Tertiary Structure Is the Overall Folding of a Polypeptide Chain
Tertiary structure refers to the overall conformation of a polypeptide chain—that is, the three-dimensional arrangement of all its amino acid residues. In contrast to secondary structures, which are stabilized only by hydrogen bonds, tertiary structure is stabilized primarily by hydrophobic interactions between nonpolar side chains, together with hydrogen bonds involving polar side chains and backbone amino and carboxyl groups. These stabilizing forces hold together elements of secondary structure—α helices, β strands, turns, and coils. Because the stabilizing interactions are often weak, however, the tertiary structure of a protein is not rigidly fixed, but undergoes continual minute fluctuations, and some segments within the tertiary structure of a protein can be so mobile that they are considered to be disordered—that is, lacking well-defined, stable, three-dimensional structure. This variation in structure has important consequences for the function and regulation of proteins.
The chemical properties of amino acid side chains help define tertiary structure. In some proteins—for example, those that are secreted from cells or are cell-surface proteins that face the extracellular environment—disulfide bonds between the side chains of cysteine residues can covalently link regions of the proteins, thus restricting the proteins’ flexibility and increasing the stability of their tertiary structures. Amino acids with charged hydrophilic polar side chains tend to be on the outer surfaces of proteins; by interacting with water, they help to make the proteins soluble in aqueous solutions and can form noncovalent interactions with other water-soluble molecules, including other proteins. In contrast, amino acids with hydrophobic nonpolar side chains are usually sequestered away from the water-facing surfaces of a protein, in many cases forming a water-insoluble central core. This observation led to what’s known as the “oil drop model” of protein conformation because the core of a protein is relatively hydrophobic, or “oily” (Figure 3-7). Uncharged hydrophilic polar side chains are found both on the surface and in the inner core of proteins.
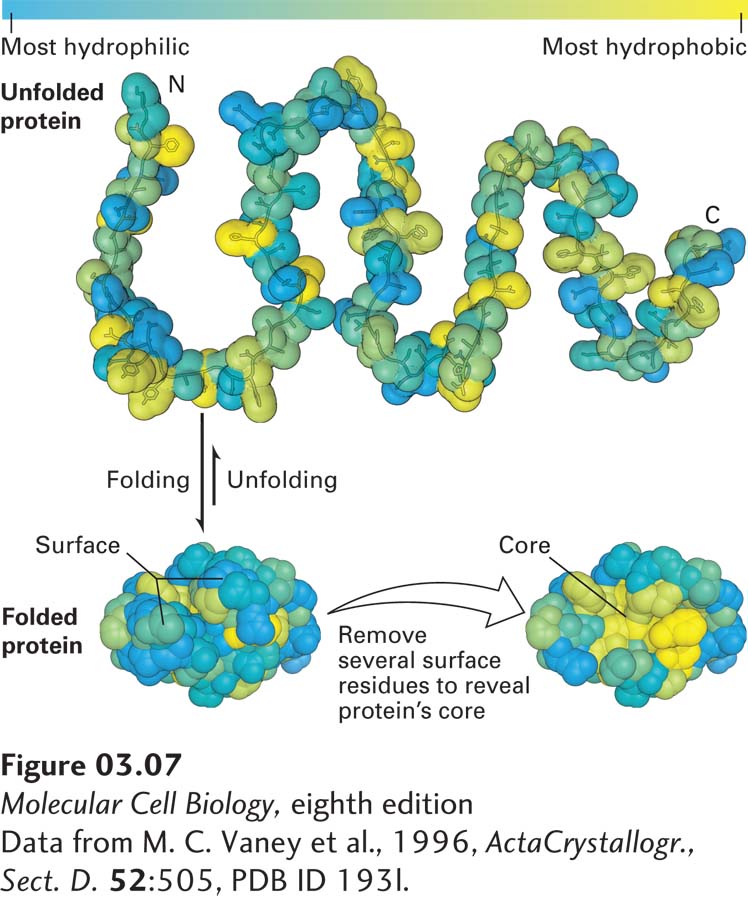
FIGURE 3-7 The oil drop model of protein folding. The hydrophobic and hydrophilic residues of a polypeptide chain can be distributed throughout its linear sequence as illustrated in the unfolded protein (top). The color scale denotes the most most hydrophilic residues (blue) to the most hydrophobic (yellow). When the protein folds (bottom left), hydrophilic (charged and uncharged polar) side chains will often be exposed on the protein’s surface, where they can form stabilizing interactions with surrounding water and ions. In contrast, the hydrophobic residues tend to cluster together in the inner core, somewhat like drops of oil in an aqueous liquid, driven away from the aqueous surroundings by the hydrophobic effect (see Chapter 2). These core residues are more easily seen when several surface residues are removed (bottom right).
[Data from M. C. Vaney et al., 1996, Acta Crystallogr., Sect. D. 52:505, PDB ID 193l.]

