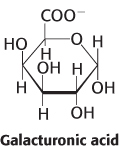
11.2Monosaccharides Are Linked to Form Complex Carbohydrates
Monosaccharides Are Linked to Form Complex Carbohydrates
Because sugars contain hydroxyl groups, glycosidic bonds can join one monosaccharide to another. Oligosaccharides are built by the linkage of two or more monosaccharides by O-glycosidic bonds (Figure 11.11). In the disaccharide maltose, for example, two D-
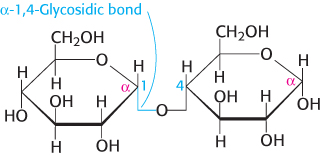
The fact that monosaccharides have multiple hydroxyl groups means that many different glycosidic linkages are possible. For example, consider three monosaccharides: glucose, mannose, and galactose. These molecules can be linked together in the laboratory to form more than 12,000 structures differing in the order of the monosaccharides and the hydroxyl groups participating in the glycosidic linkages. In this section, we will look at some of the most common oligosaccharides found in nature.
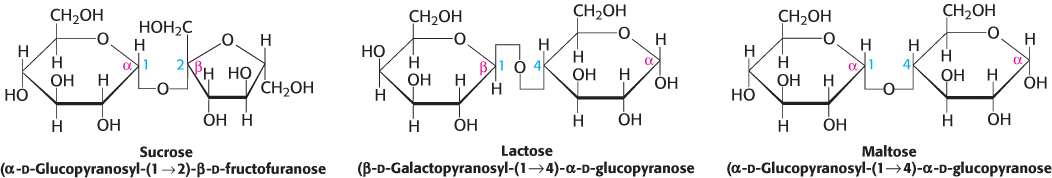
Sucrose, lactose, and maltose are the common disaccharides
A disaccharide consists of two sugars joined by an O-glycosidic bond. Three abundant disaccharides that we encounter frequently are sucrose, lactose, and maltose (Figure 11.12). Sucrose (common table sugar) is obtained commercially from sugar cane or sugar beets. The anomeric carbon atoms of a glucose unit and a fructose unit are joined in this disaccharide; the configuration of this glycosidic linkage is α for glucose and β for fructose. Sucrose can be cleaved into its component monosaccharides by the enzyme sucrase. Lactose, the disaccharide of milk, consists of galactose joined to glucose by a β-1,4-
324
Glycogen and starch are storage forms of glucose
Glucose is an important energy source in virtually all life forms. However, free glucose molecules cannot be stored because in high concentrations, glucose will disturb the osmotic balance of the cell, potentially resulting in cell death. The solution is to store glucose as units in a large polymer, which is not osmotically active.
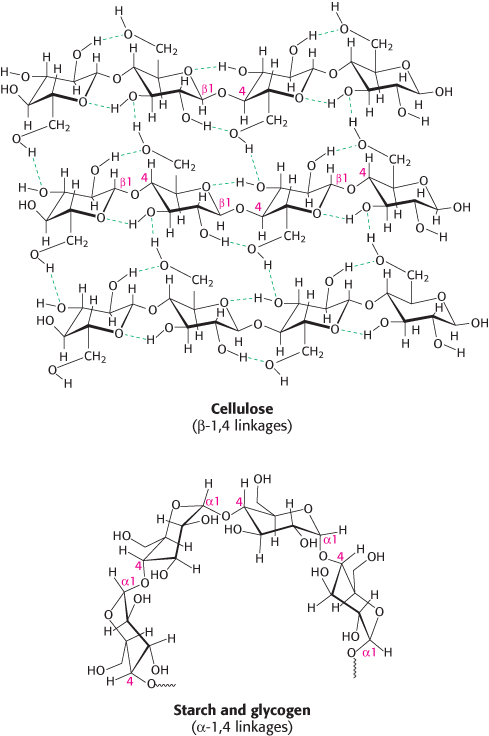
Large polymeric oligosaccharides, formed by the linkage of multiple monosaccharides, are called polysaccharides and play vital roles in energy storage and in maintaining the structural integrity of an organism. If all of the monosaccharide units in a polysaccharide are the same, the polymer is called a homopolymer. The most common homopolymer in animal cells is glycogen, the storage form of glucose. Glycogen is present in most of our tissues but is most abundant in muscle and liver. As will be considered in detail in Chapter 21, glycogen is a large, branched polymer of glucose residues. Most of the glucose units in glycogen are linked by α-1,4-
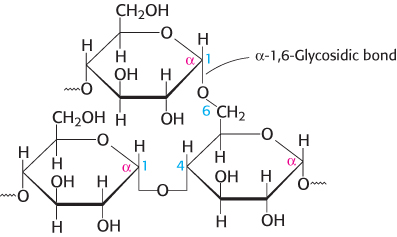
The nutritional reservoir in plants is the homopolymer starch, of which there are two forms. Amylose, the unbranched type of starch, consists of glucose residues in α-1,4 linkage. Amylopectin, the branched form, has about 1 α -1,6 linkage per 30 α -1,4 linkages, in similar fashion to glycogen except for its lower degree of branching. More than half the carbohydrate ingested by human beings is starch found in wheat, potatoes, and rice, to name just a few sources. Amylopectin, amylose, and glycogen are rapidly hydrolyzed by α-amylase, an enzyme secreted by the salivary glands and the pancreas.
Cellulose, a structural component of plants, is made of chains of glucose
Cellulose, the other major polysaccharide of glucose found in plants, serves a structural rather than a nutritional role as an important component of the plant cell wall. Cellulose is among the most abundant organic compounds in the biosphere. Some 1015 kg of cellulose is synthesized and degraded on Earth each year, an amount 1000 times as great as the combined weight of the human race. Cellulose is an unbranched polymer of glucose residues joined by β -1,4 linkages, in contrast with the α-1,4 linkage seen in starch and glycogen. This simple difference in stereochemistry yields two molecules with very different properties and biological functions. The β configuration allows cellulose to form very long, straight chains. Fibrils are formed by parallel chains that interact with one another through hydrogen bonds, generating a rigid, supportive structure. The straight chains formed by β linkages are optimal for the construction of fibers having a high tensile strength. The α-1,4 linkages in glycogen and starch produce a very different molecular architecture: a hollow helix is formed instead of a straight chain (Figure 11.14). The hollow helix formed by α linkages is well suited to the formation of a more-
325
We have considered only homopolymers of glucose. However, given the variety of different monosaccharides that can be put together in any number of arrangements, the number of possible polysaccharides is huge. We will consider some of these polysaccharides shortly.