13.6Specific Channels Increase the Permeability of Some Membranes to Water
Specific Channels Increase the Permeability of Some Membranes to Water
One more important class of channels does not take part in ion transport at all. Instead, these channels increase the rate at which water flows through membranes. As noted in Section 12.3, membranes are reasonably permeable to water. Why, then, are water-
391
The channels (now called aquaporins) were discovered serendipitously. Peter Agre noticed a protein present at high levels in red-
The structure of aquaporin has been determined (Figure 13.33). The protein consists of six membrane-
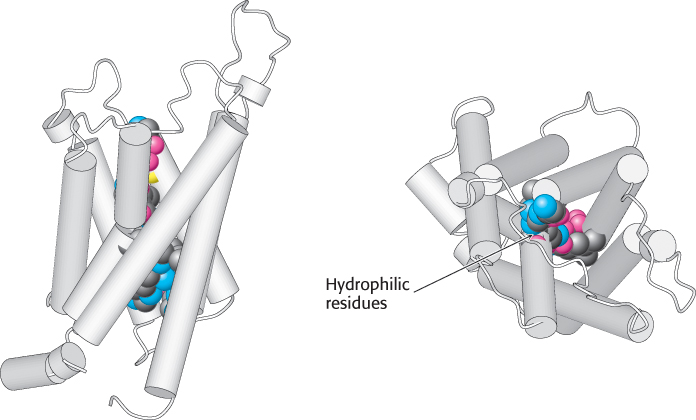
 FIGURE 13.33 Structure of aquaporin. The structure of aquaporin viewed from the side (left) and from the top (right). Notice the hydrophilic residues (shown as space-
FIGURE 13.33 Structure of aquaporin. The structure of aquaporin viewed from the side (left) and from the top (right). Notice the hydrophilic residues (shown as space-