Photorespiration leads to a net loss of energy and carbon.
A second challenge to photosynthetic efficiency is the fact that rubisco can use both CO2 and O2 as substrates. If O2 instead of CO2 diffuses into the active site of rubisco, the reaction can still proceed, although O2 is added to RuBP in place of CO2. An enzyme that adds O2 to another molecule is called an oxygenase. Recall that rubisco is shorthand for RuBP carboxylase oxygenase, reflecting rubisco’s ability to catalyze two different reactions.
When rubisco adds O2 instead of CO2 to RuBP, the result is one molecule with three carbon atoms (3-PGA) and one molecule with only two carbon atoms (2-phosphoglycolate). The production of 2-phosphoglycolate creates a serious problem because this molecule cannot be used by the Calvin cycle either to produce triose phosphate or to regenerate RuBP.
A metabolic pathway to recycle 2-phosphoglycolate is present in photosynthetic cells. A portion of the carbon atoms in 2-phosphoglycolate are converted into 3-PGA, which can reenter the Calvin cycle. However, this pathway is not able to return all of the carbon atoms in 2-phosphoglycolate to the Calvin cycle; some are released as CO2. Because the overall effect is the consumption of O2 and release of CO2 in the presence of light, this process is referred to as photorespiration (Fig. 8.17). However, whereas respiration produces ATP, photorespiration consumes ATP. In photorespiration, ATP drives the reactions that recycle 2-phosphoglycolate into 3-PGA. Thus, photorespiration represents a net energy drain on two accounts: First, it results in the oxidation and loss, in the form of CO2, of carbon atoms that had previously been incorporated and reduced by the Calvin cycle, and second, it consumes ATP.
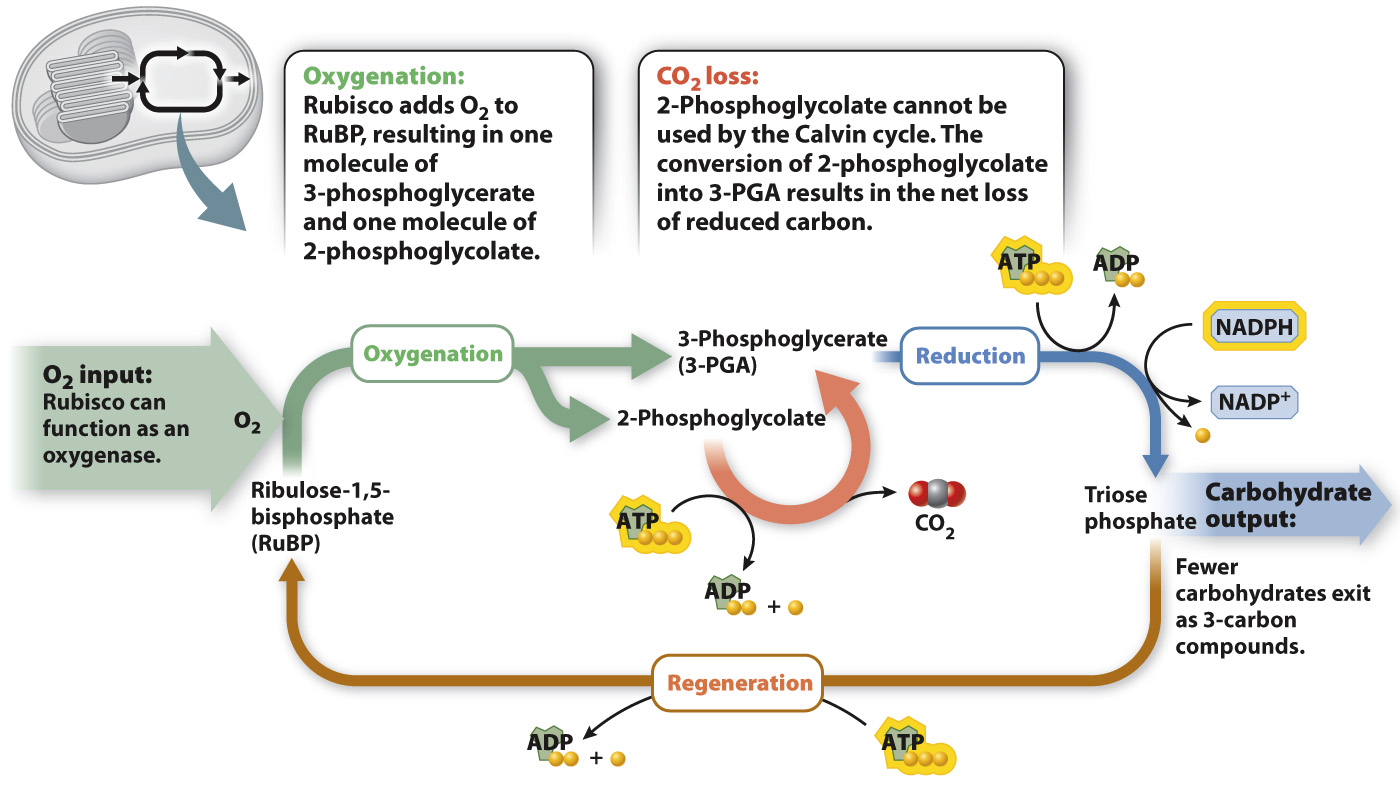
FIG. 8.17 Photorespiration. Carbon and energy are lost when rubisco acts as an oxygenase in photorespiration.
Quick Check 5 In what ways is photorespiration similar to cellular respiration (Chapter 7) and in what ways does it differ?
Quick Check 5 Answer
Like cellular respiration, photorespiration consumes O2 and releases CO2. Unlike cellular respiration, it consumes rather than produces ATP.
The Calvin cycle originated long before the accumulation of oxygen in Earth’s atmosphere, providing an explanation why an enzyme with these properties might have initially evolved. Still, why would photorespiration persist in the face of what must be strong evolutionary pressure to reduce or eliminate the unwanted reaction with oxygen?
The difficulty is that for rubisco to favor the addition of CO2 over O2 requires that the enzyme be highly selective, and the price of high selectivity is speed. CO2 and O2 are similar in size and chemical structure, and for this reason selectivity can only be achieved by rubisco binding more tightly with the transition state (Chapter 6) of the carboxylation reaction. As a result, the better rubisco is at discriminating between CO2 and O2, the slower its catalytic rate.
Nowhere is this trade-off more evident than in land plants, whose photosynthetic cells acquire CO2 from an O2-rich and CO2-poor atmosphere. The rubiscos of land plants are highly selective: If they are exposed to equal concentrations of CO2 and O2, the ratio of CO2 addition to O2 addition is approximately 80:1. As a result, rubisco is a very slow enzyme, with catalytic rates on the order of three reactions per second. To put this rate in perspective, it is not uncommon for metabolic enzymes to achieve a catalytic rate of tens of thousands of reactions per second.
This trade-off between selectivity and speed is a key constraint for photosynthetic organisms. For land plants, rubisco’s low catalytic rate means that photosynthetic cells must produce huge amounts of this enzyme; as much as 50% of the total protein within a leaf is rubisco, and it is estimated to be the most abundant protein on Earth. At the same time, because O2 is approximately 500 times more abundant in the atmosphere than CO2, as much as one-quarter of the reduced carbon formed in photosynthesis can be lost through photorespiration.
Quick Check 6 Why does rubisco have such a low catalytic rate (that is, why is it so slow)?
Quick Check 6 Answer
Rubisco faces a fundamental trade-off between selectivity and speed because it can use both CO2 and O2 as substrates. High selectivity of CO2 over O2 requires that the reaction have a high energy barrier, leading to a lower catalytic rate.
