The egg is a highly polarized cell.
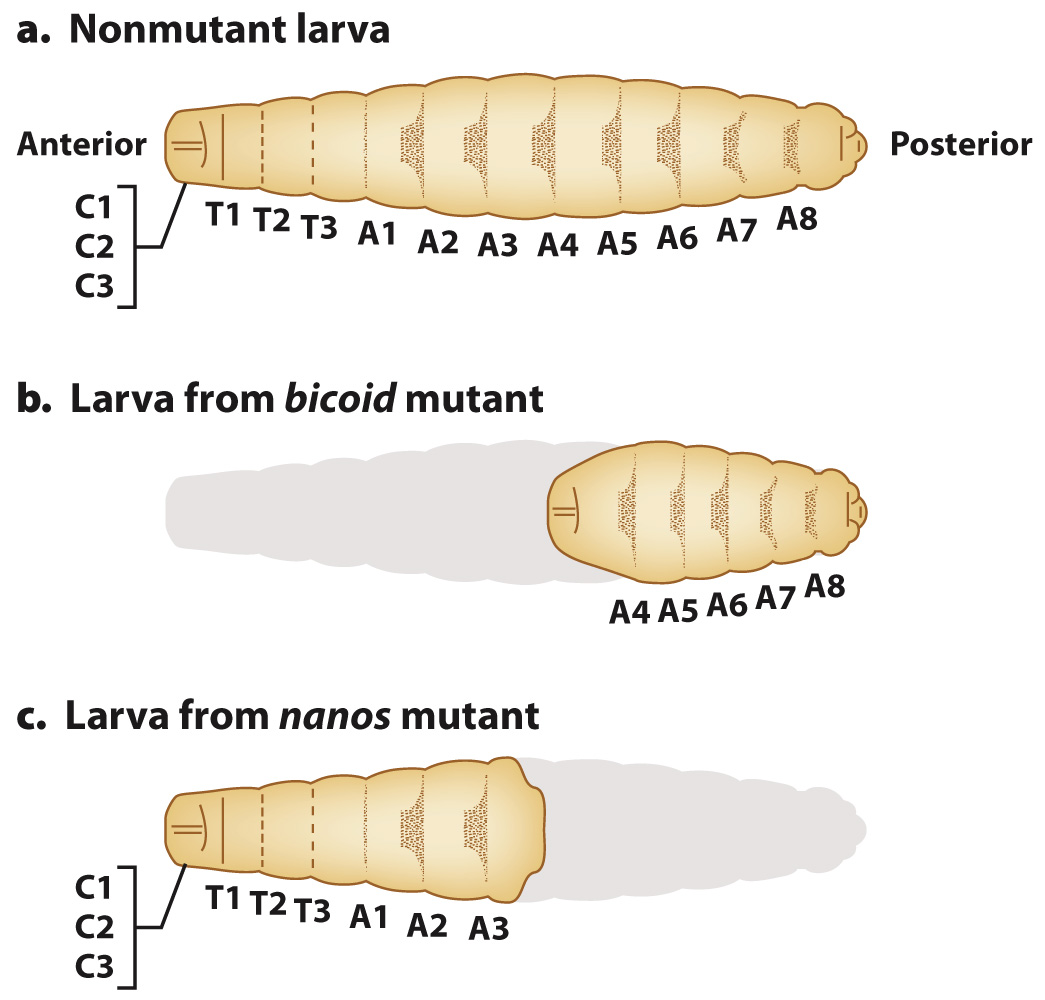
FIG. 20.6 Normal and mutant Drosophila larvae. (a) Nonmutant larvae have anterior, middle, and posterior segments. (b) Bicoid mutant larvae lack anterior segments. (c) Nanos mutant larvae lack posterior structures.
How genes control development in Drosophila was inferred from systematic studies of mutants by Christiane Nusslein-Volhard and Eric F. Wieschaus, work for which they were awarded the 1995 Nobel Prize in Physiology or Medicine. One of their findings was that development starts even before a zygote is formed, in the maturation of the oocyte, the unfertilized egg cell produced by the mother. The oocyte, which matures under control of the mother’s genes, is also important for normal embryonic development. This finding applies not only to insects like Drosophila, but also to many multicellular animals.
Among the striking mutants Nusslein-Volhard and Wieschaus generated and investigated were ones that significantly affected early development (Fig. 20.6). These defects in very early development can be easily seen by the time the mutants reach the larval stage. In one class of mutants, called bicoid, larvae are missing segments at the anterior end. In another class of mutants, called nanos, larvae are missing segments at the posterior end. The mutant larvae are grossly abnormal and do not survive. Nusslein-Volhard and Wieschaus were able to identify each segment that was missing based on each segment’s distinctive pattern of hairlike projections. In Fig. 20.6, the patterns are shown as dark shapes on each segment.
A distinguishing feature of bicoid and nanos mutants is that the abnormalities in the embryo depend on the genotype of the mother, not the genotype of the embryo. The reason the genotype of the mother can affect the phenotype of the developing embryo is that successful development requires a functioning oocyte. In Drosophila and many other organisms, the composition of the egg includes macromolecules (such as RNA and protein) synthesized by cells in the mother and transported into the egg. If the mother carries mutations in genes involved in the development of the oocyte, the offspring can be abnormal. Genes such as bicoid and nanos that are expressed by the mother but affect the phenotype of the offspring (in this case the developing embryo and larva) are called maternal-effect genes.
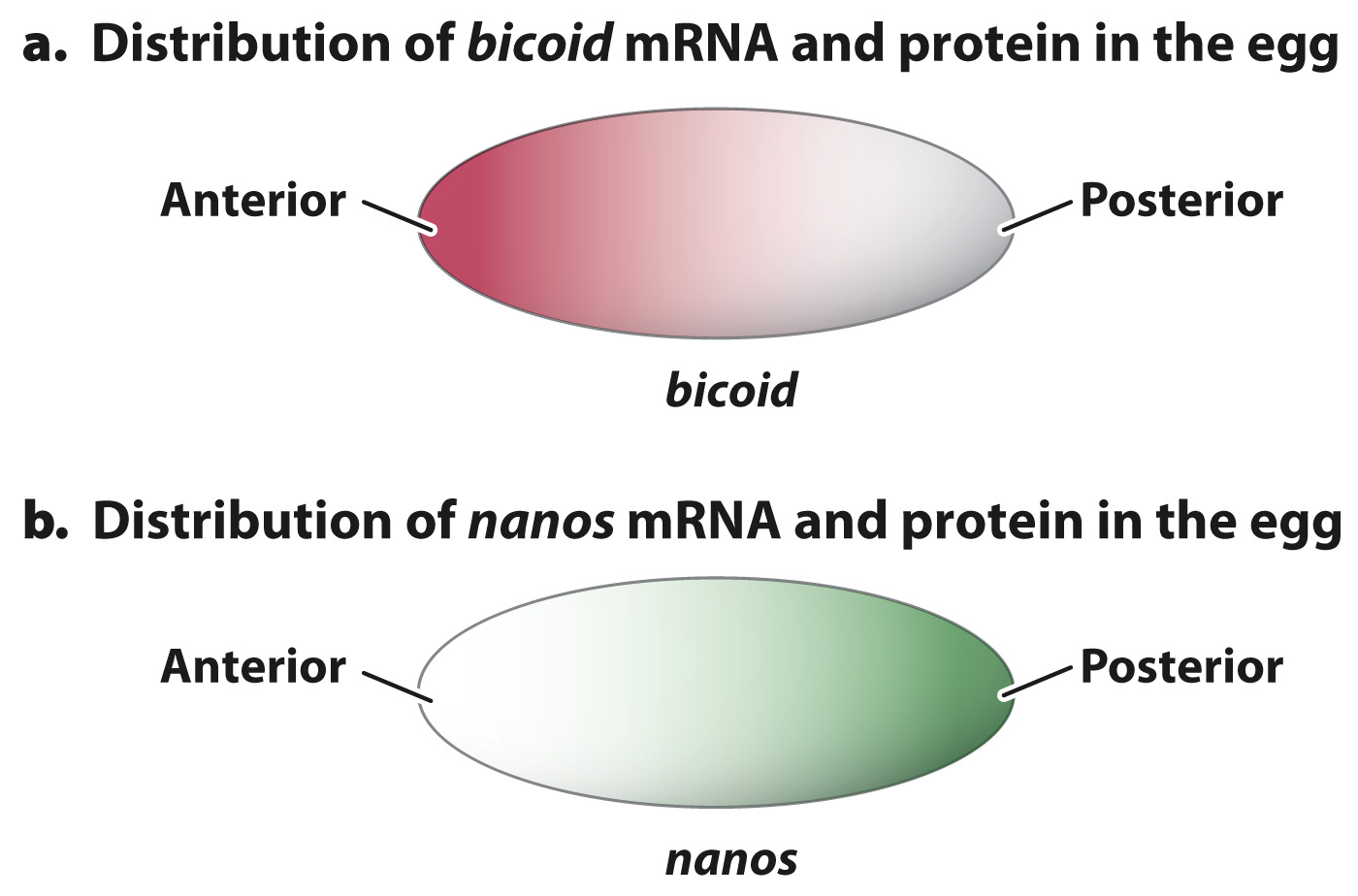
FIG. 20.7 Gradients of bicoid and nanos mRNA and protein in the developing embryo. (a) The mRNA and protein for bicoid are localized in the anterior end of the egg. (b) The mRNA and protein for nanos are localized in the posterior end of the egg.
A normal Drosophila oocyte is highly polarized, meaning that one end is distinctly different from the other. For example, there are gradients of macromolecules that define the anterior–posterior (head-to-tail) axis of the embryo as well as the dorsal–ventral (back-to-belly) axis. The best known of these gradients are those of messenger RNAs that are transcribed from the maternal-effect genes bicoid and nanos. Fig. 20.7a shows the gradient of bicoid mRNA across the oocyte. The bulk of bicoid mRNA comes from the mother, and is localized in the anterior of the egg by proteins that attach them to the cytoskeleton.
The mRNA corresponding to the maternal-effect gene nanos is also present in a gradient, but most of it is at the posterior end (Fig. 20.7b). Like bicoid, mRNA for nanos is synthesized by the mother’s cells and then imported into the oocyte. After fertilization, the zygote produces Bicoid and Nanos proteins from the localized mRNAs, and they have concentration gradients resembling those of the mRNAs in the oocyte (Fig. 20.7).
The anterior–posterior axis set up by the gradients of Bicoid and Nanos proteins is reinforced by gradients of two transcription factors called Caudal and Hunchback (Fig. 20.8). Like the mRNAs for Bicoid and Nanos, the mRNAs for Caudal and Hunchback are transcribed from the mother’s genome and transported into the egg. As shown in Fig. 20.8a, the mRNAs for caudal and hunchback are spread uniformly in the cytoplasm of the fertilized egg. However, the mRNAs are not translated uniformly in the egg. Bicoid protein represses translation of caudal, and Nanos protein represses translation of hunchback (Fig. 20.8b). Caudal protein is therefore concentrated at the posterior end and Hunchback protein is concentrated at the anterior end. The expression of Caudal and Hunchback illustrates gene regulation at the level of translation, discussed in Chapter 19. Bicoid protein is also a transcription factor that promotes transcription of the hunchback gene from zygotic nuclei, which reinforces the localization of Hunchback protein at the anterior end.
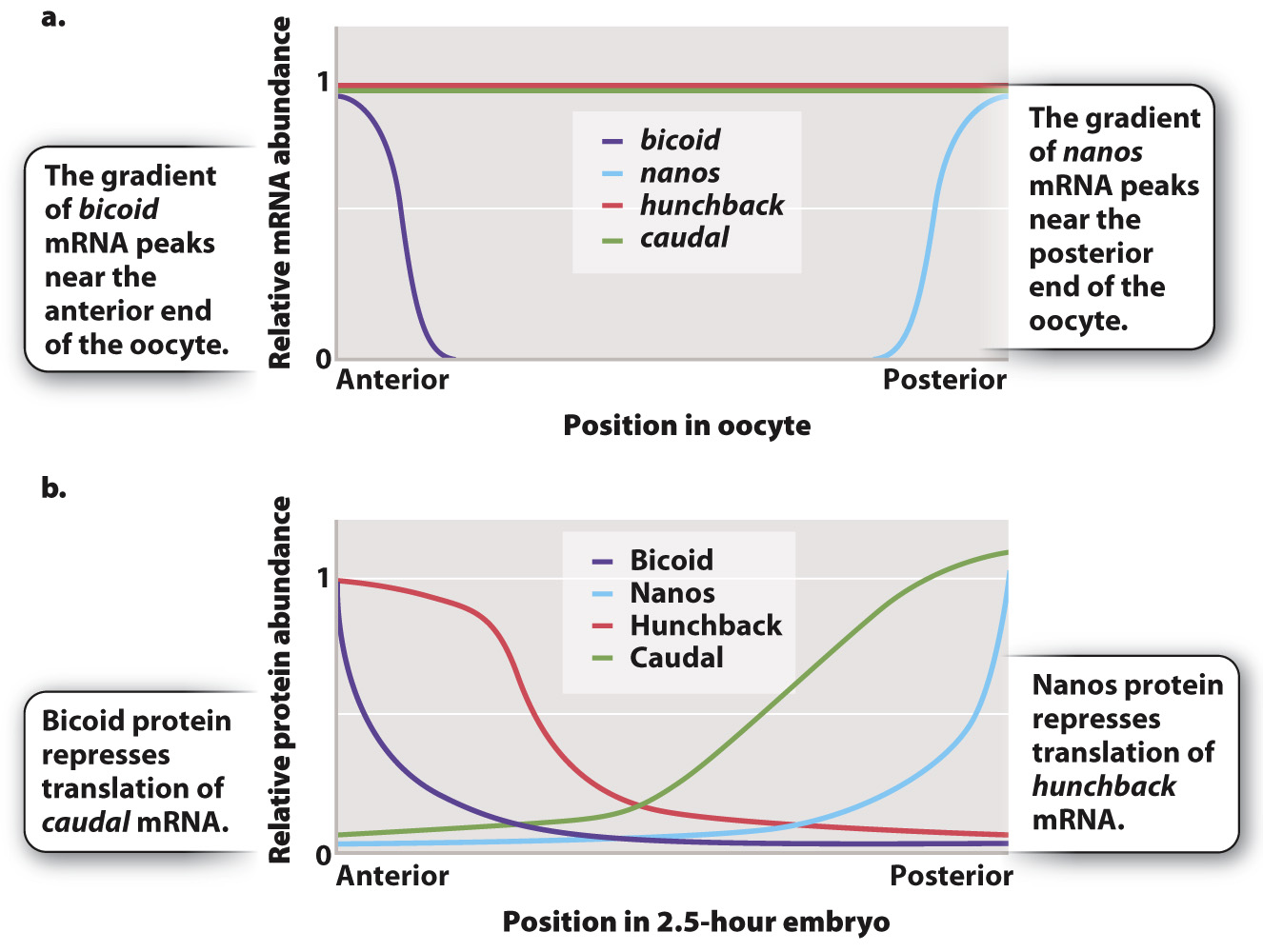
FIG. 20.8 Caudal and Hunchback gradients in the developing embryo. (a) mRNA levels of hunchback and caudal are uniform across the embryo. (b) Hunchback and Caudal protein levels are localized to the anterior and posterior ends of the embryo, respectively, because Bicoid and Nanos control the translation of hunchback and caudal mRNA.
The Hunchback and Caudal gradients set the stage for the subsequent steps in development. The Hunchback transcription factor targets genes of the embryo needed for the development of anterior structures like eyes and antennae, and Caudal targets genes of the embryo needed for the development of posterior structures like genitalia. In this way, maternal genes expressed early in development influence the expression of genes of the embryo that are important in later development. Because the products of the bicoid and nanos mRNA are the ones initially responsible for organizing the anterior and posterior ends of the embryo, respectively, mothers that are mutant for bicoid have larvae that lack anterior structures, and mothers that are mutant for nanos have larvae that lack posterior structures.
Quick Check 3 Would development happen normally if the mother has normal bicoid function, but the embryo does not? Why or why not?
Quick Check 3 Answer
Yes. The embryo would develop normally, even though the embryo does not have normal bicoid function, because bicoid is a maternal-effect gene, in which the mother’s genotype affects the phenotype of the offspring.


