The Electronic Structure of an Atom Determines the Number and Geometry of the Covalent Bonds It Can Make
Hydrogen, oxygen, carbon, nitrogen, phosphorus, and sulfur are the most abundant elements in biological molecules. These atoms, which rarely exist as isolated entities, readily form covalent bonds, using electrons in the outermost electron orbitals surrounding their nuclei (Figure 2-2). As a rule, each type of atom forms a characteristic number of covalent bonds with other atoms. These bonds have a well-defined geometry determined by the atom’s size and by both the distribution of electrons around the nucleus and the number of electrons that it can share. In some cases, the number of stable covalent bonds an atom can make is fixed; carbon, for example, always forms four covalent bonds. In other cases, different numbers of stable covalent bonds are possible; for example, sulfur can form two, four, or six stable covalent bonds.
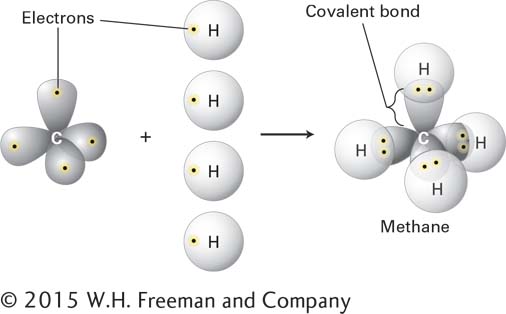
FIGURE 2-2 Covalent bonds form by the sharing of electrons. Covalent bonds, the strong forces that hold atoms together in molecules, form when atoms share electrons from their outermost electron orbitals. Each atom forms a defined number and geometry of covalent bonds.
All the biological building blocks are organized around the carbon atom, which forms four covalent bonds. In these organic biomolecules, each carbon usually bonds to three or four other atoms. [Carbon can also bond to two other atoms, as in the linear molecule carbon dioxide, CO2, which has two carbon-oxygen double bonds (O=C=O); however, such bond arrangements of carbon are not found in biological building blocks.] As illustrated in Figure 2-3a for formaldehyde, carbon can bond to three atoms, all in a common plane. The carbon atom forms two single bonds with two atoms and one double bond (two shared electron pairs) with the third atom. In the absence of other constraints, atoms joined by a single bond generally can rotate freely about the bond axis, whereas those connected by a double bond cannot. The rigid planarity imposed by double bonds has enormous significance for the shapes and flexibility of biomolecules such as phospholipids, proteins, and nucleic acids.
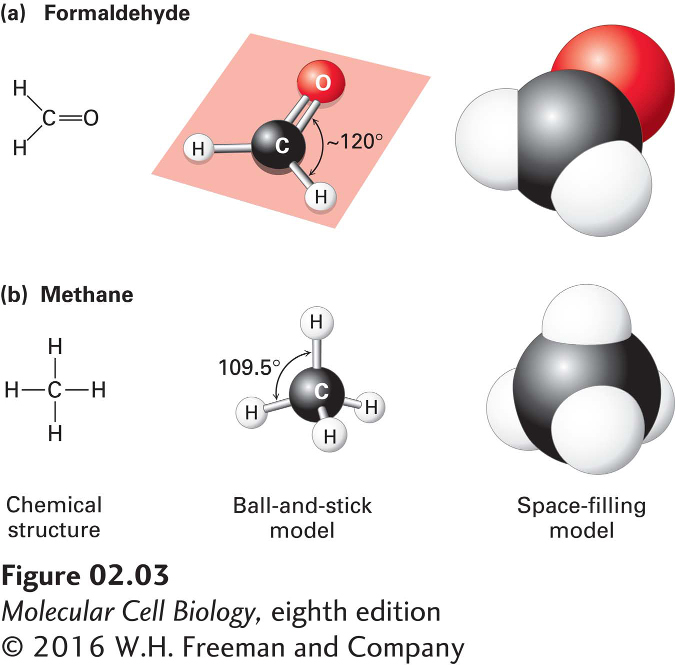
FIGURE 2-3 Geometry of bonds when carbon is covalently linked to three or four other atoms. (a) A carbon atom can be bonded to three atoms, as in formaldehyde (CH2O). In this case, the carbon-bonding electrons participate in two single bonds and one double bond, which all lie in the same plane. Unlike atoms connected by a single bond, which usually can rotate freely about the bond axis, those connected by a double bond cannot. (b) When a carbon atom forms four single bonds, as in methane (CH4), the bonded atoms (all H in this case) are oriented in space in the form of a tetrahedron. The letter representations on the left clearly indicate the atomic composition of each molecule and its bonding pattern. The ball-and-stick models in the center illustrate the geometric arrangement of the atoms and bonds, but the diameters of the balls representing the atoms and their nonbonding electrons are unrealistically small compared with the bond lengths. The sizes of the electron clouds in the space-filling models on the right more accurately represent the structure in three dimensions.
Carbon can also bond to four rather than three atoms. As illustrated by methane (CH4), when carbon is bonded to four other atoms, the angle between any two bonds is 109.5°, and the positions of bonded atoms define the four points of a tetrahedron (Figure 2-3b). This geometry defines the structures of many biomolecules. A carbon (or any other) atom bonded to four dissimilar atoms or groups in a nonplanar configuration is said to be asymmetric. The tetrahedral orientation of bonds formed by an asymmetric carbon atom can be arranged in three-dimensional space in two different ways, producing molecules that are mirror images of each other, a property called chirality (“handedness,” from the Greek word cheir, meaning “hand”) (Figure 2-4). Such molecules are called optical isomers, or stereoisomers. Many molecules in cells contain at least one asymmetric carbon atom, often called a chiral carbon atom. The different stereoisomers of a molecule usually have completely different biological activities because the arrangement of atoms within their structures, and thus their ability to interact with other molecules, differs.
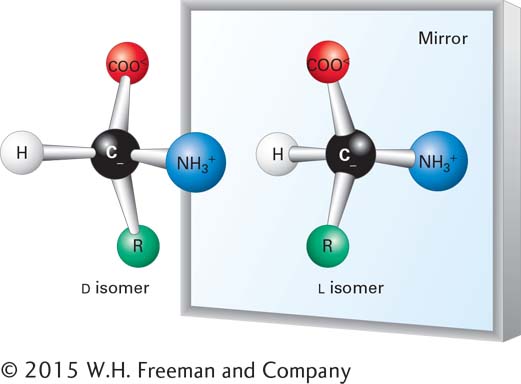
FIGURE 2-4 Stereoisomers. Many molecules in cells contain at least one asymmetric carbon atom. The tetrahedral orientation of bonds formed by an asymmetric carbon atom can be arranged in three-dimensional space in two different ways, producing molecules that are mirror images, or stereoisomers, of each other. Shown here is the common structure of an amino acid, with its central asymmetric carbon and four attached groups, including the R group, discussed in Section 2.2. Amino acids can exist in two mirror-image forms, designated L and D. Although the chemical properties of such stereoisomers are identical, their biological activities are distinct. Only L amino acids are found in proteins.
Some drugs are mixtures of the stereoisomers of small molecules in which only one stereoisomer has the biological activity of interest. The use of a pure single stereoisomer of the chemical in place of the mixture may result in a more potent drug with reduced side effects. For example, one stereoisomer of the antidepressant drug citalopram (Celexa) is 170 times more potent than the other. Some stereoisomers have very different activities. Darvon is a pain reliever, whereas its stereoisomer, Novrad (Darvon spelled backward), is a cough suppressant. One stereoisomer of ketamine is an anesthetic, whereas the other causes hallucinations.
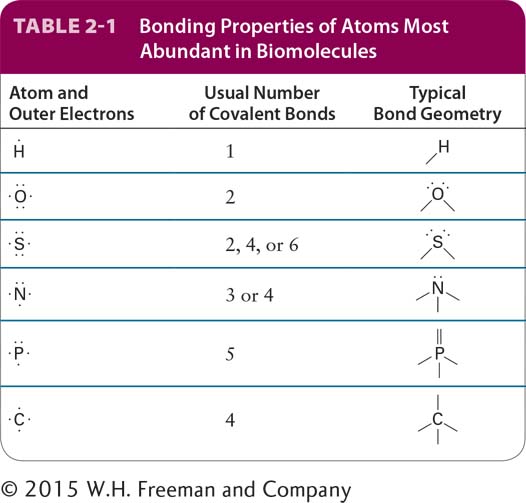
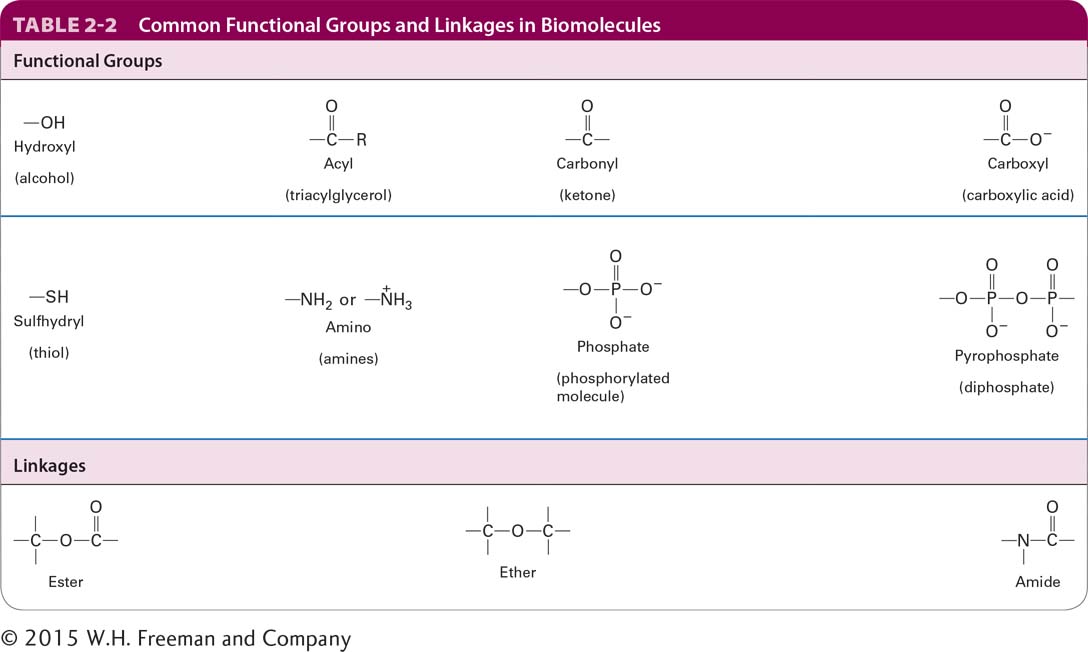
The typical numbers of covalent bonds formed by other atoms common in biomolecules are shown in Table 2-1. A hydrogen atom forms only one covalent bond. An atom of oxygen usually forms only two covalent bonds but has two additional pairs of electrons that can participate in noncovalent interactions. Sulfur forms two covalent bonds in hydrogen sulfide (H2S) but can accommodate six covalent bonds, as in sulfuric acid (H2SO4) and its sulfate derivatives. Nitrogen and phosphorus each have five electrons to share. In ammonia (NH3), the nitrogen atom forms three covalent bonds; the pair of electrons around the atom not involved in a covalent bond can take part in noncovalent interactions. In the ammonium ion (NH4+), nitrogen forms four covalent bonds, which have a tetrahedral geometry. Phosphorus commonly forms five covalent bonds, as in phosphoric acid (H3PO4) and its phosphate derivatives, which form the backbone of nucleic acids. Phosphate groups covalently attached to proteins play a key role in regulating the activity of many proteins, and the central molecule in cellular energetics, ATP, contains three phosphate groups (see Section 2.4). A summary of common covalent linkages and functional groups, which confer distinctive chemical properties on the molecules of which they are a part, is provided in Table 2-2.