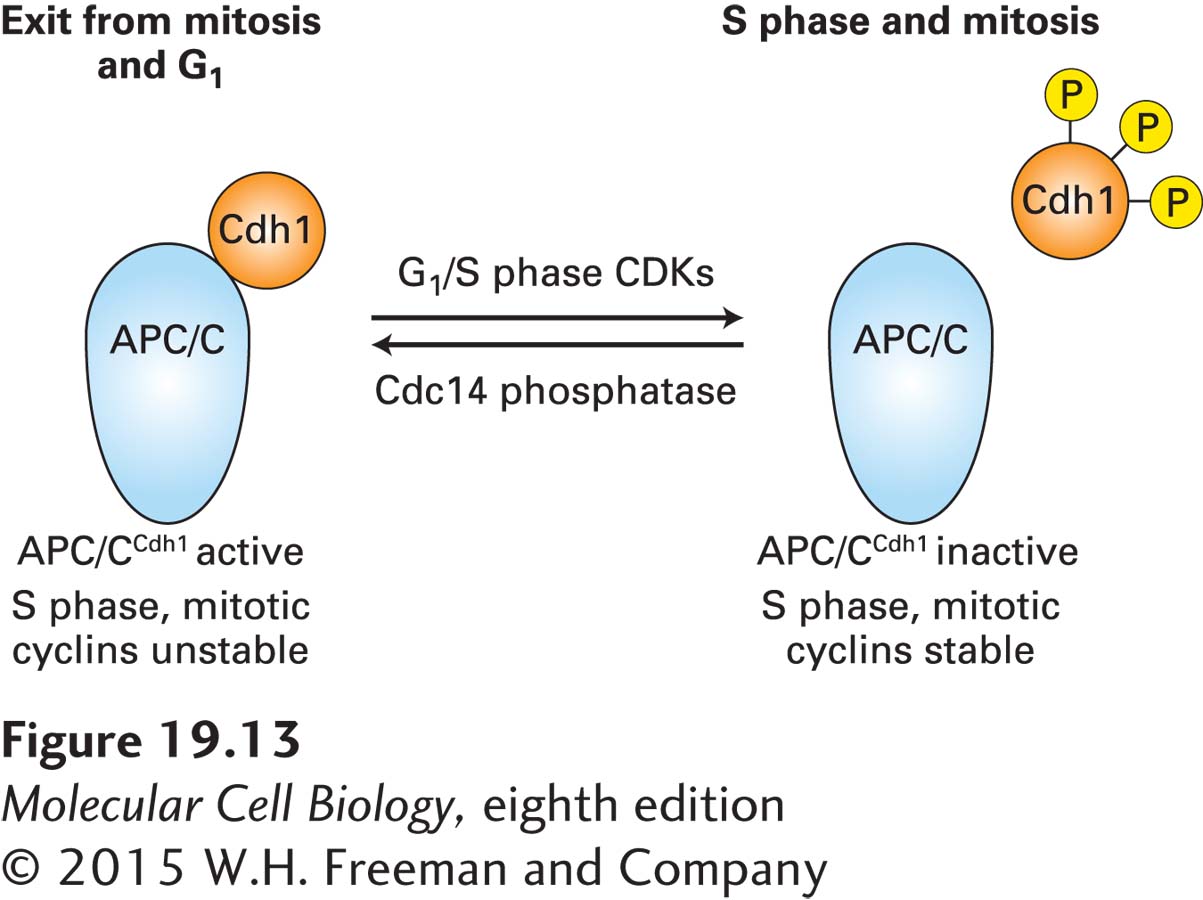
FIGURE 19-13 Regulation of S phase and mitotic cyclin levels in budding yeast. In late anaphase, the anaphase-promoting complex (APC/C) ubiquitinylates S phase and mitotic cyclins. The activity of this ubiquitin-protein ligase is directed toward mitotic cyclins by a specificity factor called Cdh1. Cdh1 activity is regulated by phosphorylation. During exit from mitosis and G1, Cdh1 is dephosphorylated and active; during S phase and mitosis, Cdh1 is phosphorylated and dissociates from APC/C, and APC/C becomes inactive. The G1/S phase CDKs, which themselves are not APC/CCdh1 substrates, phosphorylate Cdh1 at the G1–S phase transition. A specific phosphatase called Cdc14 removes the regulatory phosphate from the specificity factor late in anaphase.
In S. cerevisiae, as S phase cyclin-CDK heterodimers accumulate in late G1 following the inactivation of APC/CCdh1, they are immediately inactivated by binding of a CKI called Sic1 that is expressed late in mitosis and early in G1 (Figure 19-14). Because Sic1 specifically inhibits S phase and M phase CDK complexes, but has no effect on the G1 CDK and G1/S phase CDK complexes, it functions as an S phase inhibitor. Initiation of DNA replication occurs when the Sic1 inhibitor is precipitously degraded following its ubiquitinylation by the SCF ubiquitin-protein ligase.
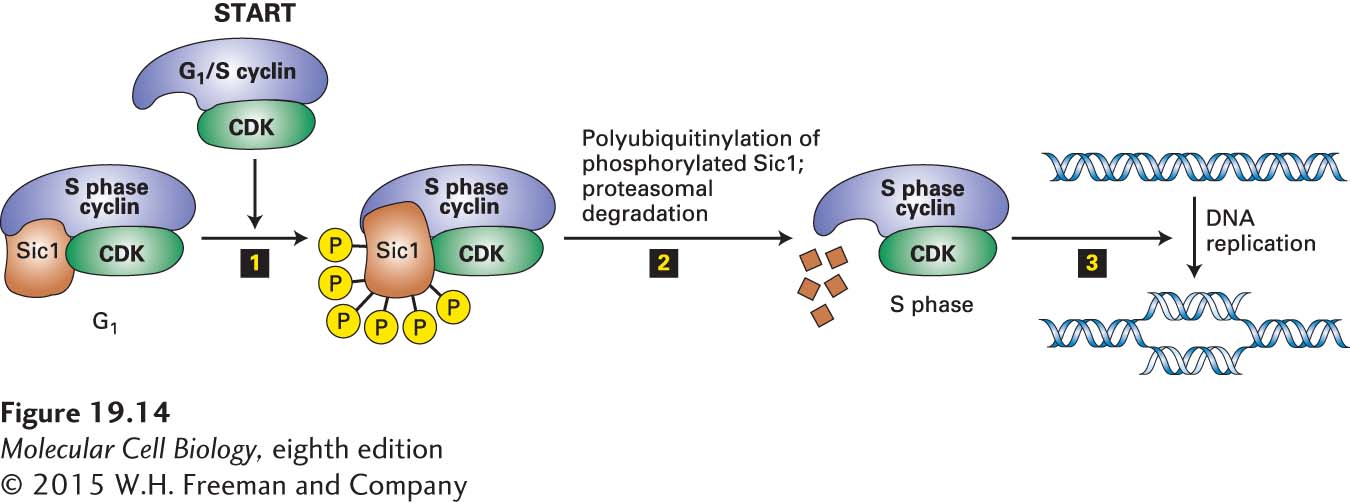
FIGURE 19-14 Control of S phase onset in S. cerevisiae by regulated proteolysis of the S phase inhibitor Sic1. The S phase cyclin-CDK complexes begin to accumulate in G1, but are inhibited by Sic1. This inhibition prevents initiation of DNA replication until the cells have completed all G1 events. G1/S phase CDKs assembled in late G1 phosphorylate Sic1 at multiple sites step 1, marking it for ubiquitinylation by the SCF ubiquitin-protein ligase and subsequent proteasomal degradation step 2. The active S phase CDKs then trigger initiation of DNA synthesis step 3 by phosphorylating and recruiting MCM helicase activators to DNA replication origins. See R. W. King et al., 1996, Science 274:1652.
Degradation of Sic1 is induced by its phosphorylation by G1/S phase CDKs (see Figure 19-14). It must be phosphorylated at no fewer than six sites, which are relatively poor substrates for the G1/S phase CDKs, before it is bound sufficiently well by SCF to be ubiquitinylated. These multiple, poor G1/S phase CDK phosphorylation sites lead to an ultrasensitive, switch-like response in Sic1 degradation and hence precipitous activation of S phase CDKs (Figure 19-15). If Sic1 were inactivated following the phosphorylation of a single site, Sic1 molecules would start to get phosphorylated as soon as the levels of G1/S phase CDK activity begin to rise, leading to a gradual decrease in Sic1 levels. In contrast, because several sites need to be phosphorylated, at low levels of G1/S phase CDK activity only a few sites are phosphorylated and Sic1 is not destroyed. Only when G1/S phase CDK levels are high is Sic1 sufficiently phosphorylated at multiple sites to target it for degradation. Thus Sic1 degradation occurs only when G1/S phase CDK activity has reached its peak and virtually all other G1/S phase CDK substrates have been phosphorylated.
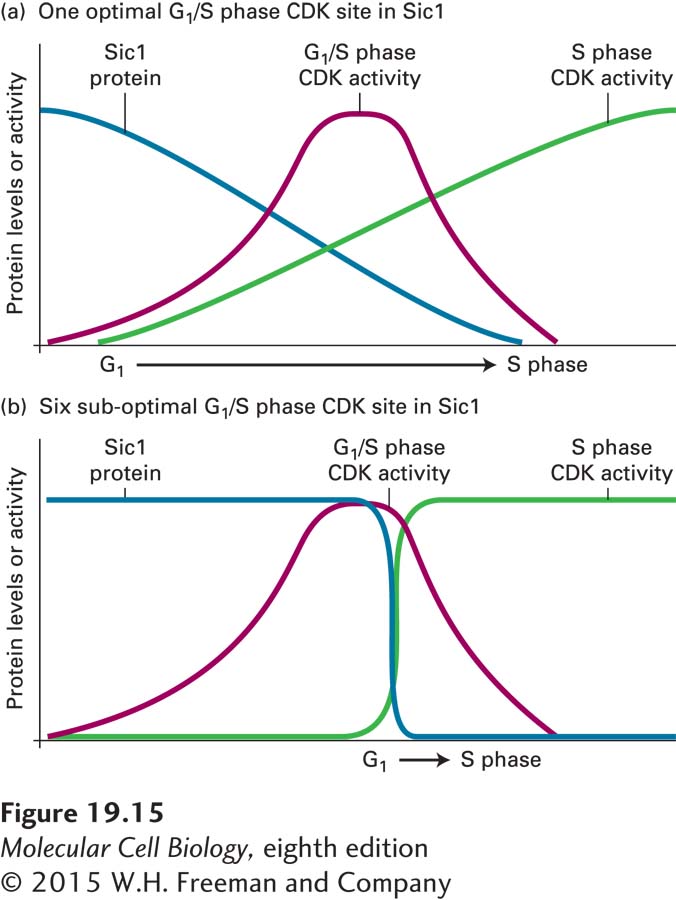
FIGURE 19-15 Six suboptimal phosphorylation sites in Sic1 create a switch-like cell cycle entry. (a) A single optimal G1/S phase CDK phosphorylation site in Sic1 would result in a sluggish G1–S phase transition. As G1/S phase CDKs accumulate during G1, Sic1 would be progressively degraded. As a result, S phase CDKs would slowly rise, and initiation of S phase would be a drawn-out event. (b) Six suboptimal phosphorylation sites ensure that Sic1 is fully phosphorylated, and hence recognized by SCF, only when G1/S phase CDKs have reached high levels. This also ensures that Sic1 degradation occurs rapidly and when G1/S phase CDKs have accomplished all their other G1 tasks. See P. Nash et al., 2001, Nature 414:514–521, and D. O. Morgan, 2006.
Once Sic1 is degraded, the S phase cyclin-CDK complexes induce DNA replication by, as we will see shortly, phosphorylating several proteins involved in activating the replicative helicases. This mechanism for activating the S phase cyclin-CDK complexes—that is, inhibiting them as the cyclins are synthesized and then precipitously degrading the inhibitor—permits the sudden initiation of replication at large numbers of replication origins. An obvious advantage of proteolysis for controlling passage through this critical point in the cell cycle is that protein degradation is an irreversible process, ensuring that cells proceed in one direction through the cycle.
Entry into S phase in metazoan cells is regulated by a mechanism similar to that in budding yeast. Like Sic1, the CKI p27 prevents the premature activation of S phase CDKs during G1. Unlike Sic1, however, this CKI inhibits both S phase CDKs and G1/S phase CDKs and has additional cell cycle functions as well. For example, while inhibiting G1/S phase CDKs and S phase CDKs, p27 helps assemble and hence activate G1 CDKs. Like Sic1 in yeast, however, p27 is removed from cyclin-CDK complexes by ubiquitin-dependent protein degradation. Two pathways contribute to its degradation. Upon stimulation by mitogens, mitogen-activated protein kinases phosphorylate p27, promoting its export from the nucleus into the cytoplasm, where one of the cellular ubiquitin-protein ligases, KPC, is found. A second pathway, analogous to the one operating on Sic1, targets p27 for degradation at the G1-S phase transition. As G1/S phase CDKs and S phase CDKs reach high levels during late G1 and early S phase, they begin to phosphorylate p27, targeting it for ubiquitinylation by SCF. Degradation of p27 causes activation of G1/S phase and S phase CDKs. These kinases then initiate S phase by phosphorylating proteins important for the initiation of DNA replication.


