26.2Cholesterol Is Synthesized from Acetyl Coenzyme A in Three Stages
Cholesterol Is Synthesized from Acetyl Coenzyme A in Three Stages
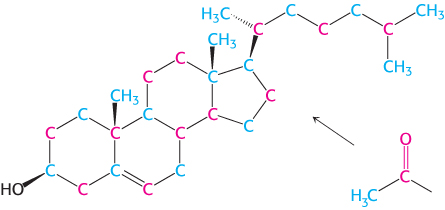
We now turn our attention to the synthesis of the fundamental lipid cholesterol. This steroid modulates the fluidity of animal cell membranes (Section 12.5) and is the precursor of steroid hormones such as progesterone, testosterone, estradiol, and cortisol. All 27 carbon atoms of cholesterol are derived from acetyl CoA in a three-
1. Stage one is the synthesis of isopentenyl pyrophosphate, an activated isoprene unit that is the key building block of cholesterol.
2. Stage two is the condensation of six molecules of isopentenyl pyrophosphate to form squalene.
3. In stage three, squalene cyclizes and the tetracyclic product is subsequently converted into cholesterol.
The first stage takes place in the cytoplasm, and the next two in the endoplasmic reticulum.
The synthesis of mevalonate, which is activated as isopentenyl pyrophosphate, initiates the synthesis of cholesterol
Cholesterol
“Cholesterol is the most highly decorated small molecule in biology. Thirteen Nobel Prizes have been awarded to scientists who devoted major parts of their careers to cholesterol. Ever since it was isolated from gallstones in 1784, cholesterol has exerted an almost hypnotic fascination for scientists from the most diverse areas of science and medicine…. Cholesterol is a Janus-
—Michael Brown and Joseph Goldstein, on the occasion of their receipt of a Nobel Prize for elucidating the control of blood levels of cholesterol.
Nobel Lectures (1985) © The Nobel Foundation, 1985
The first stage in the synthesis of cholesterol is the formation of isopentenyl pyrophosphate from acetyl CoA. This set of reactions starts with the formation of 3-
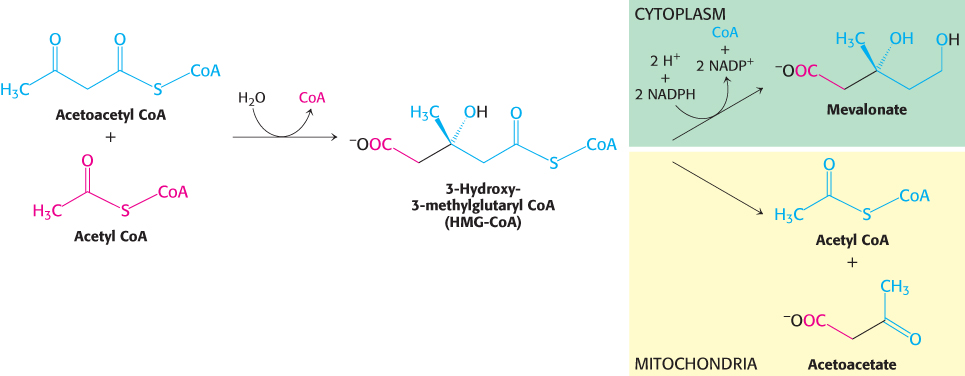
The synthesis of mevalonate is the committed step in cholesterol formation. The enzyme catalyzing this irreversible step, 3-
777
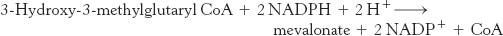
HMG-
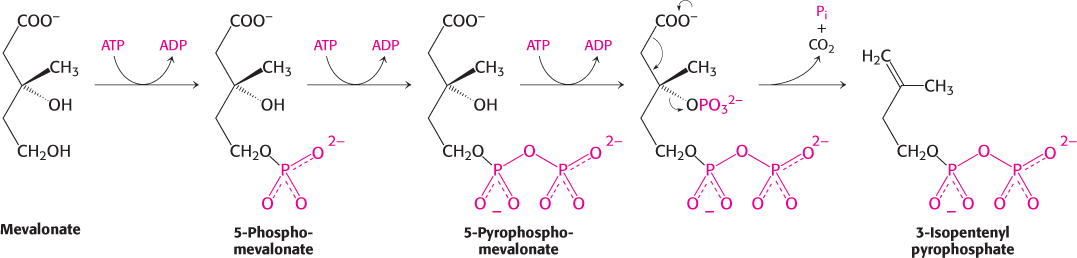
Mevalonate is converted into 3-
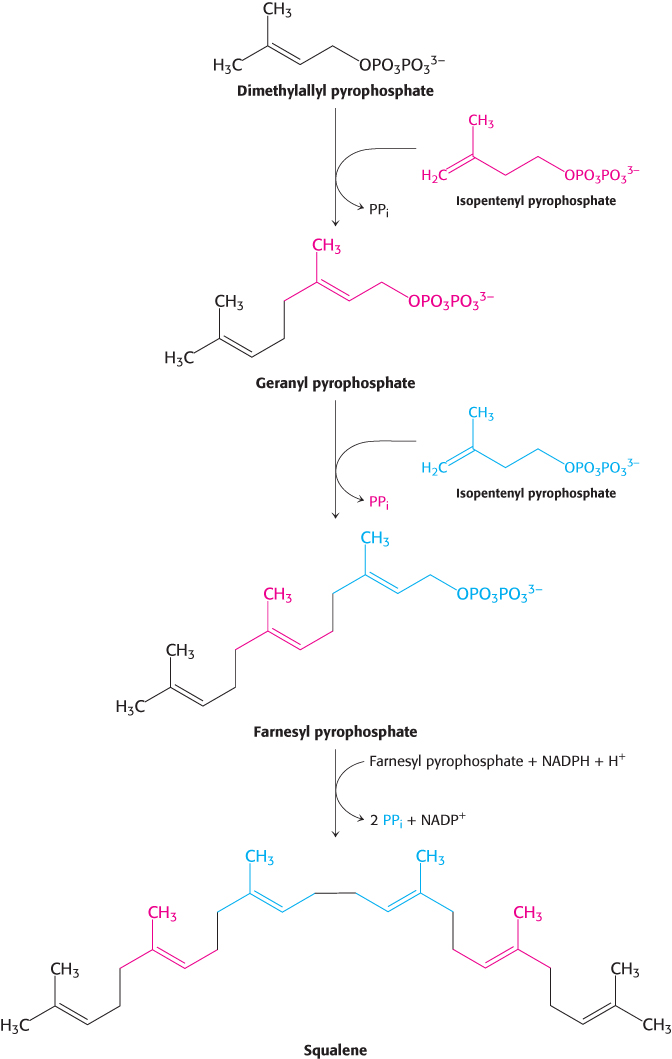
Squalene (C30) is synthesized from six molecules of isopentenyl pyrophosphate (C5)
Squalene is synthesized from isopentenyl pyrophosphate by the reaction sequence
C5 → C10 → C15 → C30
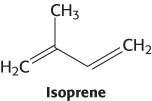
This stage in the synthesis of cholesterol starts with the isomerization of isopentenyl pyrophosphate to dimethylallyl pyrophosphate.
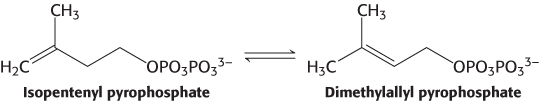
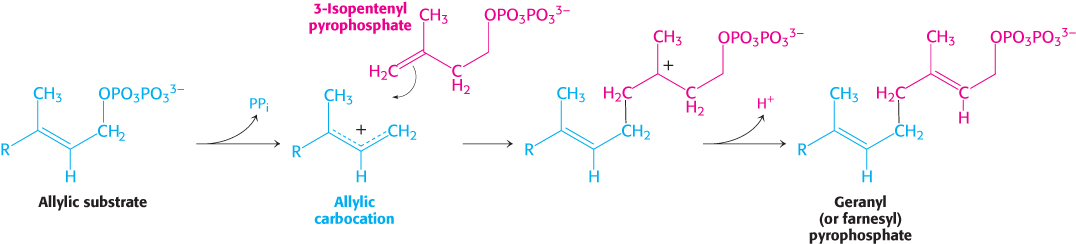
These two isomeric C5 units (one of each type) condense to form a C10 compound: isopentenyl pyrophosphate attacks an allylic carbocation formed from dimethylallyl pyrophosphate to yield geranyl pyrophosphate (Figure 26.10). The same kind of reaction takes place again: geranyl pyrophosphate is converted into an allylic carbonium ion, which is attacked by isopentenyl pyrophosphate. The resulting C15 compound is called farnesyl pyrophosphate. The same enzyme, geranyl transferase, catalyzes each of these condensations.
778
The last step in the synthesis of squalene is a reductive tail-
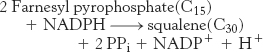
The reactions leading from C5 units to squalene, a C30 isoprenoid, are summarized in Figure 26.11.
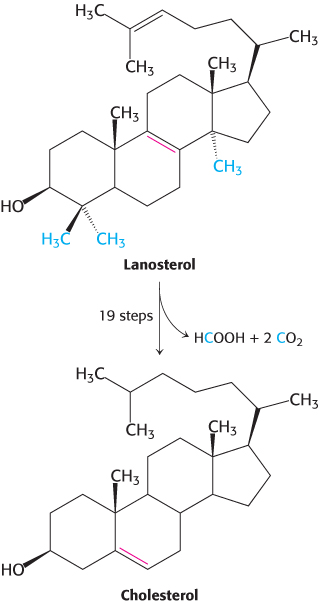
Squalene cyclizes to form cholesterol
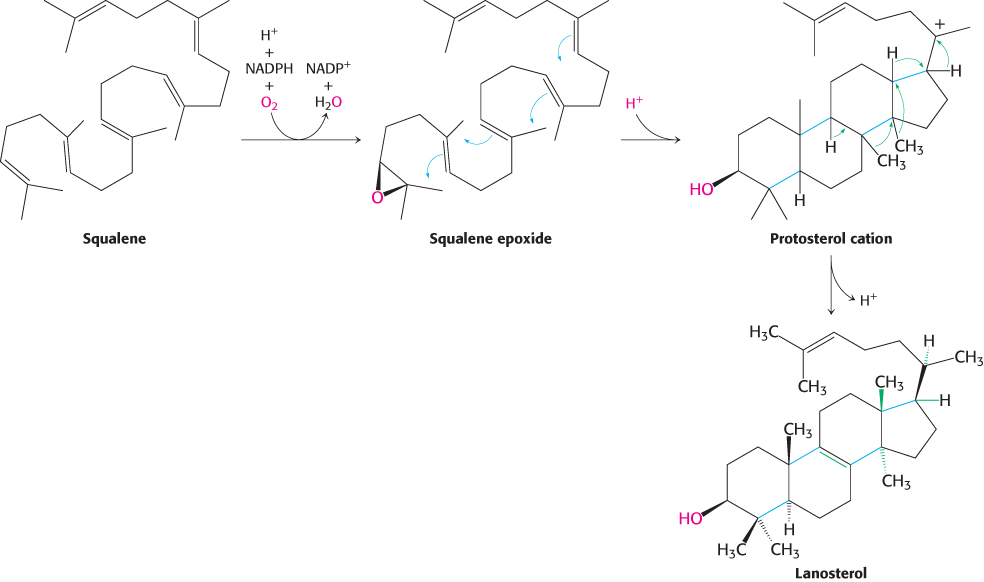
The final stage of cholesterol biosynthesis starts with the cyclization of squalene (Figure 26.12). Squalene is first activated by conversion into squalene epoxide (2,3-
779